��Ŀ����
15����ҵ������Ȼ����ڵĽ�Ĥ��þ����Ҫ�ɷ�ΪMg2B3O5•5H2O����ȡ����þ�ʹ��𣬹��չ������£�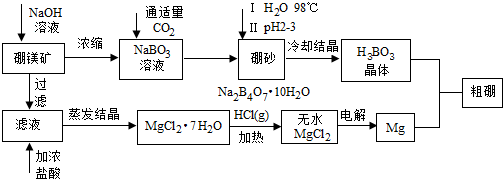
����������Ϣ�ش��������⣺
��1����ɰ����Ļ��ϼ�+3��������ˮ������Ҫ����pHΪ2-3��ȡ���ᣬд���÷�Ӧ�����ӷ���ʽ��B4O72-+2H++5H2O=4H3BO3��
��2�����Ȼ�þ�����Ƶ���ˮ�Ȼ�þ����������HCl���������м��ȣ��������Ƿ�ֹMgCl2ˮ������Mg��OH��2�����ö��Ե�ŵ��MgCl2��Һ��д�������̵����ӷ���ʽ��2H2O+Mg2++2Cl-$\frac{\underline{\;ͨ��\;}}{\;}$H2��+Mg��OH��2��+Cl2����
��3���ƵõĴ������һ��������I2��������BI2�����ᴿBI2��BI2�е�ϵͣ��ᴿ�ɲ�����������ʱBI2���ɷֽ⣬�õ��ߴ����𣮸��ݷֽ����ɵ���������Բⶨ����Ĵֶȣ��������£�
��ȡ0.04g����ת���ɴ�BI2������ȫ�ֽ⣬���ɵĵ���0.3mol/L��Na2S2O3��Һ�ζ����յ㣬���ı�Һ36.00mL���ζ�ԭ����I2+2S2O32-�T2I-+S4O62-��
�ٵζ��յ��ж�Ӧ�õ�����ָʾ����
�ڱ�ҺӦʢװ�ڼ�ʽ�ζ����У�
�۸ô���Ĵ���Ϊ99%��
��4��Mg-H2O2����Ⱦ�ϵ�صķ�Ӧ����Ϊ��Mg+H2O2+2H+=Mg2++2H2O������������ӦʽΪH2O2+2H++2e-=2H2O�������£�����ʼ���ҺpH=1�����ع���һ��ʱ����ҺpH=2����ʱ��Һ��c��Mg2+��0.045 mol•L-1������ҺpH=6ʱ��þԪ�ص���Ҫ������ʽ��Mg2+��ʽ���ڣ�û��Mg��OH��2����������֪��Ksp[Mg��OH��2]=5.6��10-12��
���� ��þ����Ҫ�ɷ�ΪMg2B2O5•H2O����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O��������þ����ȡ����þ������Ĺ�����������þ�������������Ũ��Һ���˵õ��Ȼ�þ������Ũ�����ܽ�ͨ������Ũ���õ��Ȼ�þ�ᾧˮ������Ȼ��������м��ȵõ��Ȼ�þ���壬���õ�þ����Һ����Ҫ��NaBO2��ͨ������������̼����õ���ɰ��������ˮ����H2SO4��pH2��3��ȡH3BO3�����ȵõ�B2O3��
��1����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O������Ԫ�ػ��ϼ۱�ע������Ԫ�ػ��ϼۣ���H2SO4��pH2��3����ɰ�е�Na2B4O7������Һ������H3BO3 ��
��2���Ȼ�þ��ˮ��Һ��ˮ������������þ�����Ե缫���MgCl2��Һ���������ӵõ���������������ˮ�ĵ���ƽ���ƻ���ˮ������������������Ũ������þ�����γ�������þ�������ݴ���д���ӷ��̣�
��3��BI2�е�ϵͣ��ᴿ����������е������������ķ�������õ������ɵĵ���0.3mol/L��Na2S2O3��Һ�ζ����յ㣬��Ҫ������Һ����Ӧ��ָʾ����H2S2O3Ϊ���ᣬNa2S2O3��Һ�Լ��ԣ��ݴ�ѡ��ζ��ܣ����ݹ�ϵʽB��BI3��$\frac{3}{2}$I2��3S2O32-���ζ����ݼ������������ĺ�����
��4��ȼ�ϵ�����������ǹ�������õ���������ˮ�����ݵ������ҺPH�仯��ϵ�ط�Ӧ����þ����Ũ�ȣ�PH=6��������������Ũ�ȣ��������þ����Ũ�ȼ���Ũ���̺��ܶȻ������ȽϷ����Ƿ�����������þ������
��� �⣺��1����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O����Ԫ�ػ��ϼ�Ϊ+1�ۣ���Ԫ�ػ��ϼ�-2�ۣ����ݻ��ϼ۴����ͼ���õ���Ԫ�ػ��ϼ�Ϊ+3�ۣ���H2SO4��pH2��3����ɰ�е�Na2B4O7������Һ������H3BO3 ����Ӧ�����ӷ���ʽΪ��B4O72-+2H++5H2O=4H3BO3��
�ʴ�Ϊ��+3��B4O72-+2H++5H2O=4H3BO3��
��2��MgCl2•7H2O��Ҫ��HCl��Χ�м��ȣ���Ϊ�˷�ֹ�Ȼ�þˮ������������þ�����ö��Ե缫���MgCl2��Һ�����������ӵõ���������������ˮ�ĵ���ƽ���ƻ���ˮ������������������Ũ������þ�����γ�������þ��������Ӧ�����ӷ���ʽΪ2H2O+Mg2++2Cl-$\frac{\underline{\;ͨ��\;}}{\;}$H2��+Mg��OH��2��+Cl2����
�ʴ�Ϊ����ֹMgCl2ˮ������Mg��OH��2��2H2O+Mg2++2Cl-$\frac{\underline{\;ͨ��\;}}{\;}$H2��+Mg��OH��2��+Cl2����
��3���ƵõĴ������һ��������I2��������BI2�����ᴿBI2��BI2�е�ϵͣ��ᴿ�ɲ�������ķ�������õ����ʴ�Ϊ������
�����ɵĵ���0.3mol/L��Na2S2O3��Һ�ζ����յ㣬��Ҫ������Һ����Ӧ��ָʾ�����ʴ�Ϊ��������Һ��
�������������Һ�ʼ��ԣ�ѡ���ʽ�ζ��ܣ��ʴ�Ϊ����ʽ��
����������Ƶ����ʵ���Ϊ��0.30mol/L��0.036L=0.0108mol�����ݹ�ϵʽ��B��BI3��$\frac{3}{2}$I2��3S2O32-��n��B��=$\frac{1}{3}$n��S2O32-��=0.0108mol��$\frac{1}{3}$=0.0036mol��
�������Ϊ��11g/mol��0.0036mol=0.0396g����������ĺ���Ϊ��$\frac{0.0396g}{0.04g}$��100%=99%��
�ʴ�Ϊ��99%��
��4��þ-H2O2����ȼ�ϵ�صķ�Ӧ����ΪMg+H2O2+2H+�TMg2++2H2O���������ǹ�������õ���������ˮ�ķ�Ӧ��������ӦʽH2O2+2H++2e-=2H2O������ʼ�������ҺpH=1����pH=2ʱ��Һ�У�������Ũ�ȼ�С0.1mol/L-0.01mol/L=0.09mol/L�����ݷ�Ӧ����ʽ�õ�Mg2+����Ũ��=0.045mol/L��Ksp[Mg��OH��2]=5.6��10-12������ҺpH=6ʱ��c��OH-��=10-8mol/L����Qc=c��Mg2+����c2��OH-��=0.045mol/L��10-16mol/L=4.5��10-18��Ksp[Mg��OH��2]��˵����������þ�������ɣ�ֻ��Mg2+��ʽ���ڣ�
�ʴ�Ϊ��H2O2+2H++2e-=2H2O��0.045 mol•L-1��Mg2+��ʽ���ڣ�û��Mg��OH��2������
���� ���⿼���������ˮ�⡢ԭ���ԭ���͵���ԭ���ķ����������ܽ�ƽ��ļ���Ӧ�ã���Ŀ�Ѷ��Դ������ѵ㣬����ʱҪ���������Ŀ����������ϵ�ͱ������ݣ�����������ϵ�����������ĸ�����ٽ��⣮

 A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�| A�� | 2.24L CO2�к��е�ԭ����Ϊ0.3NA | B�� | 20g D2O������������Ϊ10NA | ||
| C�� | 16g CH4�к�C-H����ĿΪNA | D�� | 1 mol �������е�ԭ����Ϊ2NA |

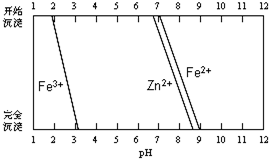
��֪�����й��������������pH��
| �������� | Fe��OH��3 | Fe��OH��2 |
| ��ʼ������pH | 1.5 | 6.5 |
| ������ȫ��pH | 3.7 | 9.7 |
��1����������Ҫ�ӿ췴Ӧ���ʣ���ʩ�г�ֽ���ͼ��ȡ��ʵ���������Ũ�ȵȣ�дһ�֣���̼���������ᷴӦ�����ӷ���ʽSrCO3+2H+=Sr2++CO2��+H2O��
��2���ڲ����-�۵Ĺ����У�����Һ��pHֵ��1������B�����õ��Լ�ΪE��
A.1.5 B.3.7 C.9.7 D����ˮ E���������ȷ�ĩ F��̼���ƾ���
��3����������������������Ҫ�ɷ���Fe��OH��3��BaSO4 ���ѧʽ����
��4����ҵ�����ȷ紵����ˮ�Ȼ��ȣ����˵��¶���A
A��50��60��B��80��100��C��100������
��5���������ѡ�õ���ϴ�Ӽ��DZ����Ȼ�����Һ��
| A�� |
| B�� |
| C�� |
| D�� |
|
| A�� | CaCl2 | B�� | Na2O | C�� | H2SO4 | D�� | NH4Cl |
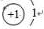 ��C�����ڱ��е�λ���ǵڶ����ڵڢ�A�壬�ӽṹ�Ƕ��ж�A2Ӧ��C2���� �����ȶ�����ã������ǵ��������ļ��ܴ���H-H
��C�����ڱ��е�λ���ǵڶ����ڵڢ�A�壬�ӽṹ�Ƕ��ж�A2Ӧ��C2���� �����ȶ�����ã������ǵ��������ļ��ܴ���H-H �����к��еĻ�ѧ�����������Ӽ����ۼ�
�����к��еĻ�ѧ�����������Ӽ����ۼ�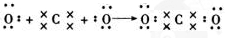 ��
��

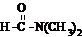 ����ˮ
����ˮ