��Ŀ����
15����ҵ����������β���ŷų���SO2��NOx�ȣ����γ���������Ҫ���أ������ɿ����еĻҳ������ᡢ���ᡢ�л�̼�⻯����������γɵ���������1��SO2���̳��Ĵ����γ�����ķ�Ӧ����ʽ��2SO2+2H2O+O2=2H2SO4��
��2����֪2SO2 ��g��+O2 ��g��?2SO3��g����H=-196kJ/mol����߷�Ӧ��SO2��ת���ʣ��Ǽ���SO2�ŷŵ���Ч��ʩ��
��T�¶�ʱ����2L�ݻ��̶�������ܱ������м���2.0mol SO2��1.0mol O2��5min��Ӧ�ﵽƽ�⣬���������ת����Ϊ50%����ԣ�O2��=0.05mol/��L•min����
���ڢٵ������£��жϸ÷�Ӧ�ﵽƽ��״̬�ı�־��bde������ĸ����
a��SO2��O2��SO3���ߵ�Ũ��֮��Ϊ2��1��2 b�������������ѹǿ����
c�������ڻ��������ܶȱ��ֲ��� d��SO3�����ʵ������ٱ仯
e��SO2 ���������ʺ�SO3�������������
��3�������е�SO2������NaOH��Һ���գ������õ�Na2SO3��Һ���е�⣬��ѭ������NaOH��ͬʱ�õ�H2SO4����ԭ����ͼ��ʾ�����缫����Ϊʯī��

��ͼ��a��Ҫ���ӵ�Դ�ģ������������������C�����������������ᣮ
��SO32-�ŵ�ĵ缫��ӦʽΪSO32--2e-+H2O=SO42-+2H+��
�۵�������������12.6gNa2SO3�����������仯������Ϊ4.4g������ù���������Һ��������ܱգ���
���� ��1��SO2���̳��Ĵ����γ�������2SO2+2H2O+O2=2H2SO4��
��2���ٸ�������ʽ���ת�������ٸ��ݷ�Ӧ���ʵĶ���ʽ���㣻
�ڻ�ѧƽ��ı�־�����淴Ӧ������ͬ������ֺ������ֲ��䣻
��3���ٸ��ݵ������������ӵ��ƶ������������������������жϢ�ͼ��a��Ҫ���ӵ�Դ�ĸ�����SO32-������ʧȥ���ӱ��SO42-������C��������������H2SO4��
��SO32-ʧȥ���ӱ�������SO42-��
�۵��������������������ˮ��Ϊ�������ƣ��������仯�����������������Ƶ�������ȥ����ˮ�����������ݵ����غ���㣮
��� �⣺��1��SO2���̳��Ĵ����γ�������2SO2+2H2O+O2=2H2SO4��
�ʴ�Ϊ��2SO2+2H2O+O2=2H2SO4��
��2����T1�¶�ʱ����2L���ܱ������м���4.0molSO2��2.0molO2��5min��Ӧ�ﵽƽ�⣬���������ת����Ϊ50%��
2SO2��g��+O2?2SO3��g����H��0
��ʼ����mol�� 2.0 1.0 0
�仯����mol��2.0��50% 0.5 0.5
ƽ������mol�� 1.0 0.5 0.5
ǰ5min��O2��ƽ����Ӧ����=$\frac{0.5mol}{2L��5min}$=0.05mol/��L•min����
�ʴ�Ϊ��0.05mol/��L•min����
�ڻ�ѧƽ��ı�־�����淴Ӧ������ͬ������ֺ������ֲ��䣻
a�����ʵ�Ũ�ȹ�ϵ����ʼ����ת�����йأ�SO2��O2��SO3���ߵ�Ũ��֮��Ϊ2��1��2 ����˵����Ӧ�ﵽƽ��״̬����a�����ϣ�
b����Ϊ�÷�ӦΪ�������ʵ�������ķ�Ӧ�����º��������£�ѹǿΪ�������������������ѹǿ����˵���ﵽƽ��״̬����b���ϣ�
c����Ӧǰ�������������䣬����������䣬�ܶ��ڷ�Ӧ�����к�ƽ��״̬�����䣬�����л��������ܶȱ��ֲ��䲻��˵�����ôﵽƽ��״̬����c�����ϣ�
d��SO3�����ʵ������ٱ仯����˵����Ӧ�ﵽƽ��״̬����d���ϣ�
e��SO2���������ʺ�SO3�������������˵�����淴Ӧ������ͬ����˵����Ӧ�ﵽƽ��״̬����e���ϣ�
�ʴ�Ϊ��bde��
��3���ٸ��ݵ������������ӵ��ƶ������������������������жϢ�ͼ��a��Ҫ���ӵ�Դ�ĸ�����SO32-������ʧȥ���ӱ��SO42-������C��������������H2SO4��
�ʴ�Ϊ���������
��SO32-ʧȥ���ӱ�������SO42-���缫��ӦʽΪ��SO32--2e-+H2O=SO42-+2H+��
�ʴ�Ϊ��SO32--2e-+H2O=SO42-+2H+��
�۵��������������������ˮ��Ϊ�������ƣ��������仯�����������������Ƶ�������ȥ����ˮ��������
���ݵ����غ㣺
2e-��Na2SO3����2H2O��2NaOH���������仯������
126g 36g 80g 44g
12.6g x
x=4.4g
������12.6g Na2SO3�����������仯������Ϊ4.4 g��
�ʴ�Ϊ��4.4��
���� ���⿼���˻�ѧ��Ӧ���ʵļ��㡢��ѧƽ����жϡ��绯ѧ�ȣ�������ض�ѧ������֪ʶ��ѵ���ͼ��飬���������ѧ�����������ѧ֪ʶ���ʵ�������������

| A�� | ƻ�����ڿ����о��ñ�ƺ�ֽ�ž��ñ��ԭ������ | |
| B�� | H2O2�����·������е�CxHy��CO�ȣ�����Ҫ��������H2O2�Ļ�ԭ�� | |
| C�� | ���õ���������Ƴ̳����Խ���PM2.5���ŷţ���С������Ⱦ | |
| D�� | ��ʯ�ҡ����ۡ��轺��ʳƷ��װ�г��õĸ���� |

| A�� | ����ʽΪC7H8O5 | |
| B�� | �����к���2�ֹ����ţ��ɷ����ӳɡ�������Ӧ | |
| C�� | ��ͬ���칹���п��ܺ�������ȩ���������ܺ��б��� | |
| D�� | �û������������� Na��NaOH��Ӧ�õ������β�����ͬ |
| A�� | 30% | B�� | 50% | C�� | 70% | D�� | 90% |

| A�� | ����̼����һ���������� | |
| B�� | 0.12g����̼���к���6.02��l022��̼ԭ�� | |
| C�� | ����̼����ʯī��Ϊͬλ�� | |
| D�� | ����̼����̼ԭ�Ӽ��Թ��ۼ���� |

 ��
��
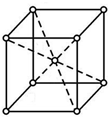 X��Y��Z��W����Ԫ�������ڱ�ǰ������Ԫ�أ�XԪ��ԭ�Ӻ�����16�ֲ�ͬ�˶�״̬�ĵ��ӣ�Y��ԭ��������X��1��Zԭ�ӵ�M�ܲ�����4��δ�ɶԵ��ӣ�W�ļ۲�����Ų�ʽΪndn+5��n+1��sn-1��
X��Y��Z��W����Ԫ�������ڱ�ǰ������Ԫ�أ�XԪ��ԭ�Ӻ�����16�ֲ�ͬ�˶�״̬�ĵ��ӣ�Y��ԭ��������X��1��Zԭ�ӵ�M�ܲ�����4��δ�ɶԵ��ӣ�W�ļ۲�����Ų�ʽΪndn+5��n+1��sn-1�� ��WԪ�ص�����Ϊ����
��WԪ�ص�����Ϊ����