��Ŀ����
3��X��Y��Z��W��M�����ֶ���������Ԫ�أ����ǵĺ˵����������������X��Wͬ���壬Wԭ�ӵĵ���������Y��Z������������֮�ͣ�Z2-��M3+������ͬ�ĵ��Ӳ�ṹ������˵����ȷ���ǣ�������| A�� | X��Y��Z�γɵĻ������в����ܼ������Ӽ� | |
| B�� | X��Z��M��Y��Z��W�����γ�ԭ�Ӹ�����Ϊ1��1�Ļ����� | |
| C�� | Y��Z��W��M�γɵļ����Ӱ뾶��С��ϵΪY��Z��M��W | |
| D�� | W��Cl�γɻ�������۵����M��Cl�γɻ�������۵� |
���� X��Y��Z��W��M�����ֶ���������Ԫ�أ����ǵĺ˵������������Z2-��M3+������ͬ�ĵ��Ӳ�ṹ�����Ӻ���2�����Ӳ㣬��ZΪ��Ԫ�ء�MΪAlԪ�أ�X��Wͬ���壬���X��W��ԭ����������Ԫ��ԭ��������С��ϵ����W���ڵ������ڣ�Wԭ�ӵĵ���������Y��Z�����������֮�ͣ�Y������������С����Ԫ�أ���������������С��12����WΪNaԪ�أ�YΪ��Ԫ�أ�XΪ��Ԫ�ػ�LiԪ�أ��ݴ˽��н��
��� �⣺X��Y��Z��W��M�����ֶ���������Ԫ�أ����ǵĺ˵������������Z2-��M3+������ͬ�ĵ��Ӳ�ṹ�����Ӻ���2�����Ӳ㣬��ZΪ��Ԫ�ء�MΪAlԪ�أ�X��Wͬ���壬���X��W��ԭ����������Ԫ��ԭ��������С��ϵ����W���ڵ������ڣ�Wԭ�ӵĵ���������Y��Z�����������֮�ͣ�Y������������С����Ԫ�أ���������������С��12����WΪNaԪ�أ�YΪ��Ԫ�أ�XΪ��Ԫ�ػ�LiԪ�أ�
A��X��Y��Z�γɵĻ��������Ϊ̼��泥�������мȺ������Ӽ��ֺ��й��ۼ�����A����
B��X��Z�����γ�H2O2��M��Y�����γ�AlN��Z��W���γ�Na2O2�����Ǿ����γ�ԭ�Ӹ�����Ϊ1��1�Ļ������B��ȷ��
C�����Ӳ�ṹ��ͬ�˵����Խ�����Ӱ뾶ԽС�������Ӱ뾶N3-��O2-��Na+��Al3+����Y��Z��W��M�γɵļ����Ӱ뾶��С��ϵΪ��Y��Z��W��M����C����
D��NaCl�����Ӿ��壬AlCl3�Ƿ��Ӿ��壬NaCl�۵���ߣ���D����
��ѡB��
���� ���⿼��ṹ����λ�ù�ϵ������ṹ�����ʡ��뾶�Ƚϵ�֪ʶ����Ŀ�Ѷ��еȣ��ƶ�Ԫ���ǽ���Ĺؼ���ͻ�ƿ�ΪZ��M���ӵ��Ӳ�ṹ��ͬ��ע������ԭ�ӽṹ��Ԫ�����ڱ���Ԫ��������֮��Ĺ�ϵ��

 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д� -CH��CH3��2]���ӵ�һ��ȡ�����ͬ���칹�����Ŀ�У�������
-CH��CH3��2]���ӵ�һ��ȡ�����ͬ���칹�����Ŀ�У�������| A�� | 3�� | B�� | 4�� | C�� | 5�� | D�� | 6�� |
| A�� | ����ˮ���������ܵ�������Ϊˮ�����Լ��� | |
| B�� | ���Ƶ�ˮʱ��������������KI��Ŀ���Ƿ�ֹI2����ԭ | |
| C�� | ���ữ�ĸ��������H2O2����ȡ����������KMnO4������ | |
| D�� | ����[KAl��SO4��2•12H2O]��ˮ��Һ�ȣ�Cl2����ˮ��ԭ������ͬ |

ʵ�鷽����һ�����ļױ���KMnO4��Һ��100�淴Ӧһ��ʱ���ֹͣ��Ӧ�����������̷����������ͻ���δ��Ӧ�ļױ���

��֪�������������122���۵�122.4�棬��25���95��ʱ�ܽ�ȷֱ�Ϊ0.3g��6.9g�����������л��ﶼ�й̶��۵㣮
��1��������Ϊ��Һ��������Ϊ����
��2����ɫҺ��A�Ǽױ������Լ���A���Լ�������KMnO4��Һ����������ɫ��Һ��ɫ��
��3���ⶨ��ɫ����B���۵㣬��������115�濪ʼ�ۻ����ﵽ130��ʱ�����������ۣ���ͬѧ�Ʋ��ɫ����B�DZ�������KCl�Ļ�����������·��������ᴿ�ͼ��飬ʵ���������Ʋ���ȷ�����ڴ������ɱ������ݣ�
| ��� | ʵ�鷽�� | ʵ������ | ���� |
| �� | ����ɫ����B����ˮ�У������ܽ⣬ ��ȴ������ | �õ���ɫ�������ɫ��Һ | |
| �� | ȡ������Һ���Թ��У����������������ữ��AgNO3��Һ | ���ɰ�ɫ���� | ��Һ��Cl- |
| �� | �����ɫ���壬����ʹ���ۻ��������۵� | �۵�Ϊ122.4�� | ��ɫ�����DZ����� |
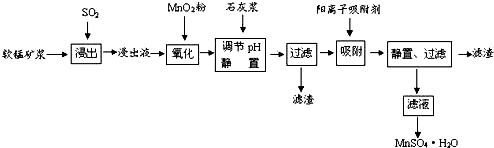
��֪������Һ��pH��2�����еĽ���������Ҫ��Mn2+��������������Fe2+��Al3+��Ca2+��Pb2+���� �����������ӣ�PbO2�������Դ���MnO2��PbSO4��һ�������ʣ��йؽ������ӵİ뾶���γ� �����������ʱ��pH������������������������������ӵ�Ч������ͼ��

| ���� | ���Ӱ뾶��pm�� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
| Fe2+ | 74 | 7.6 | 9.7 |
| Fe3+ | 64 | 2.7 | 3.7 |
| Al3+ | 50 | 3.8 | 4.7 |
| Mn2+ | 80 | 8.3 | 9.8 |
| Pb2+ | 121 | 8.0 | 8.8 |
| Ca2+ | 99 | - | - |
��1��д���������������ɷ�Ӧ�Ļ�ѧ����ʽSO2+MnO2=MnSO4��
��2��������������Ҫ��Ӧ�����ӷ���ʽ2Fe2++MnO2+4H+=2Fe3++Mn2++2H2O��
��3�����������Һ���м���ʯ�ҽ������ڵ���pHֵ���˴�����pHֵ�õ���������pH�ƣ�Ӧ����pH�ķ�ΧΪ4.7��pH��8.3��
��4�������������������ڳ�ȥ���ʽ�������•������ͼ������Ϣ�ش𣬾�������������������Ч��������������ʱ�䡢�������ӵİ뾶���������ӵĵ�ɵȣ����������ȥ����Ҫ����Ϊ��Pb2+��Ca2+��
��5��CaSO4��һ�������ʣ���֪Ksp��CaSO4��=9.10��10-6���ֽ�c mol•L-1CaCl2��Һ��2.00��10-2
mol•L-1Na2SO4��Һ�������� ����������ı仯���������ɳ���ʱ��c����Сֵ��1.82��10-3mol•L-1��
| ѡ�� | ���� | ���� | ���ӷ��� |
| A | Cu | CuO | �ڿ��������� |
| B | CaO | Ca��OH��2 | ����������ϡ���� |
| C | H2 | CO | ͨ��O2����ȼ |
| D | O2 | H2O | ͨ��Ũ����ϴ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ���Ӽ���������һ�������� | B�� | ռ�ݾ����������ԭ�� | ||
| C�� | ��ѧ���ǹ��ۼ� | D�� | ���߶��� |
 �����Ǹ�ˮ����{[Cr��CH3COO��2��]2•2H2O�����ɫ����}��һ���������ռ���ͨ���Զ�������Ӵ��ڣ���������ˮ���ѣ��������Ҵ������ᣮʵ�����Ʊ������Ǹ�ˮ�����װ����ͼ��ʾ���漰�Ļ�ѧ����ʽ���£�
�����Ǹ�ˮ����{[Cr��CH3COO��2��]2•2H2O�����ɫ����}��һ���������ռ���ͨ���Զ�������Ӵ��ڣ���������ˮ���ѣ��������Ҵ������ᣮʵ�����Ʊ������Ǹ�ˮ�����װ����ͼ��ʾ���漰�Ļ�ѧ����ʽ���£�