��Ŀ����
9����������ѧ�ҷ������ij�ԭ�ӽṹAl13��Al14��Al13��Al14��������Ԫ�����ڱ���ijЩ����Ԫ�ص��������ƣ������೬ԭ�Ӿ���40���۵��ӣ���������Ԫ�ص�ԭ�ӣ���Χ�����ֳ�Ϊ�۵��ӣ�ʱ���ȶ������������Ͽɼ��ȶ���Al13-��Al142+������˵���д�����ǣ�������| A�� | Al13��Al14��Ϊͬ�������� | |
| B�� | Al13��ԭ����Alԭ�Ӽ���ͨ�����Ӽ���ϵ� | |
| C�� | Al14������ᷴӦ�Ļ�ѧ����ʽ�ɱ�ʾΪ��Al14+2HI=Al14I2+H2�� | |
| D�� | Al13��һ���������ܺ�þ��������Mg��Al13��2 |
���� A��ͬ��������ķ�������Ϊͬ��Ԫ���γɵIJ�ͬ�ĵ��ʣ�
B��Al13��ԭ����Alԭ�Ӽ��Թ��õ��ӶԽ�ϣ�
C��Al14��ʧȥ2�����ӱ�Ϊ�ȶ��ṹ��
D���������������Ͽɼ��ȶ���Al13-�ж��γɵĻ����
��� �⣺A�����ij�ԭ�ӽṹAl13��Al14��������Ԫ���γɵIJ�ͬ���ʣ�����ͬ�������壬��A��ȷ��
B��Al13��ԭ����ֻ��Alһ��Ԫ�أ�Alԭ�Ӽ��Թ��õ��ӶԽ�ϣ���Ϊ���ۼ���ϣ���B����
C��Al14��ʧȥ2�����ӱ�Ϊ�ȶ��ṹ����Al14������ᷴӦ�Ļ�ѧ����ʽ�ɱ�ʾΪ��Al14+2HI=Al14I2+H2������C��ȷ��
D����Ϊ���������Ͽɼ��ȶ���Al13-������Al13��һ���������ܺ�þ��������Mg��Al13��2����D��ȷ��
��ѡB��
���� ��������Ϣ��ʽ����ԭ�ӵĵ����Ų���ע����Ϣ�г�ԭ�Ӿ���40���۵���ʱ���ȶ�Ϊ���Ĺؼ���ע��֪ʶ��Ǩ��Ӧ���������飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д�
�����Ŀ
19�������������ѧ������У��Ͷ������������������ǰ�ѱ���������ǰ���廪��ѧҲ���ֹ����Ƶİ��������dz������ϵ����Ԫ��֮һ���������ڱ���λ�ڵ������ڣ�������ͬ��Ԫ�أ�Ԫ�ط�����Tl�����¶�������ʵ��ƶϲ���ȷ���ǣ�������
| A�� | �������������ɫ���� | B�� | ������+3�����ӻ����� | ||
| C�� | Tl��OH��3�������������� | D�� | Tl3+������������Al3+�� |
20�����и������ʣ�����������ʡ�������˳�����е��ǣ�������
| A�� | �ɱ��������Ȼ��� | B�� | ��ʯ�ҡ����ס����� | ||
| C�� | �ռҺ̬������� | D�� | ������������������CuSO4•5H2O�� |
17�����л�ѧ������ȷ���ǣ�������
| A�� | �����ӵĵ���ʽ��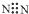 | B�� | ��ԭ�ӵĽṹʾ��ͼ�� | ||
| C�� | CO2�ĵ���ʽ��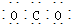 | D�� | ˮ���ӵĽṹʽ��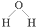 |
4�������й����ʵ����ʡ���Ӧ�������������ԭ�����;���ȷ���ҷ��������ϵ���ǣ�������
| ѡ�� | ���ʵ����ʻ�Ӧ���� | ԭ������ |
| A | SO2ͨ����ˮ����ˮ��ɫ | SO2��Ư���� |
| B | ���ԣ�NaOH��Al��OH��3 | �����ԣ�Na��Al |
| C | ���ױ�N2�����ʻ��� | �ǽ����ԣ�P��N |
| D | ����NaͶ��CuSO4��Һ�У�Na�����û���Cu | �����ԣ�Na��Cu |
| A�� | A | B�� | B | C�� | C | D�� | D |
14�� �����ᣨH3BO3����һ�ֲ�״�ṹ�İ�ɫ���壬���ڵ�H3BO3����ͨ���������������ͼ���������й�˵����ȷ���ǣ�������
�����ᣨH3BO3����һ�ֲ�״�ṹ�İ�ɫ���壬���ڵ�H3BO3����ͨ���������������ͼ���������й�˵����ȷ���ǣ�������
 �����ᣨH3BO3����һ�ֲ�״�ṹ�İ�ɫ���壬���ڵ�H3BO3����ͨ���������������ͼ���������й�˵����ȷ���ǣ�������
�����ᣨH3BO3����һ�ֲ�״�ṹ�İ�ɫ���壬���ڵ�H3BO3����ͨ���������������ͼ���������й�˵����ȷ���ǣ�������| A�� | �����ᾧ������ԭ�Ӿ��� | |
| B�� | H3BO3���ӵ��ȶ���������й� | |
| C�� | ��������ԭ�����������8�����ȶ��ṹ | |
| D�� | 1 mol H3BO3��������3 mol��� |
18��������ʵ������Ϊʵ���ж����ݵ��ǣ�������
| A�� | �ƺ�þ�ֱ�����ˮ��Ӧ���жϽ������ǿ�� | |
| B�� | ��Ϊ����HCl��H2S���ж����ȵķǽ�����ǿ�� | |
| C�� | �ȶ���CH4��SiH4���жϹ���̼�ķǽ������ǿ�� | |
| D�� | Br2��I2�ֱ���������H2��Ӧ���ж������ķǽ������ǿ�� |
8����ʯ��ʯ���Կ���CaO��Fe2O3��A12O3��SiO2��ɣ�����ʯ��ʯ��ȡ�Ȼ��ƺ���������ʵ�鲽����ͼ1��

��1����ʯ��������ܽ����Һ�����Ca2+�⣬�����еĽ���������Fe3+��Al3+��
��2������Һ���в���ʱ��������ҺpH=9��10���й��������������pH���±���NaOH���ܹ�������NaOH�������ܻᵼ��Al��OH��3�ܽ⣬Ca��OH��2������
��3����ijͬѧ��ʵ��������ͼ2��װ����ȡCO2���岢ͨ����Һͼ2���У����û�г������������ܵ�ԭ��������ӷ�����ȡ�Ķ�����̼�к������Ϊ���ܲ���������ͬѧ��ͼ��װ�ý����˸Ľ����Ľ��ķ���Ϊ�ڼ�����֮���һʢ�б���̼��������Һ��ϴ��ƿ��
������������̼����ͨ�˳���ʯ��ˮ�У�ʯ�� ˮ����ǣ������ϵ�г����ڵ���ƽ�⡢ˮ��ƽ���⣬�������ܽ�ƽ�⣬�÷���ʽ��ʾCaCO3��s��?Ca2+��aq��+CO32-��aq����

��1����ʯ��������ܽ����Һ�����Ca2+�⣬�����еĽ���������Fe3+��Al3+��
��2������Һ���в���ʱ��������ҺpH=9��10���й��������������pH���±���NaOH���ܹ�������NaOH�������ܻᵼ��Al��OH��3�ܽ⣬Ca��OH��2������
| �������� | Fe��OH��3 | Al��OH��3 | Ca��OH��2 |
| ��ʼ����pH | 1.5 | 3.3 | 12.0 |
������������̼����ͨ�˳���ʯ��ˮ�У�ʯ�� ˮ����ǣ������ϵ�г����ڵ���ƽ�⡢ˮ��ƽ���⣬�������ܽ�ƽ�⣬�÷���ʽ��ʾCaCO3��s��?Ca2+��aq��+CO32-��aq����

