��Ŀ����
(12��)����ѧ������ѧ�뼼����
��Դ�����������Ʊ�����ũҵ�����ȶ��벻����ѧ����ش��������⣺
��1����ҵ�Ʒ���ʱ����������Ӧ��������Ҫ�ڻ�����м��뱥��ʳ��ˮ�����뱥��ʳ��ˮ��Ŀ���� ��
��2��Al2O3���۵�ߴ�2050oC����ҵ��Ϊ�˽����������ģ��ڽ�������ұ����ͨ����ȡ�Ĵ�ʩ�� ��
��3����ҵ�Ϻϳɰ�����ĵ�����Դ�� ��������Դ�� ��д����ҵ���ڴ�����������ȡ����������һ����ѧ����ʽ ��
��4����ҵ������ʱ��SO3���������� (���豸����)�н��еģ���ҵ�ϳ�����Ũ��������SO3������ֱ����ˮ���յ�ԭ���� �����������У�Ϊ���SO3������������ȡ�Ĵ�ʩΪ ��
��1��������ʹ�����ӻ��Һ�з����������2�����������м������ʯ�����ͻ������۵㡣��3��������ˮ��̼�⻯���CH4+H2O CO+H2��CH4+2H2O
CO+H2��CH4+2H2O CO2+2H2����4���Ӵ��ң���ֹ�γ�������Ӱ��SO3������Ч�ʡ��������²�ͨ��SO3�������ϲ�����ŨH2SO4��SO3��ŨH2SO4�����ϱ���Ӵ��������ա�
CO2+2H2����4���Ӵ��ң���ֹ�γ�������Ӱ��SO3������Ч�ʡ��������²�ͨ��SO3�������ϲ�����ŨH2SO4��SO3��ŨH2SO4�����ϱ���Ӵ��������ա�
������������� ��1����ҵ�Ʒ���ʱ�����뱥��ʳ��ˮ��Ŀ����������ʹ�����ӻ��Һ�з����������2��Al2O3���۵�ߴ�2050oC����ҵ��Ϊ�˽����������ģ��ڽ�������ұ����ͨ����ȡ�Ĵ�ʩ�����������м������ʯ�����ͻ������۵㡣��3����ҵ�Ϻϳɰ�����ĵ�����Դ�ڿ�����������Դ��ˮ��̼�⻯�����ҵ���ڴ�����������ȡ�����Ļ�ѧ����ʽΪCH4+H2O CO+H2��CH4+2H2O
CO+H2��CH4+2H2O CO2+2H2����4����ҵ������ʱ��SO3���������ڽӴ����н��еģ���ҵ�ϳ�����Ũ��������SO3������ֱ����ˮ���յ�ԭ���Ƿ�ֹ�γ�������Ӱ��SO3������Ч�ʡ����������У�Ϊ���SO3������������ȡ�Ĵ�ʩΪ�������²�ͨ��SO3�������ϲ�����ŨH2SO4��SO3��ŨH2SO4�����ϱ���Ӵ��������ա�
CO2+2H2����4����ҵ������ʱ��SO3���������ڽӴ����н��еģ���ҵ�ϳ�����Ũ��������SO3������ֱ����ˮ���յ�ԭ���Ƿ�ֹ�γ�������Ӱ��SO3������Ч�ʡ����������У�Ϊ���SO3������������ȡ�Ĵ�ʩΪ�������²�ͨ��SO3�������ϲ�����ŨH2SO4��SO3��ŨH2SO4�����ϱ���Ӵ��������ա�
���㣺���黯ѧ�뼼�����漰��������ȡ������ұ�����ϳɰ������Ṥҵ��

[��ѧ��ѡ��2��ѧ�뼼��](15��)
��1�����й��ڹ�ҵ����˵����ȷ���� ��������ţ�
| A���ں����Ƽҵ�У����Ȼ�����Һ����ͨ������̼����ͨ���� |
| B�������Ṥҵ���ϳɰ���ҵ�����Ṥҵ�У��Բ���ѭ���������ԭ�������� |
| C�����ȼҵ�����۱����ӽ���Ĥ���������Һ������� |
| D����ҵ�ϲ��õ�������Ȼ����ķ�����ȡ������ |
��2���ҹ��涨����ˮ�������涨��������±���Ҫ��
| pH | Ca2+ ��Mg2+��Ũ�� | ϸ������ |
| 6.5��8.5 | �� 0.004 5 mol��L-1? | ��100����mL-1? |

��ԭˮ�к�Ca2+ ��Mg2+ ��HCO3-��Cl-�ȣ�����ʯ������Ca(OH)2�������������ɸ��ֽⷴӦ��д�����е����ӷ���ʽ��ֻҪ��д���������� �� ��
��FeSO4��7H2O�dz��õ����ۼ�������ˮ���������� ������ͨ�������̼��Ŀ���� �� ��
������A�������� ��ͨ��������Ca(ClO)2���A������������ ͬ��������Ϊ����A�Ĵ���Ʒ�����ţ���ѡ���ۣ���
a��ClO2 b��Ũ��ˮ c��K2FeO4 d��SO2
�ҹ��зḻ����Ȼ����Դ������Ȼ��Ϊԭ�Ϻϳ����ص���Ҫ������ͼ��ʾ(ͼ��ijЩת�����輰������δ�г�):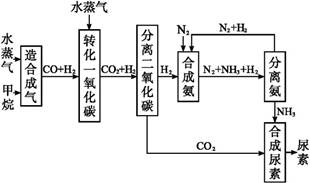
����д���пհ�:
(1)��֪0.5 mol�����0.5 molˮ������t�桢p kPaʱ,��ȫ��Ӧ����һ����̼������(�ϳ���),������a kJ�������÷�Ӧ���Ȼ�ѧ����ʽ����������������
(2)����������,��ҵ�Ϸ���H2��CO2�����ķ�������������
| A���������ͨ������������Һ,������Һ�м������� |
| B���������ѹ��ȴ,ʹCO2Һ�� |
| C��������ð�ˮϴ�� |
| D���������ͨ��ʯ�ҽ���,Ȼ��������չ��� |
(4)������������Դ����������߾���Ч��,Ҳ�Ƕ���ᡢ��ȫ���ฺ��ı���,�����߶κͼ�ͷ����ͼ�е���������������Դ�����
��ҵ������п����Ҫ�ɷ�ΪZnS��������CdS��Fe2O3�����ʣ�Ϊԭ������ZnSO4��7H2O�Ĺ����������£�����֪Cd�Ľ�����Խ���Zn��Fe֮�䣩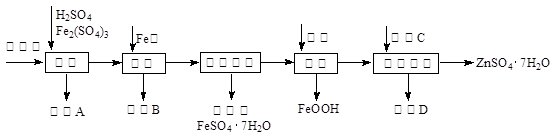
��1��������A�пɻ��һ�ֵ���ɫ�ǽ������ʵĸ���Ʒ���仯ѧʽΪ ��
��2����ȡ������Fe2(SO4)3�������� ����ȡʱFe2(SO4)3��ZnS������Ӧ�Ļ�ѧ����ʽΪ ��
��3���������̿�����Һ��pH��5.4���ң��÷�Ӧ�����ӷ���ʽΪ ���ù����ڿ�����ڴ������һ��������ԡ��ͷ��װ�ã���Ŀ���� ��
��4���û������ؽ���������Cd2+����������CΪ ��
��5������п���ܽ�����¶�֮��Ĺ�ϵ���±���
| �¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 |
| �ܽ��/g | 41.8 | 54.1 | 70.4 | 74.8 | 67.2 | 60.5 |
�ӳ��ؽ����������п��Һ�л������п�����ʵ�����Ϊ �� �����ˡ����
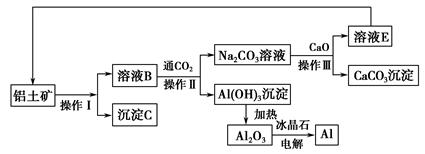
�ҹ����������ձ��ȹ��Ҷ������Ƴ�һ���մɲ��ͻ������ֲ��ͻ��ķ���������������������һ�������Ҳ��״��ȵIJ���������ģ����ֲ�����(����)��
| A���������մ� | B���������մ� |
| C�����ά | D�������� |



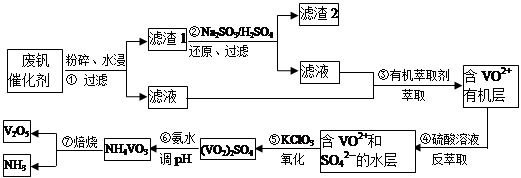
 VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________��
VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________��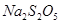 ��������ʳƷƯ�������Ʊ������������£�
��������ʳƷƯ�������Ʊ������������£�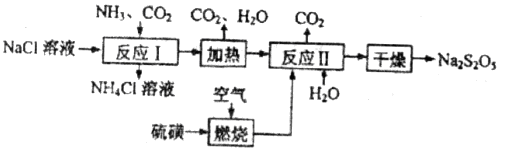
 �ȶಽ��Ӧ��
�ȶಽ��Ӧ��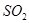 �������ӷ���ʽΪ___________��
�������ӷ���ʽΪ___________��