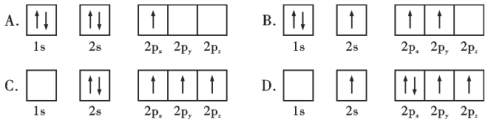
��Ŀ����
����Ŀ��ClO2��NaClO2������Ư���ԣ���ҵ����ClO2������NaClO2�Ĺ���������ͼ��ʾ��
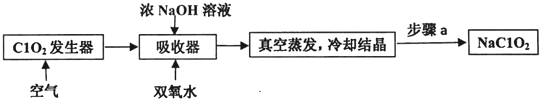
����˵������ȷ����( )
A.����a�IJ����������ˡ�ϴ�Ӻ���
B.ͨ�������Ŀ�������ϳ�ClO2��ʹ�䱻�������������
C.��ҵ�Ͽɽ�ClO2�Ƴ�NaClO2���壬�������������
D.������������NaClO2�����ӷ���ʽ��2ClO2+H2O2=2C1O2- -+O2��+2H+
���𰸡�D
��������
�����̿�֪��������ͨ��������ɽ�ClO2�ų���ȷ���䱻������գ����������з���2ClO2+2NaOH+H2O2�T2NaClO2+O2+2H2O��Ȼ�������������ȴ�ᾧ���پ������ˡ�ϴ�Ӻ���õ�NaClO2���Դ˽����⡣
A������a�ǽ��������Һ��ȡ����Ȼ�������Բ����������ˡ�ϴ�Ӻ��A��ȷ��
B����Ӧ�����������к�������ClO2��ͨ�������Ŀ������ClO2�ݳ���������������ŨNaOH��Һ��˫��ˮ������գ�B��ȷ��
C����ҵ�����û�ԭ����NaOH��Һ����ClO2�Ƴ�NaClO2���壬�����Ϊ�ȶ���������������䣬C��ȷ��
D����������Ӧ�ڼ��������½��У�����NaClO2�����ӷ���ʽ��2ClO2+H2O2+2OH-=2C1O2-+O2��+2H2O��D����ȷ��
��ѡD��

 ����ѧУ�ֲ����ܲ�ϵ�д�
����ѧУ�ֲ����ܲ�ϵ�д� �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
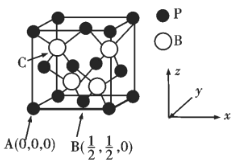
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д�����Ŀ���±��г���5��Ԫ�������ڱ��е�λ�á�
�� ���� | ��A | 0 | ||||||
1 | ��A | ��A | ��A | ��A | ��A | ��A | ||
2 | �� | �� | ||||||
3 | �� | �� | �� |
(1)�ٵ�Ԫ�ط�����_______���ݵ�ԭ�ӽṹʾ��ͼ��__________��
(2)����Ԫ���У��ǽ�������ǿ����___________(��Ԫ�ط���)��
(3)��Ԫ�ص�����������Ӧ��ˮ�����______��(������������������������)��
(4)�ۢܢ�����Ԫ�ذ�ԭ�Ӱ뾶�ɴ�С��˳����______(��Ԫ�ط���)��
(5)Ԫ�آ���Ԫ�آڵĵ��������Խ�ǿ����________(�ѧʽ)����
(6)Ԫ�آܵ������������Ԫ�آ۵�����������Ӧ��ˮ�������Ӧ�Ļ�ѧ����ʽ__��
����Ŀ���ƺͼ��Ǽ����õĽ���Ԫ�أ��ƺͼؼ����������������������й㷺��Ӧ�á�
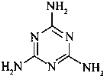
��1��д�����ֿ���ʳ�õĺ��ƻ�����Ļ�ѧʽ��________����0.01 mol��������(��Na2O2����Na2O����Na2CO3����NaCl)�ֱ����100 mL����ˮ�У��ָ������£�������Һ��������Ũ���ɴ�С��˳����(��Һ����仯���Բ���)_______��
��2�����ڼر��Ƹ����ã��Ʊ�K2Oһ�����õ��ʼػ�ԭ��Ӧ�Ĺ�����������λ��������Σ���д���ü�������ط�Ӧ��ȡK2O�Ļ�ѧ����ʽ(����һ�ֵ�������)��______________________��K2O2Ҳ��ǿ�����ԣ���д������SO2������Ӧ�Ļ�ѧ����ʽ��_______________________��
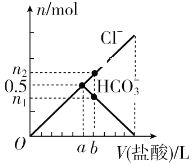
��3��ijѧ����Na2CO3�� KHCO3��ɵ�ij��������ʵ�飬����������(��������ʵ���Ũ������Ҳ�����HCl�Ļӷ�)
ʵ����� | �� | �� | �� | �� |
�������/mL | 50 | 50 | 50 | 50 |
��������/g | 3.06 | 6.12 | 9.18 | 12.24 |
�����������/L(���) | 0.672 | 1.344 | 1.568 | 1.344 |
�������ݼ���������������ʵ���Ũ��Ϊ__________��ԭ�������Ʒ��n(Na2CO3)��n(KHCO3)��_________��