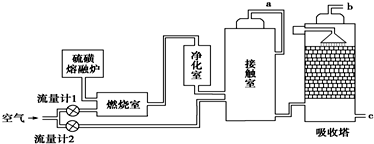
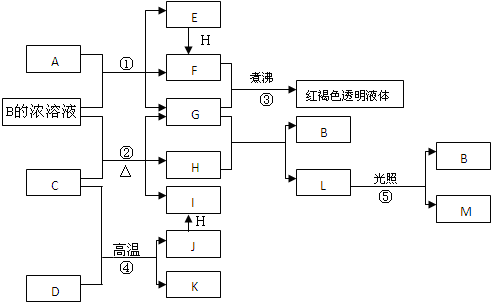
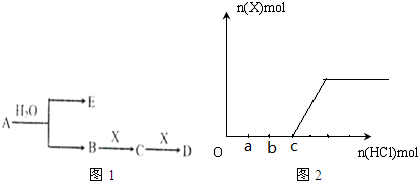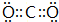��Ŀ����
7����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ���Ҫ�Ե⻯����ʽ���ڣ���һ��ѧ����С���ú���Ϊԭ����ȡ�����ⵥ�ʣ����ǽ��������ճɻң���ˮ����һ��ʱ�䣨���õ⻯�����ܽ���ˮ�У����õ�����������Һ��Ȼ��ͼ1ʵ��������ȡ���ʵ⣺
����ʾ��Cl2��I-�ɷ����û���Ӧ��
��1�����պ���ʱ��Ҫ�õ��IJ���������F��������������ѡ��������������ñ����ĸ��д�ڿհ״�����
A������ B���ձ� C������ǯ D�����ż� E�������� F���ƾ���
��2�����̢��г�������Cl2��Ŀ���ǽ�I-��ȫ����ΪI2��
��Ӧ�����ӷ���ʽΪ2I-+Cl2�TI2+2Cl-��
��3�������������õ��л��Լ�������CCl4������ֻ��һ�֣���
��4���������̢ۿ��Էֽ�Ϊ���¼�����
A����ʢ����Һ�ķ�Һ©����������̨����Ȧ�У�
B����50mL��ˮ��15mL�л��ܼ�������ѡ�������Һ©���У��Ǻò�������
C�������Һ©���������ϿڵIJ������Ƿ�©Һ��
D����ת©����������ʱ����������������رջ������ѷ�Һ©��������
E���������������ձ�������Һ��
F���ӷ�Һ©���Ͽڵ����ϲ�Һ�壻
G����©���ϿڵIJ�������ʹ���ϵİ�����©���Ͽڵ�С����
H�����á��ֲ㣮
a������������������CBDAHGEF
b��G��������Ŀ���DZ��ַ�Һ©������ѹ��ƽ�⣬ʹҺ���������£�
c��������л���Һ��ͨ��©���¿ڻ�ã��©���Ͽڡ���©���¿ڡ�����
��5����Ҫ������ȡ����ˮ��Һ���Ƿ��е��ʵ⣬��ȡ�����������Һ���۲���ɫ�仯��
��6���Ӻ�����л���Һ����ȡ��ͻ����л���Һ������Ҫ�������۲���ͼ2��ʾʵ��װ�ã�ָ���������������
��7�����������������ʱ��ʹ��ˮԡ���ŵ������Ⱦ��ȣ������¶ȣ�
���� ���������գ�Ȼ�����ҽ��ݣ����ù��˷�������õ���������Һ�к���I-������Һ�г�������Cl2��������Ӧ2 I-+Cl2=I2+2Cl-���õ����е��ˮ��Һ�����ˮ�м�����ȡ���õ������л���Һ�������������õ�����ⵥ�ʣ�
��1����������������ѡȡ������������������ת������������ǯ���ô���Ȧ������̨�����żܺ��������Ϸ����������þƾ��ƽ��м��ȣ�
��2�����˺����Һ�к������ӣ����Ա�����Ϊ�ⵥ�ʣ�
��3����ȡ����ѡȡ���ǣ���������ȡ���е��ܽ�ȴ�����ԭ�ܼ��е��ܽ�ȣ����ʺ���ȡ������Ӧ����ȡ����ԭ���ܼ����ܻ��ܣ�
��4��a����ʵ�鲽���Ǽ�©����ҩƷ�������á����á�����������������������Һ�壻
b����������Һ���ڴ���ѹ��������������
c���ϲ�Һ����Ͽڵ������²�Һ����¿�������
��5�����ݵⵥ���������ۻ����ɫ�������Ƿ��еⵥ�ʣ�
��6������ˮӦ���½��ϳ����¶ȼ�ˮ����Ӧ����ƿ֧�ܿ���ƽ��
��7��ˮԡ���ȵ��¶���ʹ�Թ��ڻ��ձ����Լ������¶Ⱦ��ȣ����г�ʱ������¶ȱ��ֺ㶨���ص㣮
��� �⣺��1�����պ���ʱ������ʢ�ź������ô���Ȧ������̨�����żܺ��������Ϸ����������þƾ��ƽ��м��ȣ��������պ���ʱ��Ҫ�õ���ʵ������������������Ȧ������̨�����żܡ������ǡ��ƾ��ƣ�������ǯת������������ʹ���ձ��������Dz���������ֻ�оƾ��ƣ�ѡF��
�ʴ�Ϊ��F��
��2��������������û������ӣ��䷴Ӧ���ӷ���ʽΪCl2+2I-=I2+2Cl-��
�ʴ�Ϊ��ʹI-ת��ΪI2��Cl2+2I-=I2+2Cl-��
��3��������ȡ��CCl�е��ܽ�ȴ�����ԭ�ܼ�ˮ�е��ܽ�ȣ�������Ȼ�̼����Ӧ�����Ȼ�̼��ˮ���ܻ��ܣ����Կ��������Ȼ�̼������ȡ����
�ʴ�Ϊ��CCl4������
��4��a����ʵ�鲽���Ǽ�©����ҩƷ�������á����á�����������������������Һ�壬���������˳����CBDAHGEF��
�ʴ�Ϊ��CBDAHGEF��
b����������Һ���ڴ���ѹ��������������
�ʴ�Ϊ�����ַ�Һ©������ѹ��ƽ�⣬ʹҺ���������£�
c���ϲ�Һ����Ͽڵ������²�Һ����¿�������CCl4 ���ܶȴ���ˮ���ܶȣ��ʴ�©���¿�������
�ʴ�Ϊ��©���¿ڣ�
��5����ȡ����ˮ��Һ�����Ǻ��е��ʵ⣬���������ۻ����ɫ������ȡ������ȡ����ˮ��Һ���Թ��У����뼸�ε�����Һ���۲��Ƿ������ɫ�����������˵�����е��ʵ⣩��
�ʴ�Ϊ��������Һ��
��6������ˮӦ���½��ϳ����¶ȼƲ����������¶ȣ��¶ȼ�ˮ����Ӧ����ƿ֧�ܿ���ƽ���ձ���Ҫ��ʯ�������ȣ��������������
�ʴ�Ϊ������
��7��ˮԡ���ȵ��¶���ʹ�Թ��ڻ��ձ����Լ������¶Ⱦ��ȣ����г�ʱ������¶ȱ��ֺ㶨���ص㣬���Ȼ�̼�е㣺76.8�棬�ⵥ�ʷе�Ϊ184�棬�������̬����������ƿ��ۼ���
�ʴ�Ϊ�����ȱȽϾ��ȣ��¶������ƣ�
���� ���⿼���˺�ˮ��Դ���ۺ����ã�Ϊ��Ƶ���㣬�漰֪ʶ��϶࣬����ʵ�����̼������ķ�Ӧ����ȡ����ѡȡ��������ʵ��ԭ����װ�õ�֪ʶ��Ϊ���Ĺؼ������ظ�Ƶ����Ŀ��飬��Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | �ۢ٢ܢߢ� | B�� | �ڢۢܢޢ� | C�� | �ۢܢڢߢ� | D�� | �ڢܢۢߢ� |
| A�� | �������廯������������������Ӧ��2Fe2++Cl2�T2Fe3++2Cl- | |
| B�� | ��KI��ϡ����Ļ����Һ��ͨ��������4H++O2+4I-�T2I2+2H2O | |
| C�� | ����SO2ͨ��Ca��ClO��2��Һ�У�SO2+H2O+Ca2++2ClO-�TCaSO3��+2HClO | |
| D�� | ̼����þ��Һ�м��������ij���ʯ��ˮ��Mg2++2HCO3-+2Ca2++4OH-�TMg��OH��2��+2CaCO3��+2H2O |
| A�� | Cu��OH��2�����ᣬCu��OH��2����� | |
| B�� | BaCl2��Һ��Na2SO4��Һ��Ba��OH��2��Һ��H2SO4��Һ | |
| C�� | NaHCO3��Һ��NaHSO4��Һ��NaHCO3��Һ������ | |
| D�� | ʯ��ʯ��ϡ���ᷴӦ����ʯ����ϡ���� |
| A�� | A��ƽ����Ӧ����Ϊ 0.1 mol/��L•min�� | |
| B�� | ƽ��ʱ��C��Ũ��Ϊ 0.125 mol/L | |
| C�� | ƽ��ʱ��B��ת����Ϊ 20% | |
| D�� | ƽ��ʱ��������ѹǿΪԭ���� 0.8 �� |
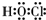 ����Ӧ�ڵĻ�ѧ����ʽΪPbO2+4HCl��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$PbCl2+Cl2��+2H2O��
����Ӧ�ڵĻ�ѧ����ʽΪPbO2+4HCl��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$PbCl2+Cl2��+2H2O��