��Ŀ����
17����ҵ�ϳ���ˮ��������������Ӧ��ȡ������ʹҩ��˾ƥ�֣�����ˮ���ᣩ������Ӧԭ����
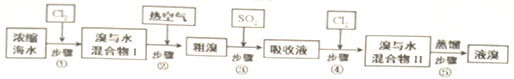
��ͼ1
���������ʡ�
| �Լ� | �е㣨�棩 | �ܽ�� | ��ѧ���� |
| ������ | 139 | ��ˮ���ֽ� | �� |
| ����ˮ���� | �� | ����ˮ | ��̼���Ʒ�Ӧ����ˮ������ |
| ˮ���� | 211 | ������ˮ����������ˮ | �� |
| ���� | 117.9 | �� | �� |
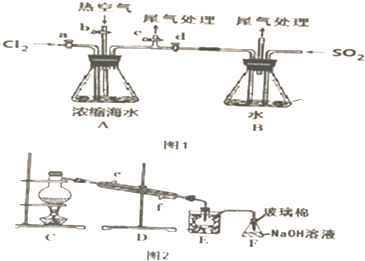
��1�������Ʊ�������Թ������μ���2gˮ���ᡢ5mL���������ܶ�Ϊ1.08g/mL����5��Ũ���ᣬ���Թ���ˮ����ȫ���ܽ⣬�������ʢ����ˮ�ձ��У��̶�������̨�ϣ���85�桫90�������¼���5��10min��װ����ͼ2��
��2����Ʒ�ᾧ��ȡ�´��Թܣ�������ˮ����ȴ10min����������ȫ�����������ˣ���ϴ�Ӿ��壬��ɣ�
��3����Ʒ�ᴿ�����ֲ�Ʒת����150mL�ձ��У����������������Լ�NaHCO3�����Ͻ��������ٲ�������Ϊֹ����һ���ᴿ���ջ������ˮ����1.8g��
��4�����ȼ��飺ȡ������Ʒ����ʢ��5mLˮ���Թ��У�����1��2��FeCl3��Һ����Һ��dz��ɫ��
���������ۡ�
��1��д��ˮ������̼��������Һ��Ӧ�Ļ�ѧ��Ӧ����ʽ
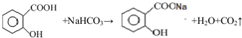 ��
����2��д��ˮ����������̼������Һ��Ӧ������������Ľṹ��ʽ
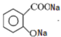 ��
����3�����̣�1��װ��ͼ�г����ܵ���������������ƽ��ѹǿ��
��4�����Ʒ�Ӧ�¶�85�桫90���ԭ��ȱ����нϸߵķ�Ӧ�����ּ������ʵĻӷ���
��5�����̣�2����֤������ϴ�Ӹɾ��IJ���������ȡ���һ��ϴ����Һ�������Թ��У��μ������Ȼ������ٵμ�ϡ���ᣬ������������ϴ�Ӹɾ�����֮��δϴ�Ӹɾ�
��6�����̣�4������Һ����ɫ�����ܵ�ԭ���Dz�Ʒ����Ȼ������ˮ���ᣮ
��7��1mol����ˮ����������������Һ��Ӧ���ܣ��������3mol NaOH��
��8��ʵ��������ˮ����IJ���Ϊ69%����֪��ˮ���ᡢ����ˮ�������Է��������ֱ�Ϊ138��180����
���� ��1������ˮ����Ľṹ��ʽ��֪��ˮ���������Ȼ����Ȼ�����̼��������Һ��Ӧ����������̼��
��2������ˮ����Ľṹ��ʽ��֪��ˮ�����е��Ȼ��ͷ��ǻ�����������̼������Һ��Ӧ���ݴ˴��⣻
��3��ͼ2���Թ���ˮԡ���ȣ��������̣�1��װ��ͼ�г����ܿ�������������ƽ��ѹǿ�����ã�
��4��������ˮ�����������ᣬ���������ṩ��Ϣ��֪�������ӷ����������ʶȵķ�Ӧ���ʣ��ݴ˿��Ʒ�Ӧ�¶ȣ�
��5�����̣�2���о�������ʱ�����п�������������ӣ������ж�ϴ�Ӹɾ��IJ����������Ǽ���ϴ��Һ���Ƿ�����������ӣ�
��6����ʹFeCl3��Һ��dz��ɫ���Ƿ��ǻ���
��7������ˮ�����е��������Ȼ����������ƶ��ܷ�Ӧ��������ˮ�����е�����ˮ������ɷ��ǻ�Ҳ�����������Ʒ�Ӧ���ݴ˴��⣻
��8������ͼ1����Ϣ��֪��1molˮ��������1mol����ˮ���ᣬ�ݴ˼���2gˮ���������ϲ�������ˮ��������������ݵIJ���Ϊ$\frac{ʵ�ʲ���}{���۲���}$��100%���㣮
��� �⣺��1������ˮ����Ľṹ��ʽ��֪��ˮ���������Ȼ����Ȼ�����̼��������Һ��Ӧ����������̼������ˮ������̼��������Һ��Ӧ�Ļ�ѧ��Ӧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��2������ˮ����Ľṹ��ʽ��֪��ˮ�����е��Ȼ��ͷ��ǻ�����������̼������Һ��Ӧ������ˮ����������̼������Һ��Ӧ������������Ľṹ��ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��3��ͼ2���Թ���ˮԡ���ȣ��������̣�1��װ��ͼ�г����ܿ�������������ƽ��ѹǿ�����ã�
�ʴ�Ϊ������������ƽ��ѹǿ��
��4��������ˮ�����������ᣬ���������ṩ��Ϣ��֪�������ӷ����������ʶȵķ�Ӧ���ʣ����Կ��Ʒ�Ӧ�¶�Ϊ85�桫90����Լȱ����нϸߵķ�Ӧ�����ּ������ʵĻӷ���
�ʴ�Ϊ���ȱ����нϸߵķ�Ӧ�����ּ������ʵĻӷ���
��5�����̣�2���о�������ʱ�����п�������������ӣ������ж�ϴ�Ӹɾ��IJ���������ȡ���һ��ϴ����Һ�������Թ��У��μ������Ȼ������ٵμ�ϡ���ᣬ������������ϴ�Ӹɾ�����֮��δϴ�Ӹɾ���
�ʴ�Ϊ��ȡ���һ��ϴ����Һ�������Թ��У��μ������Ȼ������ٵμ�ϡ���ᣬ������������ϴ�Ӹɾ�����֮��δϴ�Ӹɾ���
��6����ʹFeCl3��Һ��dz��ɫ���Ƿ��ǻ����������̣�4������Һ����ɫ��˵����Ʒ����Ȼ������ˮ���ᣬ
�ʴ�Ϊ����Ʒ����Ȼ������ˮ���
��7������ˮ�����е��������Ȼ����������ƶ��ܷ�Ӧ��������ˮ�����е�����ˮ������ɷ��ǻ�Ҳ�����������Ʒ�Ӧ������1mol����ˮ����������������Һ��Ӧ���ܣ��������3mol NaOH��
�ʴ�Ϊ��3��
��8������ͼ1����Ϣ��֪��1molˮ��������1mol����ˮ���ᣬ����2gˮ���������ϲ�������ˮ���������Ϊ$\frac{180}{138}$��2g=2.61g����������ˮ����IJ���Ϊ$\frac{ʵ�ʲ���}{���۲���}$��100%=$\frac{1.8g}{2.61g}$��100%=69%��
�ʴ�Ϊ��69%��
���� ���⿼���л����Ʊ�ʵ�鷽�����漰�Բ������������Ƶķ������ۡ�ϴ�ӡ����ʵķ����ᴿ�����ʼ���ȣ����ؿ���ѧ����֪ʶ��Ǩ�����á��������������������Ѷ��еȣ�

 ��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
��ʦ����ɳ���ʱͬ��ѧ����ϵ�д����Ȼ��� ��̼���� ���������� �ܹ����ƣ�
| A�� | ���٢� | B�� | ���ڢ� | C�� | ���٢ۢ� | D�� | ���٢ڢ� |
| A�� | ��pH=0����Һ�У�Fe2+��Mg2+��SO42-��NO3- | |
| B�� | ��ʹ���۵⻯����ֽ����ɫ����Һ��K+��SO42-��S2-��SO32- | |
| C�� | ������c��H+��/c��OH-��=1012����Һ��Fe3+��Mg2+��NO3-��Cl- | |
| D�� | ����ʹ��̪������ɫ��Һ��Na+��CO32-��K+��ClO-��AlO2- |
| ѡ�� | �������ʵ | ���� |
| A | ���ȵĴ�����Һϴȥ���� | Na2CO3��ֱ�Ӻ����۷�Ӧ |
| B | Ư���ڿ����о��ñ��� | Ư���е�CaCl2������е�CO2��Ӧ����CaCO3 |
| C | ʩ��ʱ����ľ�ң���Ч�ɷ�ΪK2CO3��������NH4Cl���ʹ�� | K2CO3��NH4Cl�������з�Ӧ���ɰ����ή�ͷ�Ч |
| D | �����ڳ�ʪ�Ŀ������������� | �����ڿ����з��������ⸯʴ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | Ba��OH��2��Һ�еμ�NaHSO4��Һ�����ԣ�Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O | |
| B�� | ����ˮ��Ӧ��Na+2H2O=Na++2OH-+H2�� | |
| C�� | ���Ȼ�����Һ�ͷ�ˮ��Ӧ��ȡ�����������壺Fe3++3H2O����ˮ���TFe��OH��3��+3H+ | |
| D�� | Ư����Һ�ڿ�����ʧЧ��ClO-+CO2+H2O�THClO+HCO3- |
| A�� | ��ǦԪ��ԭ�ӵ�����������Ϊ6 | B�� | �䳣����̬Ϊ+2��+3��+4 | ||
| C�� | ���Ľ����Ա�Ǧǿ | D�� | ����ԭ�Ӱ뾶��ǦС? |
��1����������������Ӧ����1molˮ��������241.8kJ��д���÷�Ӧ���Ȼ�ѧ����ʽH2��g��+$\frac{1}{2}$O2��g��=H2O��g����H=-241.8kJ/mol����1gˮ����ת����Һ̬ˮ����2.444kJ����Ӧ2H2��g��+O2��g���T2H2O��l���ġ�H=-571.6kJ/mol��������ȼ����Ϊ285.8kJ/mol��
��2������̬��̬ԭ���γ�1mol��ѧ���ͷŵ���������м��ܣ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̣��ڻ�ѧ��Ӧ�����У��ƻ��ɻ�ѧ����Ҫ�����������γ��»�ѧ���ֻ��ͷ�������
| ��ѧ�� | H-H | N-H | N��N |
| ����/kJ•mol-1 | 436 | 391 | 945 |
��3�����ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ���ʱ�������㣮
��֪��C��s��ʯī��+O2��g���TCO2��g����H1=-393.5kJ•mol-1
2H2��g��+O2��g���T2H2O��l����H2=-571.6kJ•mol-1
2C2H2��g��+5O2��g���T4CO2��g��+2H2O��l����H3=-2 599kJ•mol-1
���ݸ�˹���ɣ�����298Kʱ��C��s��ʯī����H2��g������1mol C2H2��g����Ӧ���ʱ䣨�г��ļ���ʽ������H=2��H1+$\frac{1}{2}$��H2-$\frac{1}{2}$��H3=+226.7kJ•mol-1��