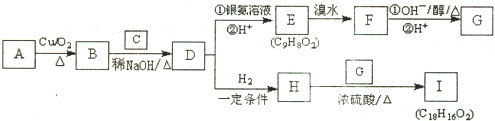
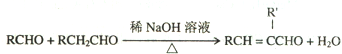
��Ŀ����
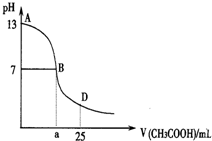 ��ͼΪ��25mL 0.1mol?L-1NaOH��Һ����εμ�0.2mol?L-1CH3COOH��Һ��������ҺpH�ı仯���ߣ���ش�
��ͼΪ��25mL 0.1mol?L-1NaOH��Һ����εμ�0.2mol?L-1CH3COOH��Һ��������ҺpH�ı仯���ߣ���ش���1��B����Һ�����ԣ����˾ݴ���Ϊ����B��ʱNaOH��CH3COOHǡ����ȫ��Ӧ�����ֿ����Ƿ���ȷ����ѡ��ǡ���
��2��AB���䣬c��OH-����c��H+������c��OH-����c��CH3COO-����С��ϵ��
A��c��OH-��һ������c��CH3COO-��
B��c��OH-��һ��С��c��CH3COO-��
C��c��OH-��һ������c��CH3COO-��
D��c��OH-�����ڡ�С�ڻ����c��CH3COO-��
��3����D��ʱ����Һ��c��CH3COO-��+c��CH3COOH��
��4�������£���VmL��0.1000mol?L-1����������Һ��μ��뵽20.00mL��0.1000mol?L-1������Һ�У���ַ�Ӧ���ش��������⣮��������Һ����ı仯��
�������ҺpH=7����ʱV��ȡֵ
�����V=40.00�����ʱ��Һ��c��OH-��-c��H+��-c��CH3COOH��=
���㣺�����ʱ�Ķ����жϼ��й�ph�ļ���
ר�⣺
��������1��NaOH��CH3COOHǡ����ȫ��Ӧ�����ɴ����ƣ�������Ϊǿ�������Σ�ˮ�����Һ�Լ��ԣ�pH��7��
��2����AB�����ڣ�����CH3COOH��NaOHǡ����ȫ��Ӧ�Լ�CH3COOH���㣬����������Һ������Һ�Լ������ֿ����ԣ�
��3����D��ʱ��NaOH��CH3COOH��Ӧ��ʣ��CH3COOH����Һ�����Ϊ��Ũ�ȵ�CH3COOH��CH3COONa�Ļ���
��4������Һ��������Ǹ�����Һ��H+Ũ����OH-Ũ�ȵ���Դ�С�жϵģ�ֻҪ��Һ��c��H+��=c��OH-������Һ�ͳ����ԣ�CH3COOH��������ʣ�����̶Ȳ���NaOH��ǿ����ʣ���ȫ���룬��Ӧ���ɵ���������ǿ��������ˮ��ʼ��ԣ�����Һ�����ԣ����ټӼ������Һ������pH=7c��H+��=c��OH-����ϵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-�����н��
�ڸ��ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�c��Na+��=2[c��CH3COO-��+c��CH3COOH��]���н��
��2����AB�����ڣ�����CH3COOH��NaOHǡ����ȫ��Ӧ�Լ�CH3COOH���㣬����������Һ������Һ�Լ������ֿ����ԣ�
��3����D��ʱ��NaOH��CH3COOH��Ӧ��ʣ��CH3COOH����Һ�����Ϊ��Ũ�ȵ�CH3COOH��CH3COONa�Ļ���
��4������Һ��������Ǹ�����Һ��H+Ũ����OH-Ũ�ȵ���Դ�С�жϵģ�ֻҪ��Һ��c��H+��=c��OH-������Һ�ͳ����ԣ�CH3COOH��������ʣ�����̶Ȳ���NaOH��ǿ����ʣ���ȫ���룬��Ӧ���ɵ���������ǿ��������ˮ��ʼ��ԣ�����Һ�����ԣ����ټӼ������Һ������pH=7c��H+��=c��OH-����ϵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-�����н��
�ڸ��ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�c��Na+��=2[c��CH3COO-��+c��CH3COOH��]���н��
���
�⣺��1��NaOH��CH3COOHǡ����ȫ��Ӧ��NaOH+CH3COOH=CH3COONa+H20�����ɵĴ�����Ϊǿ�������Σ���Һ�Լ��ԣ�pH��7������AB֮�䣬
�ʴ�Ϊ����AB��
��2����AB�����ڣ�c��OH-����c��H-����˵����Һ�Լ��ԣ���NaOH��CH3COOHǡ�÷�Ӧʱ���Լ��ԣ���ʱ���ɵ���Һ����Ϊ�����ƣ�c��OH-��С��c��CH3COO-������NaOH��CH3COOH��Ӧ��ʣ��NaOH����Һ��Ȼ�Լ��ԣ���ʱ��ʣ���NaOH���ܴ���c��OH-������c��CH3COO-����Ҳ�п���ʣ���NaOH��CH3COONa��CH3COO-ˮ��֮��ʣ���CH3COO-��Ũ����ȣ�
��ѡD��
��3����D��ʱ����Ӧ��CH3COOHʣ�࣬��Һ�����Ϊ��Ũ�ȵ�CH3COOH��CH3COONa�Ļ������������غ㣬��ʱ��c��CH3COO-��+c��CH3COOH��=2c��Na+����
�ʴ�Ϊ��=��
��4����CH3COOH��������ʣ�����̶Ȳ���NaOH��ǿ����ʣ���ȫ���룬��Ӧ���ɵ���������ǿ�������Σ�ˮ��ʼ��ԣ�����Һ������pH=7�����ټӼ���Գ����£���V mL��0.1000mol?L-1����������Һ��μ��뵽20.00mL��0.1000mol?L-1������Һ�У���ַ�Ӧ��V��20.00mL��Һ������pH=7��c��H+��=c��OH-�������ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-����c��H+��=c��OH-������Һ�е�����Ϊ��������Һ��ˮ�ĵ��������ģ�����c��Na+��=c��CH3COO-����c��H+��=c��OH-����
�ʴ�Ϊ������c��Na+��=c��CH3COO-����c��H+��=c��OH-����
�ڸ��ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�c��Na+��=2[c��CH3COO-��+c��CH3COOH��]���õ�c��H+��+c��CH3COO-��+2c��CH3COOH��=c��OH-������c��OH-��-c��H+��-c��CH3COOH��=c��CH3COO-��+c��CH3COOH������Ӧ����Һ�������Ϊ60mL����c��CH3COO-��+c��CH3COOH���T
=
mol/L����0.033mol/L��
�ʴ�Ϊ��
��0.033��
�ʴ�Ϊ����AB��
��2����AB�����ڣ�c��OH-����c��H-����˵����Һ�Լ��ԣ���NaOH��CH3COOHǡ�÷�Ӧʱ���Լ��ԣ���ʱ���ɵ���Һ����Ϊ�����ƣ�c��OH-��С��c��CH3COO-������NaOH��CH3COOH��Ӧ��ʣ��NaOH����Һ��Ȼ�Լ��ԣ���ʱ��ʣ���NaOH���ܴ���c��OH-������c��CH3COO-����Ҳ�п���ʣ���NaOH��CH3COONa��CH3COO-ˮ��֮��ʣ���CH3COO-��Ũ����ȣ�
��ѡD��
��3����D��ʱ����Ӧ��CH3COOHʣ�࣬��Һ�����Ϊ��Ũ�ȵ�CH3COOH��CH3COONa�Ļ������������غ㣬��ʱ��c��CH3COO-��+c��CH3COOH��=2c��Na+����
�ʴ�Ϊ��=��
��4����CH3COOH��������ʣ�����̶Ȳ���NaOH��ǿ����ʣ���ȫ���룬��Ӧ���ɵ���������ǿ�������Σ�ˮ��ʼ��ԣ�����Һ������pH=7�����ټӼ���Գ����£���V mL��0.1000mol?L-1����������Һ��μ��뵽20.00mL��0.1000mol?L-1������Һ�У���ַ�Ӧ��V��20.00mL��Һ������pH=7��c��H+��=c��OH-�������ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-����c��H+��=c��OH-������Һ�е�����Ϊ��������Һ��ˮ�ĵ��������ģ�����c��Na+��=c��CH3COO-����c��H+��=c��OH-����
�ʴ�Ϊ������c��Na+��=c��CH3COO-����c��H+��=c��OH-����
�ڸ��ݵ���غ�c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�c��Na+��=2[c��CH3COO-��+c��CH3COOH��]���õ�c��H+��+c��CH3COO-��+2c��CH3COOH��=c��OH-������c��OH-��-c��H+��-c��CH3COOH��=c��CH3COO-��+c��CH3COOH������Ӧ����Һ�������Ϊ60mL����c��CH3COO-��+c��CH3COOH���T
| 0.1000mol/L��20ml |
| 60ml |
| 1 |
| 30 |
�ʴ�Ϊ��
| 1 |
| 30 |
������������Ҫ�������к͵ζ������Լ���Һ��PH�ļ��㣬��ѧƽ����й�֪ʶ����Ŀ�Ѷ��еȣ�ע����������кͷ�Ӧ����ҺpH�ļ��㷽�����к͵ζ���������������������ѧ�����Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
1molH2��O2��ȼ������Һ̬ˮ�ų�285.8kJ���������������Ȼ�ѧ����ʽ��ȷ���ǣ�������
| A��2H2��g��+O2��g��=2H2O��l����H=+571.6kJ?mol-1 | ||
| B��2H2��g��+O2��g��=2H2O��l������H=-571.6kJ?mol-1 | ||
C��H2��g��+
| ||
D��H2��g��+
|
��֪���ᣨHA�������Աȼ��ᣨHB�����������ʵ���Ũ�Ⱦ�Ϊ0.1mol?L-1��NaA��NaB�����Һ�У�����������ȷ���ǣ�������
| A��c��OH-����c��HA����c��HB����c��H+�� |
| B��c��OH-����c��A-����c��B-����c��H+�� |
| C��c��OH-����c��B-����c��A-����c��H+�� |
| D��c��OH-����c��HB����c��HA����c��H+�� |
�����ᡢ���ᡢ��������ᶼ��ǿ�ᣬ��������ˮ��Һ�в��������ij�¶������������ڱ������еĵ��볣����
�����ϱ����ж�����˵���в���ȷ���ǣ�������
| �� | HClO4 | H2SO4 | HCl | HNO3 |
| Ka | 1.6��10-5 | 6.3��10-9 | 1.6��10-9 | 4.2��10-10 |
| A���ڱ��������������ᶼû����ȫ���� |
| B���ڱ������и�������������������ǿ���� |
| C���ڱ�����������ĵ��뷽��ʽΪ H2SO4=2H++SO42- |
| D��ˮ�������������ǿ��û����������������������������������ǿ�� |
��״���£���V L�������е���������ΪN�����ӵ������ɱ�ʾΪ��������
A��
| ||
B��
| ||
C��
| ||
D��
|