��Ŀ����
����ĺ�ˮ����Լ��1.4��1018t��������������Դ�⡣
��1����ͼ���ú�ˮ�õ���ˮ�ķ���Ϊ ��
��2�����������ǽ��귢չ������һ�ֽϺõĺ�ˮ������������ԭ������ͼ��a�ǵ�Դ�� ��������ų����� �����ˮ�� ��Ũˮ������
��3����ˮ�������Ũˮ�к������η֣���Ҫ����Mg2����Ca2+��Fe3����SO42����������ˮ�л�ı�ˮ�ʣ��ŵ������лᵼ�������μ���ʲ���ֱ���ŷţ��������ȼҵ������
���ǰ��Ҫ��Ũˮ���ƣ������Լ���Ҫ������HCl��NaOH��BaCl2��Na2CO3�ȣ�������HCl��������Ҫ�� ��
��4�����Ǻ˷�Ӧ����Ҫ��ȼ�ϣ�����������ֱ�ӹ�ϵ��һ�����Һ˹�ҵ��������ķ�չˮƽ����ˮ������UCl4��ʽ���ڣ���������ʽ���ڣ���ÿ�ֺ�ˮֻ��3.3�����ˣ���ˮ���������������൱���ٹ�������̽����ˮ���˵ķ��������ڣ��Ѿ����Ƴɹ�һ�����������ӽ�����֬����ר��������ˮ�е��ˣ�������������Ԫ�ء��䷴Ӧԭ��Ϊ��___________________________����֬��HR���棩���������ӽ���������ӽ���Ĥ���ᴦ�������������õ����˵���Һ���䷴Ӧԭ��Ϊ��________________________��
��5�����ӽ�����֬�����Ʊ���ˮ��ȥ����ˮ�� ����Ҫ������ij�����ӽ��������ľֲ��ṹ��д�ɣ���ͼ�����������ӽ�����֬���ɵ��屽��ϩ�ͽ������Զ���ϩ�����ۺϺ��پ� ��Ӧ�õ��ġ�����ˮ������ӽ�����֬�������ú��� ������ԡ��������ԡ������ԡ�����
��6���й���������ˮ���������й涨��ˮ����Ӳ�Ȳ��ܹ������Ӳ�ȹ������ú�����彡�����ճ�������һ��Ӱ�졣��ʱӲˮ��Ӳ������ ���������ӷ��ţ�����ģ��� ����������ƣ���ɱ�ȥ��������Ӳˮ��Ӳ�ȿ������ӽ�����ȥ����
(1)����1�֣�
(2)����1�֣� ��ˮ��1�֣�
(3)��ȥ������Na2CO3����������Һ��pHֵ��1�֣�
(4)4HR��U4��=UR4��4H�� UR4��4H��=4HR��U4�� ��2�֣�
��5���ǻ���ȡ����1�֣� ���ԣ�1�֣���6��HCO3����1�֣� ������У�1�֣�
���������������1������̫����ʹˮ������Ϊ����
(2)a������������Ϊ��Դ�ĸ���������ų����ǵ��������������ˮ���ǵ�ˮ��
��3��HCl����������Na2CO3��������Һ��pHֵ�����ԣ�
��4��U4���滻HR�е�H�������ᴦ��ʱH�����滻U4����
��5������������˱����ϵ�H������ȡ����Ӧ��ǻ���Ӧ������ˮ������ӽ�����֬�������ú�����H�������ԣ�
��6����ʱӲˮ��Ӳ������HCO3�����𣬼���������ɳ�������ȥ��
���㣺���⿼�麣ˮ�ĵ��������ʵ�ȥ�����л���Ӧ���͡�

��ˮ��Դ���ۺ�����ʮ����Ҫ������Ҫͨ����ѧ�仯���ܹ��Ӻ�ˮ�л�õ�������
| A��ʳ�Ρ���ˮ | B���ơ�þ | C���ռ���� | D���塢�� |
�ݿ�ѧ��Ԥ�⣺�ٹ�100���ȫ�����¹��ƽ�������Լ1.4��5.8 �档������һԤ�⣬ȫ��������������ȫ���������ɹ����Ӱ�죬����ˮ��Դ���ѷ���������塣��ˮռ�����ܴ�ˮ����97.2%�����Ѻ�ˮ�����ͻ�������������������ܽ����ˮ��Դȱ�������⣬���ܳ�����ú�����Դ��
��1����Ŀǰ�������ԣ������Դ���ĵ����⣬���������ڡ���ˮ�������ļ����� ������ţ���
| A������ | B������������ | C����ᷨ�� | D�����ӽ�������E�������� |
��3����ˮɹ�εõ���ĸҺ�У����д�����þ�������Ӻ�һ�������塢�⻯�������ͨ���Ƚ��ķ��뼼���õ�MgCl2��6H2O��Ʒ���˲�Ʒ����Ҫ�ڲ���ͨ�롰�����Ȼ��⡱����������ˮ���ܵõ���ˮMgCl2��ԭ���� ��
���ʵ���Ժ�ˮ��������Ϊ����ԭ�����Ƶõ����塢�⣬������IJ��������� �� �� ��
��4���ѱ���Ϊ21���ͽ����������ܶ�С��ǿ�ȴ�����һ���ǿ�ᡢǿ����������ܣ��㷺���ں��ա������Լ�����ҽѧ������ҵ�Ͽ�����Mg���»�ԭTiCl4���Ƶá�����ƺ�����ʵ�����������������Ӧ����õ����ѣ������������̣� ��
��������ۺ����ü������ڽ�Լ��Դ���������ڱ���������ʵ�������÷Ͼɻ�ͭ(Cu��Zn�Ͻ𣬺���������Fe)�Ʊ���������(CuSO4��5H2O)��������ZnO���Ʊ�����ͼ���£� 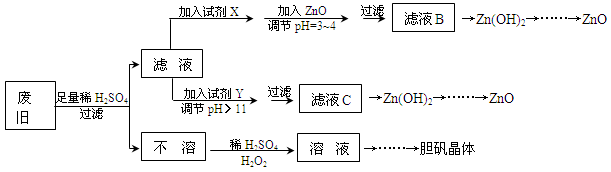
��֪��Zn���������������Al����������������ƣ�pH��11ʱZn(OH)2������NaOH��Һ����[Zn(OH)4]2�����±��г��˼������������������������pH(��ʼ������pH����������Ũ��Ϊ1.0mol��L��1����)��
| | Fe3�� | Fe2�� | Zn2�� |
| ��ʼ������pH | 1.1 | 5.8 | 5.9 |
| ������ȫ��pH | 3.0 | 8.8 | 8.9 |
��ش��������⣺
��1���Լ�X������__________����������____________________��
��2������ZnO����pH=3��4��Ŀ����____________________��
��3���ɲ�����������ҺD�Ļ�ѧ����ʽΪ______________________________��
��4������ҺD�Ƶ��������������Ҫ����������______________________________��
��5�������Լ�����ΪY�Լ�����______��
A��ZnO B��NaOH C��Na2CO3 D��ZnSO4
������ҺC����μ�������ֱ���������������������______________________________��
��6���ⶨ��������Ĵ���(��������I��������Ӧ������������)��ȷ��ȡ0.5000g��������������ƿ�У�������ˮ�ܽ⣬�ټ������KI����0.1000mol��L��1Na2S2O3����Һ�ζ����յ㣬����Na2S2O3����Һ19.40mL����֪�������ζ������е����ӷ���ʽ���£�
2Cu2����4I��
 2CuI(��ɫ)����I2��I2��2S2O32��
2CuI(��ɫ)����I2��I2��2S2O32�� 2I����S4O62��
2I����S4O62���ٵ�������Ĵ���Ϊ_______________��
���ڵζ������о���ҡ��(��Һ���⽦)��ƿ��������õĴ��Ƚ���__________(�ƫ�ߡ�����ƫ�͡����䡱)��
��ҵ���������������ͼ���£���ش��������⣺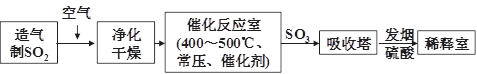
��1���������������Ի�����Ϊԭ�ϣ������ڹ����������������Ϊԭ�ϣ������� ��
��2��������������Ӧ��ǰ�辻����ԭ���� ��
��3���ڴ���Ӧ����ͨ��ʹ�ó�ѹ���ڴ�������SO2��ת����Ϊ90%�����Dz��ַ�����Ҳ�ȡ��ѹ��������ȡSO3����ȡ��ѹ��ʩ��Ŀ�ij��˼ӿ췴Ӧ�����⣬������ ���Ӷ��������Ч�ʡ�
��4����ҵ�����г��ð����ᷨ����β�������Դﵽ������Ⱦ���������õ�Ŀ�ġ��û�ѧ����ʽ��ʾ�䷴Ӧԭ���� ��
��5�������Ṥҵ�⣬�������ҵ������������صĹ�ҵ������������ȷ���� ��
A����ˮ���壺��ˮŨ�� ������ ������ Һ�� Һ�� |
B����ˮ��þ����̲���� ʯ��ˮ ʯ��ˮ MgO MgO þ þ |
C����ҵ��������� NO2 NO2 �����β������ �����β������ |
D����ҵ�ϳɰ�����Ȼ�� ���� ���� NH3��H2��N2 NH3��H2��N2 �� �� |
����þ����Ҫ�ɷ�ΪMgCO3������ȼ��������þ�Ĺ����������£�
��1����������ͼ���Եó��Ľ���Ϊ �� ��
ͼ1 25��ʱMgOˮ����ʱ��仯X����������ͼ 
ͼ2 90��ʱMgOˮ����ʱ��仯X����������ͼ
��2��ˮ����ӦMgO+H2O = Mg(OH)2���Է����е�ԭ���� ��
��3�����Ԫ�������ɺͱ�1��֪�����������������ȷֽ�Ĺ����� ����дһ�����ɣ�
��1 ��������Ԫ�صĽ������������ȷֽ��¶�/��
| LiOH | NaOH | KOH | Al(OH)3 | Mg(OH)2 | Ca(OH)2 | Ba(OH)2 |
| 924 | ���ֽ� | ���ֽ� | 140 | 258 | 390 | 700 |
��4����֪�Ȼ�ѧ����ʽ��Mg(OH)2 (s) =" MgO" (s)+H2O (g) ��H =" 81.5" kJ��mol��1
��Mg(OH)2����ȼ���õ���Ҫԭ���� ��
���볣��±ϵ�����������飩���л���ϵ����������������ȼ����ȣ�Mg(OH)2��ȼ�����ŵ��� ��
[��ѧ��ѡ��ѧ�뼼��]��15�֣�
��ˮ����þ(MgSO4��7H2O)��ӡȾ����ֽ��ҽҩ�ȹ�ҵ�϶��й㷺��Ӧ�ã����û�����������ɰ�ķ�������þ�����ȡ��ˮ����þ����þ�����Ҫ�ɷ���MgCO3����������������(MgO��SiO2��Fe2O3��FeO��CaO��Al2O3��MnO�ȣ���
��1 ����������������������ʽ��ȫ����ʱ��Һ��pH
| ������ | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Mn(OH)2 | Mg(OH)2 |
| pHֵ | 5.2 | 3.2 | 9.7 | 10.4 | 11.2 |
��2 �����ε��ܽ��(��λΪg��100gˮ)
| �¶�/�� | 10 | 30 | 40 | 50 | 60 |
| CaSO4 | 0.19 | 0.21 | 0.21 | 0.20 | 0.19 |
| MgSO4��7H2O | 30.9 | 35.5 | 40.8 | 45.6 | / |
��þ����ȡ��ˮ����þ�Ĺ����������£�

������������ͼ���ο�����pH���ݺ��ܽ�����ݣ��Իش��������⣺
��1������I����Һ�м�����þ�࣬������Һ��pH��5��6���ټ���NaClO��Һ������У�����Һ�е� Mn2+������MnO2����Ӧ�����ӷ�Ӧ����ʽΪ ��������е���ҪĿ�� �� ��
��2������B�г�MnO2��SiO2����� (�ѧʽ)�����ʡ�
��3��������ˢ�����Һ���Ƿ���Fe3+��ʵ�鷽���� ��
��4������C�Ļ�ѧʽ�� ������III����ȹ��˵�������