��Ŀ����
13��ʵ��������Na2CO3•10H2O��������1mol/L��Na2CO3��Һ100mL������˵����ȷ���ǣ�������| A�� | Ҫ���ʵ�����ȡ10.6gNa2CO3•10H2O���� | |
| B�� | ���ݺ������ƿ��תҡ�ȣ�����Һ����ڿ̶ȣ��ٲ��伸��ˮ���̶��ߣ��ᵼ����ҺŨ��ƫ�� | |
| C�� | ����ʱ������ƿ�����������������ˮ�ᵼ��Ũ��ƫ�� | |
| D�� | ����ʱ���ӿ̶��ᵼ��Ũ��ƫ�� |
���� A������m=CVM������Ҫ���ʵ�������
B����������һ�����ʵ���Ũ����Һ�IJ���ѡ��������
C������ʱ����Ҫ������ƿ�м�������ˮ��
D������ʱ���ӿ̶��ߣ�������Һ���ƫС��
��� �⣺A������1mol/L��Na2CO3��Һ100mL����Ҫ̼���ƾ��������m=1mol/L��286g/mol��0.1L=28.6g����A����
B������һ�����ʵ���Ũ����Һ���ƵIJ��裺�������ܽ⡢��Һ��ϴ�ӡ����ݵȣ��õ�����������ƽ��ҩ�ס����������ձ���100mL����ƿ����ͷ�ιܣ���B����
C������ʱ����Ҫ������ƿ�м�������ˮ����������ʱ������ƿ�����������������ˮ������ҺŨ����Ӱ�죬��C����
D������ʱ���ӿ̶��ߣ�������Һ���ƫС������C=$\frac{n}{V}$��֪��ҺŨ��ƫ�ߣ���D��ȷ��
��ѡ��D��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���Ϥ���ƹ����ǽ���ؼ���ע������C=$\frac{n}{V}$�������ķ�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
1������������CrO5���Ľṹ����ͼ���ɴ˿���֪��CrO5����Ԫ�صĻ��ϼ�Ӧ���ǣ�������


| A�� | ȫ��-1�� | B�� | $\frac{4}{5}$Ϊ-1�ۣ�$\frac{1}{5}$Ϊ-2�� | ||
| C�� | ȫ��-2�� | D�� | $\frac{4}{5}$Ϊ-2�ۣ�$\frac{1}{5}$Ϊ-1�� |
8��Ϊ̽����������ƣ�Na2S2O3����Һ��ϡ���ᷴӦ��Ӱ�����أ�ij�����о�С������һϵ��ʵ�飬ʵ���������±���
�ش��������⣺
��1����ȡҺ�����õ��������������Ͳ��50mL��
��2��Ϊ����ijһ���ʴﵽij�ص�ֵ���õ�ʱ�䣬���ײ��������ʵ���C��
A��Na2S2O3Ũ�� B��H2SO4��Ũ�� C��S���ʵ��� D��SO2����
��3��ʵ��١��ڡ��۵ó��Ľ�������ͬ�¶��£���Ӧ��Ũ��Խ��Ӧ����Խ�죬���� V1=15mL��
��4��ʵ���ʱ�о��¶ȶԻ�ѧ��Ӧ�ٶȵ�Ӱ�죬�� V2=15mL��V3=10mL��
��5��Ϊ����ʵ���ķ�Һ��ʵ��С�����Һ�м���������NaOH��Һ����Һ�����ԣ�������Һ����壬д��������Ӧ�Ļ�ѧ����ʽ��6NaOH+3S=2Na2S+Na2SO3+3H2O��
| ��� | �¶� | 0.1mol•L-1Na2S2O3��Һ | 0.1mol•L-1H2SO4��Һ | H2O | �ﵽ��ͬ�̶����õ�ʱ��/s |
| �� | 25�� | 25mL | 25mL | 0mL | 4 |
| �� | 25�� | 25mL | 15mL | 10.0mL | 6 |
| �� | 25�� | V1 | 25mL | 10mL | 6 |
| �� | 45�� | V2 | 25mL | V3 | 2 |
��1����ȡҺ�����õ��������������Ͳ��50mL��
��2��Ϊ����ijһ���ʴﵽij�ص�ֵ���õ�ʱ�䣬���ײ��������ʵ���C��
A��Na2S2O3Ũ�� B��H2SO4��Ũ�� C��S���ʵ��� D��SO2����
��3��ʵ��١��ڡ��۵ó��Ľ�������ͬ�¶��£���Ӧ��Ũ��Խ��Ӧ����Խ�죬���� V1=15mL��
��4��ʵ���ʱ�о��¶ȶԻ�ѧ��Ӧ�ٶȵ�Ӱ�죬�� V2=15mL��V3=10mL��
��5��Ϊ����ʵ���ķ�Һ��ʵ��С�����Һ�м���������NaOH��Һ����Һ�����ԣ�������Һ����壬д��������Ӧ�Ļ�ѧ����ʽ��6NaOH+3S=2Na2S+Na2SO3+3H2O��
18����ͬ���pH=3��ǿ����Һ��������Һ�ֱ���������þ��ȫ��Ӧ������˵����ȷ���ǣ�������
| A�� | ���߲������������� | B�� | ǿ����Һ�����������ٶȿ� | ||
| C�� | ������Һ�����϶������ | D�� | ���Ƚ����߲����������� |
5��������������ȷ���ǣ�������
| A�� | ����������Ư�۴�������ˮ�����ߵ�����ԭ����ͬ | |
| B�� | ��������10mL pH=3�Ĵ�����Һ�м�ˮϡ�ͺ���Һ��$\frac{c��C{H}_{3}CO{O}^{-}��}{c��C{H}_{3}COOH��•c��O{H}^{-}��}$���� | |
| C�� | ��ˮ����ˮ����ˮ�ǻ���ˮ����ˮ�����Ǵ����� | |
| D�� | ���ó����ʯ��ˮ����̼���ƺ�̼������ |
2��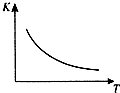 ��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺
��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺
��1��Q��0�����������=����������
��2������850��ʱ��Ӧ�����г���H2O��g����K ֵ���䣨���������С�����䡱����
��3���ϱ��� c2Ϊ0.18mol/L��CO��g����ת����Ϊ60%��
 ��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺
��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO��g��+H2O��g��?CO2 ��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ��K���¶�T�Ĺ�ϵ��ͼ��ʾ������һ�̶��ݻ����ܱ������У�850��ʱ����������Ӧ����������ڸ����ʵ�Ũ�� ��mol/L����ʱ��ı仯��ϵ�������֪��850��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=1.0����ش��������⣺| ʱ��/min | CO��g�� | H2O��g�� | CO2��g�� | H2��g�� |
| 0 | 0.200 | 0.300 | 0 | 0 |
| 2 | 0.138 | 0.238 | 0.062 | 0.062 |
| 3 | c1 | c2 | c3 | c4 |
| 4 | c1 | c2 | c3 | c4 |
��2������850��ʱ��Ӧ�����г���H2O��g����K ֵ���䣨���������С�����䡱����
��3���ϱ��� c2Ϊ0.18mol/L��CO��g����ת����Ϊ60%��
3����ͬ�¶���ͬ���ʵ���Ũ�ȵ�������Һ����NaHCO3 ��CH3COONa ��NaOH ��Ba��OH��2 ��NaHSO4��pHֵ�ɴ�С��˳���ǣ�������
| A�� | �ݢڢ٢ܢ� | B�� | �ݢ٢ڢۢ� | C�� | �ܢۢڢ٢� | D�� | �ܢۢ٢ڢ� |
