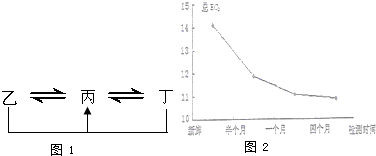
��Ŀ����
A��B��C��D��E��F����������һ��������������ͼ��ʾ���ת����ϵ�����з�Ӧ������������������
��1������Ӧ�١��ڡ��۾�Ϊ��Һ�е��û���Ӧ��A��D��EΪ�ڢ�A��Ԫ�ص��ʡ���A��D��E����������ǿ������˳��Ϊ(�û�ѧʽ��ʾ)________________________��
��2����B��ijԪ��R���⻯�B�ķе������Rͬ���������Ԫ���⻯��ķе㣬��B�ڳ�����ΪҺ̬��C��һ���д��ԵĻ����E��һ����ɫ����ζ���ж����塣��Ӧ�ٵĻ�ѧ����ʽ________________________________________���۵Ļ�ѧ����ʽ_____________________________��
��3����B�ɶ�����Ԫ��X��Y��ɣ���X��Yԭ�Ӹ�����Ϊ1��1��B�����мȺ����Ӽ����ֺ��Ǽ��Թ��ۼ�����ɫֲ��Ĺ�����úͺ������ÿ�ʵ����Ȼ����D��E��ѭ������B�ĵ���ʽΪ_________________����Ӧ�ٵ����ӷ���ʽ______________________________��
��2����B��ijԪ��R���⻯�B�ķе������Rͬ���������Ԫ���⻯��ķе㣬��B�ڳ�����ΪҺ̬��C��һ���д��ԵĻ����E��һ����ɫ����ζ���ж����塣��Ӧ�ٵĻ�ѧ����ʽ________________________________________���۵Ļ�ѧ����ʽ_____________________________��
��3����B�ɶ�����Ԫ��X��Y��ɣ���X��Yԭ�Ӹ�����Ϊ1��1��B�����мȺ����Ӽ����ֺ��Ǽ��Թ��ۼ�����ɫֲ��Ĺ�����úͺ������ÿ�ʵ����Ȼ����D��E��ѭ������B�ĵ���ʽΪ_________________����Ӧ�ٵ����ӷ���ʽ______________________________��
��1��Cl2��Br2��I2
��2��3Fe+4H2O Fe3O4+4H2��CO+H2O
Fe3O4+4H2��CO+H2O CO2+H2
CO2+H2
��3��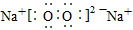 ��2Na2O2+2H2O==4Na++4OH-+O2��
��2Na2O2+2H2O==4Na++4OH-+O2��
��2��3Fe+4H2O
 Fe3O4+4H2��CO+H2O
Fe3O4+4H2��CO+H2O CO2+H2
CO2+H2��3��
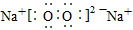 ��2Na2O2+2H2O==4Na++4OH-+O2��
��2Na2O2+2H2O==4Na++4OH-+O2�� 
��ϰ��ϵ�д�
�����Ŀ
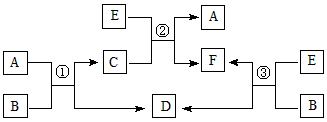
 [��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�
[��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�