��Ŀ����
11����ij�¶�ʱ��pH=3��ijˮ��Һ��c��OH-��=10-9 mol/L�����и��¶��µ��ķ���Һ����pH=2��CH3COOH����0.01mol/L��HCl����pH=11�İ�ˮ����pH=11��NaOH��Һ������˵����ȷ���ǣ�������| A�� | ����ˮ�ĵ���̶���С������ˮ�ĵ���̶���� | |
| B�� | ���ڡ��ۻ�ϣ���pH=7����������Һ�������=�� | |
| C�� | ���ķ���Һϡ����ͬ�ı�������Һ��pH���ۣ��ܣ��ڣ��� | |
| D�� | ���١��ܻ�ϣ�����c��CH3COO-����c��H+����������Һһ���ʼ��� |
���� ��ij�¶�ʱ��pH=3��ijˮ��Һ��c��OH-��=10-9 mol/L������¶���ˮ�����ӻ�����K=10-3.10-9=10-12��
��������Һ��pH=6�����¶��£�pH=2�Ĵ�����c��H+��=10-2 mol/L��0.01mol/L������c��H+��=10-2 mol/L��pH=11�İ�ˮ��NaOH��Һ��c��OH-��=0.1mol/L������ˮŨ�ȴ���NaOHŨ�ȣ�
A����������ˮ���룬����������Ũ�Ȼ��������������Ũ��Խ��������ˮ����̶�Խ��
B��������ߵ������ϣ���ˮ��������Һ�ʼ��ԣ���Һ��pH��7��
C��ϡ����ͬ�ı�����pH��6���ڣ��٣��ۣ��ܣ�6��
D�����١��ܻ�ϣ�����c��CH3COO-����c��H+�������ݵ���غ�֪����Һ�д���c��OH-����c��Na+����Ҫ������Һ��c��OH-����c��H+������Դ�С�ж���Һ����ԣ�
��� �⣺��ij�¶�ʱ��pH=3��ijˮ��Һ��c��OH-��=10-9 mol/L������¶���ˮ�����ӻ�����K=10-3.10-9=10-12��
��������Һ��pH=6�����¶��£�pH=2�Ĵ�����c��H+��=10-2 mol/L��0.01mol/L������c��H+��=10-2 mol/L��pH=11�İ�ˮ��NaOH��Һ��c��OH-��=0.1mol/L������ˮŨ�ȴ���NaOHŨ�ȣ�
A����������ˮ���룬����������Ũ�Ȼ��������������Ũ��Խ��������ˮ����̶�Խ�٢���������Ũ����ȡ��ۢ�������������Ũ����ȣ��Ңۡ���������������Ũ�ȴ��ڢ٢���������Ũ�ȣ���������ˮ����̶ȣ��ۢ�С�ڢ٢ڣ���A����
B��������ߵ������ϣ���ˮ���ʵ���ԶԶ�������ᣬ��Һ�ʼ��ԣ���Һ��pH��7����B����
C��ϡ����ͬ�ı�����pH��6���ڣ��٣��ۣ��ܣ�6��������Һ��pH���ۣ��ܣ��ڣ��٣���C��ȷ��
D�����١��ܻ�ϣ�����c��CH3COO-����c��H+�������ݵ���غ�֪����Һ�д���c��OH-����c��Na+���������Һ��c��OH-����c��H+������Һ�ʼ��ԣ������Һ��c��OH-����c��H+��������Һ�����ԣ���D����
��ѡC��
���� ���⿼��������ʵĵ��롢�������Һ�����жϵ�֪ʶ�㣬Ϊ��Ƶ���㣬��ȷ������ʵ����ص��ǽⱾ��ؼ���ע����¶���ˮ�����ӻ���������10-14���״�ѡ����B����������˼ά�����жϼ��ɣ���Ŀ�ѶȲ���

��ҵ�������ظ����ƾ��壨Na2Cr2O7•2H2O�����Ը�����[��Ҫ�ɷ֣�FeCr2O4���Ǹ���������]Ϊԭ�ϣ�����Ҫ�������£�
��4FeCr2O4+8Na2CO3+7O2$\frac{\underline{\;����\;}}{\;}$8Na2CrO4+2Fe2O3+8CO2
����H2SO4������ҺpH��ʹNa2CrO4ת��ΪNa2Cr2O7
��1����ҵ�ϲ�����ڷ�Ӧ�������費�Ͻ��裬��Ŀ��������Ӧ��ĽӴ�������ӿ췴Ӧ���ʣ�
��2����֪��Cr2O72-+H2O?2CrO42-+2H+��д�������Na2CrO4ת��ΪNa2Cr2O7�Ļ�ѧ����ʽ2Na2CrO4+H2SO4�TNa2C2O7+Na2SO4+H2O��
��ʽ������[Fe��OH��SO4]��һ��������ˮ����������Ч����������ҽҩ��Ҳ���������������������Ѫ����ҵ�����÷���м������������������������������ʽ�������Ĺ���������ͼ��

��֪������������������������ʽ����ʱ��Һ��pH������
| ������ | Fe��OH��3 | Fe��OH��2 | A1��OH��3 |
| ��ʼ���� | 2.3 | 7.5 | 3.4 |
| ��ȫ���� | 3.2 | 9.7 | 4.4 |
��4����������NaHCO3��Ŀ���ǵ�����Һ��pH��Ӧ����pH�ķ�Χ����Ϊ[4.4��7.5����
��5����ʵ�������У���ӦII��ͬʱͨ��O2�Լ���NaNO2����������ͨ��2.8L O2����״���������൱�ڽ�ԼNaNO2������Ϊ34.5g��
��6����ʽ����������ˮ�������[Fe��OH��]2+���ӣ��ɲ���ˮ������[Fe2��OH��4]2+�ۺ����ӣ���ˮ�ⷴӦ�����ӷ���ʽΪ2[Fe��OH��]2++2H2O�T[Fe2��OH��4]2++2H+��
�����Ļ�����Ҳ���Ʊ�����﮵�ص���Ҫԭ�ϣ���֪LiFePO4��ط�ӦΪFePO4+Li $?_{���}^{�ŵ�}$LiFePO4������еĹ������ʿɴ���Li+����д���õ�س��ʱ������ӦʽLiFePO4-e-�TFePO4+Li+���������Ըõ��Ϊ��Դ���500mL����ʳ��ˮ��������0.35g Liʱ����Һ��pHΪ13��������Һ������仯����
��1����Fe��OH��3��NaClO��Һ��ϣ����Ƶ�Na2FeO4����ƽ�������ӷ���ʽ��
2Fe��OH��3+3ClO-+OH-�T2FeO42-+3Cl-+5H2O
��2����Fe��������Cu��������NaOH��Һ�����Һ���õ��ķ���Ҳ���Ƶ�Na2FeO4����������������ķ�ӦʽΪFe+8OH--6e-=FeO42-+4H2O��
��3��ij�غ�ˮ��Ʒ��Na2FeO4�������������Ӽ���Ũ�����±���ʾ��H+��OH-δ�г�����
| ���� | SO42- | Mg2+ | Fe3+ | Na+ | Cl- |
| Ũ�ȣ�mol/L�� | a | 0.05 | 0.10 | 0.50 | 0.58 |
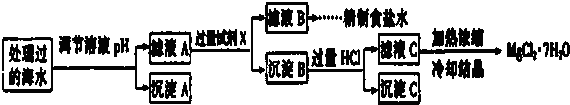
ע������Ũ��С��10-5mol/L ����Ϊ�����Ӳ����ڣ�Ksp[Fe��OH��3]=1.0��10-38��Ksp[Mg��OH��2]=1.0��10-12��������������Һ����ı仯���Բ��ƣ�
�ٱ����е�a��0.16 ���������=����������
�ڳ���A�����ΪFe��OH��3���ѧʽ�����ڵ�����ҺpHʱ��������Ӧ���ڵ�pH�ķ�Χ��3��pH��9��
�ۼ���Ĺ����Լ�XΪBa��OH��2 ���ѧʽ����
�ܼ������HCl������ΪʹMg��OH��2ȫ���ܽ�ת��ΪMgCl2���ڼ���Ũ����ҺCʱ����Mg2+ˮ�⣮
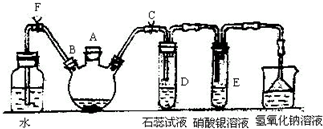 ʵ�����Ʊ��屽������ͼ��ʾ��װ�ã���д���пհף�
ʵ�����Ʊ��屽������ͼ��ʾ��װ�ã���д���пհף� ��
��

 ��
��