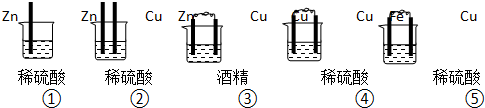
��Ŀ����
13������ȩ����������Ʊ�1-������Ϊ�ᴿ����������ȩ���ʵ�1-���������������·�ߣ���Ʒ$��_{����1}^{����NaHSO3��Һ}$��Һ$��_{��ȡ}^{��ȩ}$����2�л���$��_{����3}^{��������}$1-�������ѣ�����4����Ʒ
��֪��������ȩ�뱥��NaHSO3��Һ��Ӧ�����ɳ����������ѵķе���34�棬������ˮ����1-�������ܣ���1-�����ķе���118�森�����1��4�ֱ��ǣ�������
| A�� | ��ȡ�����ˡ��������� | B�� | ���ˡ���Һ��������ȡ | ||
| C�� | ���ˡ������ˡ����� | D�� | ���ˡ���Һ�����ˡ����� |
���� ���ᴿ����������ȩ���ʵ�1-���������̿�֪����Ʒ�мӱ���NaHSO3��Һ������ȩ��Ӧ���ɳ����������1Ϊ���ˣ�����Һ��ȡ���Һ�õ����Ѻ�1-����������2Ϊ��Һ���л����мӹ�����������ȥˮ������3Ϊ���ˣ��������Ѻʹ��������ɣ�
��� �⣺���ᴿ����������ȩ���ʵ�1-���������̿�֪����Ʒ�мӱ���NaHSO3��Һ������ȩ��Ӧ���ɳ�����������������Һ����룬�����1Ϊ���ˣ�����Һ��ȡ���Һ�õ����Ѻ�1-����������2Ϊ��Һ���л����мӹ�����������ȥˮ������3Ϊ���ˣ��������Ѻʹ��Ļ������÷е㲻ͬ�������������4Ϊ����
��ѡD��
���� ���⿼����������ᴿʵ�鷽������ƣ�Ϊ��Ƶ���㣬�����л�������ʼ��������뷽��Ϊ���Ĺؼ���ע�������з����Ļ�ѧ��Ӧ��ϰ���е���Ϣ����Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ
3���������ö��Ե缫���1L������KCl����Һ����ͨ��n mol���ӵĵ���������Һ��pH��n�Ĺ�ϵ�ǣ�����ǰ����Һ��������䣬���ɵ�����ȫ���ݳ�����������
| A�� | pH=n | B�� | pH=-lg n | C�� | pH=14-lgn | D�� | pH=lgn+14 |
4�����³�ѹ����7.8g��Na2O2����������1H218O�У���Ӧ��ȫ���й����ʵ���������ȷ���ǣ�������
| A�� | ת�Ƶĵ�������0.1NA | B�� | ��Ӧ��Һ����������1.6g | ||
| C�� | ���ɵ������к���0.8mol���� | D�� | ��������������1.12L |
8�����й����л��������˵����ȷ���ǣ�������
| A�� | ���͵����ʶ��Ǹ߷��ӻ����ˮ�������ͬ | |
| B�� | ���ͼ�ϩ����ʹ��ˮ��ɫ��������ˮ�����ӳɷ�Ӧ | |
| C�� | ����̼��������Һ����������Ҵ� | |
| D�� | ����ˮ��ȥ�����л��е������������� |
5��������Ԫ��X��Y��Z��W��ԭ��������������Ԫ��X��һ������Ľ�����Yԭ�ӵ�����������������Ӳ�����2��������ѧ��Χ��Z�ĵ���ֻ�������ԣ����ǿ�������Ҫ�ɷ�֮һ��W���⻯������Z��ij���⻯�ﷴӦ����Z������һ���⻯���W���ʣ�����˵����ȷ���ǣ�������
| A�� | ԭ�Ӱ뾶��W��Z��Y��X | |
| B�� | W���������ڻ�����YW2 | |
| C�� | �⻯����ȶ��Ժͷе����Z��W | |
| D�� | X�����ڼ��ȵ���������Z��ij�ֵ��ʷ�Ӧ����X2Z2 |
3����ҵ�ϳ���ϡ������ϴ���������㣬������������ģ�������
| A�� | ǿ������ | B�� | ���ӷ��� | C�� | ��ˮ�� | D�� | ���� |
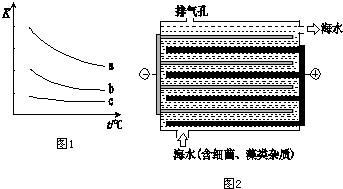
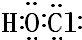 ��
��