��Ŀ����
��ૼ������ȵõ�����ͪ���������������ϣ��ɵõ��˼������ӿ���ϩ���й�ʵ��ԭ�����������£� 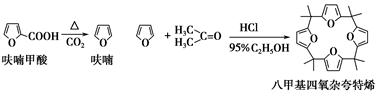
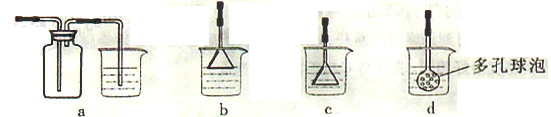
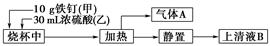
����1����Ʊ�
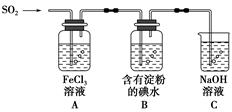
��Բ����ƿ�з���4.5 gૼ��ᣨ100 ��������ૼ�����133 �����ڣ�230��232 ����ڣ����ڴ��¶������ȣ�����ͼ��װ���������ȴ�����ʹૼ�������ۻ���Ȼ����ڼ���ǿ�ȣ��������У���ૼ������ȷ�Ӧ��ϣ�ֹͣ���ȡ�����ɫҺ��ૣ��е㣺31��32 �棬������ˮ����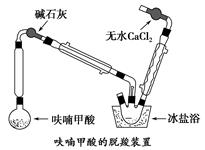
����2��������˼������ӿ���ϩ�ĺϳ�
��25 mL��ƿ�м���2.7 mL 95%�Ҵ���1.35 mLŨ���ᣬ���ȣ��ڱ�ԡ������5 �����£�Ȼ��3.3 mL��ͪ��1.35 mLĻ��ҺѸ�ٵ�����ƿ�У���ֻ��ȣ���ԡ��ȴ�����õ�һ��ɫ��״���塣���ˣ�����3 mL��ˮ�Ҵ�ϴ�ӣ��ñ��ؽᾧ���ð�ɫ�ᾧ�˼������ӿ���ϩ��
��1������1���ô���ټ��ȣ�����ҪĿ����____________________________��
��2��װ��ͼ�м�ʯ�ҵ�������__________________________________________��
��ˮ�Ȼ��Ƶ�������________________________________________________��
��3������װ�����ñ���ԡ��Ŀ����_____________________________________��
��4���ϳɰ˼������ӿ���ϩ���������Ŀ����_________________________��
��5��ȷ�۲�ƷΪ�˼������ӿ���ϩ����ͨ���ⶨ�е㣬���ɲ��õļ�ⷽ����__________________________________________________________��
��1����ֹ��������࣬��������ܵĶ�������Ⱦ����
��2���������ɵ�CO2����ֹˮ�������룬�����
��3����ӷ�������ԡ���ٻӷ�����߲���
��4��������
��5������Ʒ�ĺ˴Ź������ͺ������
����

 ��У����ϵ�д�
��У����ϵ�д�����ȩ��һ�ֻ���ԭ�ϡ�ijʵ��С����������װ�úϳ�����ȩ��
�����ķ�Ӧ���£�
CH3CH2CH2CH2OH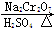 CH3CH2CH2CHO
CH3CH2CH2CHO
��Ӧ��Ͳ������������б����£�
| | �е�/�� | �ܶ�/(g��cm��3) | ˮ���ܽ��� |
| ������ | 117.2 | 0.810 9 | �� |
| ����ȩ | 75.7 | 0.801 7 | �� |
ʵ�鲽�����£�
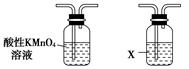
��6.0 g Na2 Cr2O7����100 mL�ձ��У���30 mLˮ�ܽ⣬�ٻ�������5 mLŨ���ᣬ��������ҺС��ת����B�С���A�м���4.0 g�������ͼ�����ʯ�����ȡ�������������ʱ����ʼ�μ�B����Һ���μӹ����б��ַ�Ӧ�¶�Ϊ90��95�棬��E���ռ�90�����µ���֡�
������ﵹ���Һ©���У���ȥˮ�㣬�л������������ռ�75��77����֣�����2.0 g��
�ش��������⣺
(1)ʵ���У��ܷ�Na2Cr2O7��Һ�ӵ�Ũ�����У�˵������______________________��
(2)�����ʯ��������________�������Ⱥ���δ�ӷ�ʯ��Ӧ��ȡ����ȷ������________��
(3)����װ��ͼ�У�B������������________��D������������________��
(4)��Һ©��ʹ��ǰ������еIJ�����________(����ȷ�𰸱��)��
a����ʪ������b���������c����©����d���궨
(5)������ȩ�ֲ�Ʒ���ڷ�Һ©���з���ˮʱ��ˮ��________��(��ϡ����¡�)��
(6)��Ӧ�¶�Ӧ������90��95�棬��ԭ����__________________________________��
�������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���
һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ����(��ѭ��ʹ��)����CCl4��Һ�У�����������(����ˮ����)��45 �淴Ӧ1 h����Ӧ�������ˣ���Һ�ֱ���5% NaHCO3��Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ���
(1)����ʵ���й��˵�Ŀ����_____________________________________��
(2)��Һ�ڷ�Һ©����ϴ�Ӿ��ú��л��㴦��________��(��ϡ����¡�)����Һʱ��������Һ�����������������ԭ�����Һ©�����������⣬����_________________________________________________________��
(3)���и����˴������༰�����Լױ�������ӦӰ���ʵ������
| ���� |  | ���������и����칹����������(%) | �ܲ���(%) | ||
| �������ױ� | �������ױ� | �������ױ� | |||
| ŨH2SO4 | 1.0 | 35.6 | 60.2 | 4.2 | 98.0 |
| 1.2 | 36.5 | 59.5 | 4.0 | 99.8 | |
| NaHSO4 | 0.15 | 44.6 | 55.1 | 0.3 | 98.9 |
| 0.25 | 46.3 | 52.8 | 0.9 | 99.9 | |
| 0.32 | 47.9 | 51.8 | 0.3 | 99.9 | |
| 0.36 | 45.2 | 54.2 | 0.6 | 99.9 | |
��NaHSO4���Ʊ��������ױ�ʱ��������ױ���������ʵ���֮��Ϊ________��
���ɼױ������õ��ĸ��ֲ���ĺ�����֪���ױ�������Ӧ���ص���_________________________________________________________��
����Ũ������ױ�������ȣ�NaHSO4���ױ��������ŵ���________________��________________��
 ���������������Է�ȩ��֬�ȡ�ʵ�����Ա��ӡ��嶡����[(CH3)3CCl]��Ϊԭ���Ʊ����嶡�����ӵ�ʵ�鲽�����£�
���������������Է�ȩ��֬�ȡ�ʵ�����Ա��ӡ��嶡����[(CH3)3CCl]��Ϊԭ���Ʊ����嶡�����ӵ�ʵ�鲽�����£�

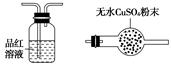
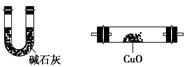

 H+ + OH����HCO3��
H+ + OH����HCO3��