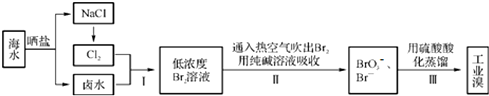
��Ŀ����
7��ij����Һ���л����ԪIV�ĺϳ���·���£�
��1����ķ���ʽC7H6O3�����к��������ŵ����Ʒ��ǻ����Ȼ�����Ӧ�ٵ�����Ϊȡ����Ӧ��
��2��CH2=CHCH2Br��NaOHˮ��Һ��Ӧ�Ļ�ѧ����ʽCH2=CHCH2Br+NaOH$\stackrel{��}{��}$CH2=CHCH2OH+NaBr��ע����������
��3����ɷ����Ӿ۷�Ӧ�����ò���Ľṹ��ʽ
 ��
����4���йػ�������II��˵���У�����ȷ����A��
A��1mol����ȼ����Ҫ����6mol O2 B��1mol����������2mol NaOH
C������ʹ���CCl4��Һ��ɫ D������ʹ����KMnO4��Һ��ɫ
��5��һ�������£�
 Ҳ����������Ʒ�Ӧ�۵ķ�Ӧ�������л���V��V�Ľṹ��ʽ��
Ҳ����������Ʒ�Ӧ�۵ķ�Ӧ�������л���V��V�Ľṹ��ʽ�� ��
��
���� ��1�������л���Ľṹ��ʽ�жϺ��е�Ԫ�������ԭ�Ӹ�������ȷ������ʽ�����������ŵ�����Ϊ���ǻ����Ȼ�����Ӧ��Ϊ��ķ��ǻ��ϵ��ⱻȡ���ķ�Ӧ��
��2���ȴ����ڼ���������ˮ������-OH��
��3��II����̼̼˫�����ɷ����Ӿ۷�Ӧ���ݴ���д�ṹ��ʽ��
��4�����ݻ�������II�Ľṹ�жϣ�
��5��������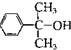 ��III����ȡ����Ӧ����-COO-��ˮ��
��III����ȡ����Ӧ����-COO-��ˮ��
��� �⣨1���ɽṹ��ʽ��֪��������к���7��C��6��H��3��O�������ʽΪC7H6O3����������з��ǻ����Ȼ�����Ӧ��Ϊ��ķ��ǻ��ϵ��ⱻȡ���ķ�Ӧ��
�ʴ�Ϊ��C7H6O3�����ǻ����Ȼ���ȡ����
��2���ȴ����ڼ���������ˮ������-OH��CH2=CH-CH2Br��NaOHˮ��Һ��Ӧ�Ļ�ѧ����ʽΪCH2=CHCH2Br+NaOH$\stackrel{��}{��}$CH2=CHCH2OH+NaBr��
�ʴ�Ϊ��CH2=CHCH2Br+NaOH$\stackrel{��}{��}$CH2=CHCH2OH+NaBr��
��3��II����̼̼˫�����ɷ����Ӿ۷�Ӧ�����ò���Ľṹ��ʽΪ
�ʴ�Ϊ��
��4��A����Ϊ��ķ���ʽΪC7H6O3����1mol����ȼ����Ҫ����7mol O2���ʴ���
B����Ϊ���з��ǻ����Ȼ�������1mol����������2molNaOH������ȷ��
C�����ݢ�Ľṹ������̼̼˫�������Ԣ���ʹ���CCl4��Һ��ɫ������ȷ��
D�����ݢ�Ľṹ������̼̼˫��������II��ʹ����KMnO4��Һ��ɫ������ȷ��
��ѡ��A��
��5���ɹ����ŵ�ת����֪��Ӧ������ȡ����Ӧ�������� ��III����ȡ����Ӧ����-COO-����Ӧ��Ϊ
��III����ȡ����Ӧ����-COO-����Ӧ��Ϊ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɣ������Ĺؼ�������ȷ�����л�������ŵ����ʣ�ע����ݹ����ŵ�ת���жϷ�Ӧ���ͣ��״���Ϊͬ���칹�����д��ע����ᣮ

 ��������������������ϵ�д�
��������������������ϵ�д�| A�� | SiO2 | B�� | NO | C�� | Al2O3 | D�� | SO3 |
| A�� | ��С�մ�����θ����ࣺ2H++CO32-�TH2O+CO2�� | |
| B�� | �Ȼ�����Һ�м��������ˮ��Al3++4NH3•H2O�TAlO2-+4NH4++2H2O | |
| C�� | ��NaHSO4��Һ�еμ�Ba��OH��2��Һ��ǡ��Ϊ���ԣ�2H++SO42-+Ba2++2OH-�TBaSO4��+2H2O | |
| D�� | ̼��������Һ�����������������Һ��Ca2++2HCO3-+2OH-�TCaCO3��+CO32-+2H2O |
| A�� | Na2O2 | B�� | NaHSO4 | C�� | HCl | D�� | H2O |
��1��ʵ������ȡ��Ȳ����ķ���ʽ��CaC2+2H2O��C2H2��+Ca��OH��2
��2���������������͵�����װʵ��װ�ã�
| ��� | 1 | 2 | 3 | 4 | 5 | 6 |
| ���ܼ����� |  |  |  |  |  |  |
| ÿ����Ƥ���϶����������� | ||||||
��3���������Ӻú���ʵ��ʱ�������в�����ÿ�����ֻ����һ�Σ�
�ٳ�ȡһ������ʯ����������3�У�������Ƥ���� �ڼ��װ�õ������� ��������6��5��ע������ˮ�� �ܴ�����3�ָ�������ʱ����ȡ����4��ˮ�����������2��ˮ��������Բ��ƣ� ��������������6�Ļ�����ʹˮ��ε���������������ʱ���رջ�������ȷ����˳����B
A���٢ۢڢݢ�B���ڢ٢ۢݢ�C���٢ۢݢܢ�
��4����ʵ���в���������������ŵ���ζ���Ҳⶨ���ƫ��������Ϊ��ʯ�к����������壬����������ͭ��Һ������������Һ���Գ�ȥ��