��Ŀ����
19��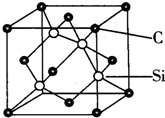 ͭ���ء��������Ԫ�صĻ�����������������̫���ܵ�ص���Ҫ���ϣ���ش�
ͭ���ء��������Ԫ�صĻ�����������������̫���ܵ�ص���Ҫ���ϣ���ش���1����̬ͭԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s1��[Ar]3d104s1����ͭԭ�Ӽ۲���ӽṹ�仯�Ƕ�����������ʱCuO��Cu2O���ȶ�����CuO��Cu2O����д����������=����������
��2���������������Ԫ���γ���̬�⻯��������γɵ��������⻯���У����ӹ��ͷֱ�ΪV�Ρ��������壬����Si-H���й��õ��Ӷ�ƫ����Ԫ�أ�����������Ӧʱ�����������������������ĵ縺����Դ�СΪSe��Si�������������������
��3��SeO2�����°�ɫ���壬�۵�Ϊ340��350�棬315��ʱ��������SeO2����ľ�������Ϊ���Ӿ��壻SeO2������Seԭ�ӵ��ӻ�����Ϊsp2��
��4����Ԫ����Ԫ�����ڱ��е�λ���ǵ������ڵ�IIIA�壬����Ԫ�ض�����ȱ�����ԣ��۵��������ڼ۲����������仯���������й¶Ե��ӵķ��ӻ���������������BF3����NH3��Ӧ����BF3•NH3��BF3•NH3�Ľṹ��ʽ�ɱ�ʾΪ
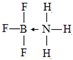 ��
����5�����ɰ��SiC���ľ����ṹ��ͼ��ʾ������SiC�У�ÿ��Cԭ����Χ���ڵ�Cԭ����ĿΪ12�����������ı߳�Ϊapm������ɰ���ܶ�Ϊ2.66��108a-3��1.6��1032a-3NA-1g/cm3��
���� ��1������Ԫ�ط��ţ��ж�Ԫ��ԭ�ӵĺ�����������ٸ��ݺ�������Ų�������д��Cu+�ĺ�����28�����ӣ����ݹ���ԭ����д���̬���Ӻ�������Ų�ʽ��ԭ�ӹ������ȫ�ա�������ȫ��ʱ���ȶ���
��2���������������Ԫ���γ���̬�⻯��ֱ�ΪH2Se��SiH4������ӽṹ�ֱ���H2O��CH4���ƣ�����Si-H���м��ϵ���ƫ����ԭ�ӣ�˵���������ۣ�����������Ӧʱ�������������������Ը��ۣ�
��3�����ݷ��Ӿ�����ۡ��е�ͣ����ݼ۲���ӶԻ�������ȷ���ӻ����ͣ�
��4����Ԫ����Ԫ�����ڱ��е�λ���ǵ������ڵڢ�A�壬�ṩ�չ����ԭ�Ӻ��ṩ�µ��ӶԵ�ԭ��֮���γ���λ����
��5��ÿ��̼ԭ������4����ԭ�ӣ�ÿ����ԭ������������3��̼ԭ�ӣ��ݴ��ж�ÿ��Cԭ����Χ�����Cԭ����Ŀ���þ�����Cԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Siԭ�Ӹ���Ϊ4�����ݦ�=$\frac{m}{V}$�������ܶȣ�
��� �⣺��1��CuԪ��Ϊ29��Ԫ�أ�ԭ�Ӻ�����29�����ӣ����Ժ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1��CuO��ͭ�ļ۲�����Ų�Ϊ3d94s0��Cu2O��ͭ�ļ۲�����Ų�Ϊ3d10��3d10Ϊ�ȶ��ṹ�������ڸ���ʱ��������Cu2O�����ȶ�����CuO��Cu2O��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��[Ar]3d104s1������
��2���������������Ԫ���γ���̬�⻯��ֱ�ΪH2Se��SiH4������ӽṹ�ֱ�V�Σ��������壻����Si-H���м��ϵ���ƫ����ԭ�ӣ�˵���������ۣ�����������Ӧʱ�������������������Ը��ۣ����������ĵ縺����Դ�СΪSe��Si��
�ʴ�Ϊ��V�Ρ��������壻����
��3��SeO2�����°�ɫ���壬�ۡ��е�ͣ�Ϊ���Ӿ��壻�������������м۲���Ӷ�=2+$\frac{1}{2}$��6-2��2��=3��Seԭ�ӵ��ӻ�����Ϊsp2���Һ���һ���µ��Ӷԣ���������V�Σ�
�ʴ�Ϊ�����Ӿ��壻sp2��
��4����Ԫ����Ԫ�����ڱ��е�λ���ǵ������ڵڢ�A�壬BF3•NH3��Bԭ�Ӻ���3���� ����1����λ����������۲��������4��Bԭ�Ӳ�ȡsp3�ӻ����û������У�Bԭ���ṩ�չ����ԭ�ӡ�Nԭ���ṩ�µ��Ӷԣ�B��Nԭ��֮���γ���λ������ṹ��ʽ�ɱ�ʾΪ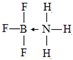 ��
��
�ʴ�Ϊ���������ڵڢ�A�壻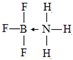 ��
��
��5��ÿ��̼ԭ������4����ԭ�ӣ�ÿ����ԭ������������3��̼ԭ�ӣ�����ÿ��Cԭ����Χ�����Cԭ����ĿΪ3��4=12���þ�����Cԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Siԭ�Ӹ���Ϊ4�������߳�=a��10-10cm�����V=��a��10-10cm��3�����ܶ�=$\frac{\frac{M}{{N}_{A}}��4}{V}$=$\frac{\frac{40}{{N}_{A}}��4}{��a��1{0}^{-10}��^{3}}$g/cm3=2.66��108a-3 g/cm3��1.6��1032a-3 NA-1g/cm3��
�ʴ�Ϊ��12��2.66��108a-3 ��1.6��1032a-3 NA-1��
���� ���⿼�����ʽṹ�����ʣ�Ϊ��Ƶ���㣬�漰�������㡢���������жϡ�ԭ���ӻ���ʽ�жϡ�ԭ�Ӻ�������Ų���֪ʶ�㣬���ؿ���ѧ���������㼰�ռ������������ѵ��Ǿ������㣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| A�� | ͭ�缫Ӧ��X������ | |
| B�� | H+�����ӽ���Ĥ���������ƶ� | |
| C�� | M�缫��ӦʽΪCO��NH2��2+H2O-6e-�TCO2��+N2��+6H+ | |
| D�� | ��N�缫����0.25 mol����ʱ�������������缫����32 g |
I����ʵ������ģ�ҵ�������̿���Ҫ�ɷ�ΪMnO2��������SiO2��Fe2O3��A12O3�ȣ��Ʊ������̵����ʣ����������ͼ��
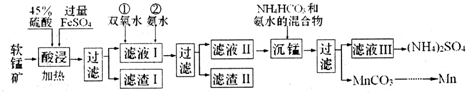
��֪��̼�����ڿ����и��¼��ȹ������ΪMn2O3���������������Ksp������ֵ�������
| ���� | Mn��OH��2 | Fe��OH��2 | Fe��OH��3 | Al��OH��3 |
| Ksp | 10-13 | 10-17 | 10-39 | 10-33 |
��2������ҺI �еμ�˫��ˮ��Ŀ���ǽ�Fe2+����ΪFe3+������ת��ΪFe��OH��3��ȥ�������ҺI ��c��Mn2+��=0.1mol•L-1��Ϊ��֤�μӰ�ˮ������Mn��OH��2��Ӧ������ҺpH���Ϊ8��
��3�������̡������е����ӷ���ʽΪMn2++HCO3-+NH3•H2O=MnCO3��+NH4++H2O��
��4����̼���̺ͽ�����Ϊԭ�Ͽ����Ʊ����̣�д���Ʊ����������һ����Ӧ�Ļ�ѧ����ʽMn2O3+2Al$\frac{\underline{\;����\;}}{\;}$Al2O3+2Mn��
��ѧ�ҷ�����NH3ȼ�ϵ�أ���KOHΪ����ʣ�ԭ����ͼ��ʾ��
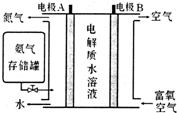
��5����ȼ�ϵ�صĸ�����ӦʽΪ2NH3+6OH--6e-=N2+6H2O��
��6���Ըõ��Ϊ��Դ����ʯī�缫�������أ�K2MnO4����Һ��ȡ������أ�ͨ��10���ӣ�����ǿ��Ϊ0.5A�������Ƶø������0.49g����֪1mol������������Ϊ96500C����
| A�� | NaHCO3��Һ�ʼ��Ե�ԭ����HCO3-��ˮ��̶ȴ��ڵ���̶� | |
| B�� | ����CaCl2�ٽ���HCO3-�ĵ��� | |
| C�� | ��Ӧ�����ӷ���ʽ��2HCO3-+Ca2+�TCaCO3��+H2CO3 | |
| D�� | ��Ӧ�����Һ�д��ڣ�c��Na+��+2c��Ca2+��=c��HCO3-��+2c��CO32-��+c��Cl-��+c��H2CO3�� |

| A�� | HRΪǿ�� | |
| B�� | a��b������ʾ��Һ��ˮ�ĵ���̶���ͬ | |
| C�� | b����Һ��c�� NH4+����c�� R-����c��OH-����c��H+�� | |
| D�� | a��c�������Һ����c��H+����c��OH-��=Kw=l.0��l0-14 |
| A�� | ���ó�ϫ�����ǽ���ѧ��ת��Ϊ���� | |
| B�� | ��������ˮ�Ĺ�����һ�������˻�ѧ�仯 | |
| C�� | �Ӻ�ˮ�п��Ի��MgCl2���������MgCl2���Ʊ�Mg | |
| D�� | �����÷�Һ�ķ�������ˮ���� |
| A�� | 1mol����1molCl2��ַ�Ӧ��ת�Ƶ�����Ϊ3NA | |
| B�� | ���³�ѹ�£�2gD216O �к������������������������������ | |
| C�� | 14g ����ʽΪCnH2n �ģ�n��2�������к��е�C=C ����Ŀһ��Ϊ$\frac{{N}_{A}}{n}$ | |
| D�� | �ں�Al3+����ΪNA��AlCl3��Һ�У�Cl-����Ϊ3NA |
| A�� | ����Һ�У�H+��Mg2+��NO3-��Cl-���Դ������� | |
| B�� | ����Һ��ʹ���۵⻯����ֽ��������Ӧ�����ӷ���ʽΪ��Fe3++2I-�TFe2++I2 | |
| C�� | ����Һ������Ba��OH��2��Һ��Ӧ�����ӷ���ʽΪ��Fe3++SO42-+Ba2++3OH-�TFe��OH��3��+BaSO4�� | |
| D�� | ����Һ������ͭ�۷�Ӧ�����ӷ���ʽΪ��3Cu+2Fe3+�T2Fe+3Cu2+ |
 ����Ԫ�صķ�����S��
����Ԫ�صķ�����S��