��Ŀ����
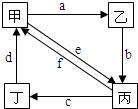
17��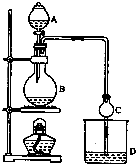 ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ����֪��
ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ����֪������ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2•6C2H5OH
���й��л���ķе㣺
| �Լ� | ���� | �Ҵ� | ���� | �������� |
| �е�/�� | 34.7 | 78.5 | 118 | 77.1 |
��1��Ũ��������������ԡ���������ˮ��������ͬλ��18Oʾ�ٷ�ȷ����Ӧ����ˮ��������ԭ�ӵ��ṩ�ߣ�д���ܱ�ʾ18Oλ�õĻ�ѧ����ʽ��CH3CO18OH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOC2H5+H218O��
��2�����θ����C�������Ƿ�ֹ����������������Ӧǰ��D�м��뼸�η�̪����Һ�ʺ�ɫ����Ӧ������D�е���������Һ�ֲ㣬�ϲ���ɫ����Һ�壬�²���Һ��ɫ��dz��
��3�����÷�Һ���������ƣ����� D�з���������������г�����һ�������Ҵ������Ѻ�ˮ��Ȼ�������ˮ�Ȼ��ƣ�������Ҵ����ټ��루������ѡ����ѡ��C��Ȼ����������ռ�77�����ҵ���֣��Եýϴ���������������
A������������ B����ʯ�� C����ˮ������ D����ʯ�ң�
���� ��1���������Ҵ���Ũ���������������ȵ������·���������Ӧ���������������÷�ӦΪ���淴Ӧ��Ũ������ˮ����ƽ���������������������ƶ���
�����봼������������Ӧ�У������е��Ȼ��ṩ-OH�����е�-OH�ṩ-H����������ˮ��
��2��Բ����ƿ���Ȳ��������θ���ܵĹܿ�����Һ���¿��ܷ���������ͬʱ���������ã�
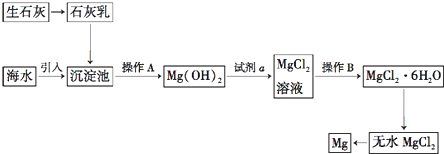
�������ᶼ�ӷ����Ʊ����������������Ҵ������ᣬͨ���ñ���̼������Һ����������������Ӧ���ӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У�������������������ζ���ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����ڷֲ�õ�������D�е���ҺΪ���͵�̼������Һ��̼���ˮ�⣬��Һ�ʼ��ԣ���Ӧ����Һ�ֲ㣬�ϲ���ɫ����Һ�壬����̼���������ᷴӦ�����²���Һ��ɫ�䣻
��3���÷�Һ��������ᴿ����������Ȼ�����Ȼ��Ƴ�ȥ�����Ҵ�������ˮ�����Ƴ�ȥ������ˮ����ˮ��������ˮ�γ������ƽᾧˮ����������������ķе��ж��¶ȣ�
��� �⣺��1�������Ҵ���Ũ�������������÷�ӦΪ���淴Ӧ��Ũ������ˮ����ƽ���������������������ƶ������ڷ�Ӧ������������ƣ���Ũ���ỹ����������ã���Ũ��������������ԡ���������ˮ����
�����봼������������Ӧ�У������е��Ȼ��ṩ-OH�����е�-OH�ṩ-H����������ˮ����������������������ͬʱ�÷�Ӧ���棬��Ӧ�Ļ�ѧ����ʽΪCH3CO18OH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOC2H5+H218O��
�ʴ�Ϊ�����ԡ���������ˮ����CH3CO18OH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOC2H5+H218O��
��2��Բ����ƿ���Ȳ��������θ���ܵĹܿ�����Һ���¿��ܷ������������θ����������Է�ֹ������ͬʱ���������ã�
̼���ˮ��CO32-+H2O?HCO3-+OH-����Һ�ʼ��ԣ����뼸�η�̪����Һ�ʺ�ɫ��
̼����ˮ��ʼ��ԣ��������������ڱ���̼������Һ���ܶȱ�ˮС����Һ�ֲ㣬�ϲ���ɫ����Һ�壬�����̼���Ʒ�Ӧ��ʹ��Һ��ɫ��dz��
�ʴ�Ϊ����ֹ��������������Һ�ֲ㣬�ϲ���ɫ����Һ�壬�²���Һ��ɫ��dz��
��3���÷�Һ��������ᴿ����������Ȼ�����Ȼ��Ƴ�ȥ�����Ҵ�������ˮ�����Ƴ�ȥ������ˮ����ˮ��������ˮ�γ������ƽᾧˮ������������ķе�Ϊ77�棬�����ռ� 77�����ҵ���֣��Եýϴ���������������
�ʴ�Ϊ����Һ���Ҵ���C��77��
���� ���⿼�������������Ʊ���ʵ��Ľ����Ѷ��еȣ�ע�⣨3���漰�����������ľ��ƣ�����ˮ�����Ƴ�ȥ������ˮ������ѡ��P2O5����ʯ�Һ�NaOH�ȹ����������Է��������������ԣ�P2O5��ˮ�����ᣩ�����������ˮ�⣮

 ������и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������
������и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������| �� | �� | �� | �� | |
| �� | Cu | CuO | CuCl2 | Cu��NO3��2 |
| �� | Na2CO3 | NaOH | NaHCO3 | CO2 |
| �� | ��NH4��2SO3 | CaSO3 | SO2 | NH4HSO3 |
| �� | Al��OH��3 | Al2O3 | NaAlO2 | AlCl3 |
| A�� | �٢ڢۢ� | B�� | ���ڢۢ� | C�� | ���ڢ� | D�� | ���ۢ� |
| A�� | ��ѹ�����ڰ��ĺϳ� | |
| B�� | ʵ�����г����ű���ʳ��ˮ�ķ�ʽ�ռ����� | |
| C�� | ����ˮƿ�������ݴ���Һ��ð�� | |
| D�� | ��˫��ˮ�м�������������������������� |
| A�� | ��C��s��ʯī���TC��s�����ʯ����H=+1.9 kJ•mol-1����֪ʯī�Ƚ��ʯ���ȶ� | |
| B�� | ���������������������ֱ���ȫȼ�գ����߷ų����������� | |
| C�� | ��H+��aq��+OH-��aq���TH2O��l����H=-57.3 kJ•mol-1����֪��1 mol CH3COOH����Һ�뺬1 mol NaOH����Һ��ֻ�Ϸ�Ӧ���ų�����������57.3 kJ | |
| D�� | 2g H2��ȫȼ������Һ̬ˮ�ų�285.8 kJ������������ȼ�յ��Ȼ�ѧ����ʽΪ2H2��g��+O2��g���T2H2O��l����H=+571.6 kJ•mol-1 |
| A�� | $\frac{1-W}{7}$ | B�� | $\frac{3W}{4}$ | C�� | $\frac{6��1-W��}{7}$ | D�� | $\frac{12��1-W��}{13}$ |
| A�� | C2H2 | B�� | C3H6 | C�� | C7H8 | D�� | C2H4 |