��Ŀ����
9������������������Ҫ��ҽҩ�м��壬��A��B Ϊԭ�Ϻϳɱ�����������F·����ͼ��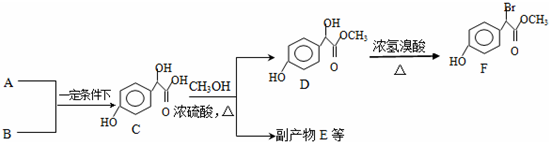
��1��A�ķ���ʽΪC2H2O3���ɷ���������Ӧ���Ҿ������ԣ�A��������������Ϊ��ȩ�����Ȼ���д��A+B��C�Ļ�ѧ��Ӧ����ʽΪ
 ��
����2��
 �Т١��ڡ��ۣ������ұ�ţ�3��-OH��������ǿ������˳���ǣ��ۣ��٣��ڣ�
�Т١��ڡ��ۣ������ұ�ţ�3��-OH��������ǿ������˳���ǣ��ۣ��٣��ڣ���3��E����2����C���ɵĺ���3����Ԫ���Ļ����E�ķ����в�ͬ��ѧ��������ԭ����4�֣�
��4��D��F�ķ�Ӧ������ȡ����Ӧ��1mol F��һ��������������NaOH��Һ��Ӧ���������NaOH�����ʵ���Ϊ��3mol��д����������������F������ͬ���칹�壨�����������칹���Ľṹ��ʽ��
 ��
��������һԪ������
�ڱ�����ֻ��2��ȡ�����Ҵ��ڶ�λ������һ�����ǻ���
���� ��1��A�ķ���ʽΪC2H2O3���ɷ���������Ӧ���Ҿ������ԣ�����ȩ�����Ȼ�����A��OHC-COOH������C�Ľṹ��֪B��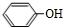 ��A+B��C�����ӳɷ�Ӧ��
��A+B��C�����ӳɷ�Ӧ��
��2���Ȼ�������ǿ�ڷ��ǻ������ǻ�������ǿ�ڴ��ǻ���
��3��C�����ǻ����Ȼ���2����C���Է���������Ӧ����������3����Ԫ���Ļ����C���Ӽ䴼�ǻ����Ȼ�����������Ӧ����EΪ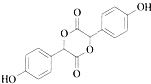 ��
��
��4���Ա�D��F�Ľṹ����֪��ԭ��ȡ��-OHλ�ã�F����ԭ�ӡ����ǻ��������������봼�γɵ������������������������Ʒ�Ӧ��
F������ͬ���칹����ϣ�������һԪ�������ڱ�����ֻ��2��ȡ�����Ҵ��ڶ�λ������һ�����ǻ�������ȡ����Ϊ-CBr��CH3��COOH��-CH��CH2Br��COOH��-CH2CHBrCOOH��-CHBrCH2COOH��
��� �⣺��1��A�ķ���ʽΪC2H2O3���ɷ���������Ӧ���Ҿ������ԣ�����ȩ�����Ȼ�����A��OHC-COOH������C�Ľṹ��֪B�� ��A+B��C�����ӳɷ�Ӧ����Ӧ����ʽΪ��
��A+B��C�����ӳɷ�Ӧ����Ӧ����ʽΪ�� ��
��
�ʴ�Ϊ��ȩ�����Ȼ��� ��
��
��2���Ȼ�������ǿ�ڷ��ǻ������ǻ�������ǿ�ڴ��ǻ�����ǿ��˳��Ϊ���ۣ��٣��ڣ�
�ʴ�Ϊ���ۣ��٣��ڣ�
�Ȼ�������ǿ�ڷ��ǻ������ǻ�������ǿ�ڴ��ǻ���
��3��C�����ǻ����Ȼ���2����C���Է���������Ӧ����������3����Ԫ���Ļ����C���Ӽ䴼�ǻ����Ȼ�����������Ӧ����EΪ ��Ϊ�Գƽṹ����������4�ֻ�ѧ������ͬ��Hԭ�ӣ��ֱ�Ϊ������2�֡����ǻ���1�֡��Ǽ���1�֣�
��Ϊ�Գƽṹ����������4�ֻ�ѧ������ͬ��Hԭ�ӣ��ֱ�Ϊ������2�֡����ǻ���1�֡��Ǽ���1�֣�
�ʴ�Ϊ��4��
��4���Ա�D��F�Ľṹ����֪��ԭ��ȡ��-OHλ�ã�D��F�ķ�Ӧ�����ǣ�ȡ����Ӧ��F����ԭ�ӡ����ǻ��������������봼�γɵ������������������������Ʒ�Ӧ��1molF�������3mol NaOH��
F������ͬ���칹����ϣ�������һԪ�������ڱ�����ֻ��2��ȡ�����Ҵ��ڶ�λ������һ�����ǻ�������ȡ����Ϊ-CBr��CH3��COOH��-CH��CH2Br��COOH��-CH2CHBrCOOH��-CHBrCH2COOH�����ܵĽṹ��ʽΪ�� ��
��
�ʴ�Ϊ��ȡ����Ӧ��3�� ��
��
���� ���⿼���л�����ƶ���ϳɡ�ͬ���칹�����д�������л���Ӧ���͡������ŵ����ʵȣ��Ƕ��л���ѧ�������ۺϿ��飬�Ѷ��еȣ�


| A�� | 4Fe��0H��2+O2+2H2O�T4Fe��OH��3 | B�� | 2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2�� | ||
| C�� | 4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O | D�� | 2CuO+C$\frac{\underline{\;����\;}}{\;}$2Cu+CO2�� |
| A�� | 920 kJ | B�� | 557 kJ | C�� | 436 kJ | D�� | 188 kJ |
| A�� | ԭ�Ӱ뾶��D��E��B��C��A | |
| B�� | ���ȶ��ԣ�EA4��A2C | |
| C�� | BԪ�ص�����������ˮ������D���ʷ�Ӧ��������A2���� | |
| D�� | ������DC�뻯����EC2�л�ѧ��������ͬ |
 �̣�Mn����ά������������������������Ԫ�أ����̻�������������������Ҳ���㷺Ӧ�ã����ͼ����Ϣ������������ȷ���ǣ�������
�̣�Mn����ά������������������������Ԫ�أ����̻�������������������Ҳ���㷺Ӧ�ã����ͼ����Ϣ������������ȷ���ǣ�������| A�� | ��ԭ�Ӻ�����25�����ӣ�һ����ԭ�ӵ�������54.94g | |
| B�� | ��ԭ�Ӻ�����4�����Ӳ㣬�仯ѧ���������ڲ���Ӿ��� | |
| C�� | MnO2�����������H2O2�ֽ���O2��ʵ���������� | |
| D�� | KMnO4Ϊ�Ϻ�ɫ���壬������ˮ�����͵��ܼ����γ��Ϻ�ɫ��Һ |
| A�� | Al3+Na+HCO3- Cl- | B�� | K+ Na+ HCO3-AlO2- | ||
| C�� | Mg2+ NH4+ SO42-Cl- | D�� | Fe2+ Na+Cl- NO3- |
| A�� | Na2CO3 | B�� | Na2O | C�� | CaO | D�� | Al��OH��3 |

 ��
��