��Ŀ����
7��ijͬѧ��ʵ����̽��NaHCO3�����ʣ������£�����0.10mol/LNaHCO3��Һ������pHΪ8.4��ȡ��������Һ�μ�CaCl2��Һ��pH=7���μӹ����в�����ɫ��������������ų�������˵������ȷ���ǣ�������| A�� | NaHCO3��Һ�ʼ��Ե�ԭ����HCO3-��ˮ��̶ȴ��ڵ���̶� | |
| B�� | ����CaCl2�ٽ���HCO3-�ĵ��� | |
| C�� | ��Ӧ�����ӷ���ʽ��2HCO3-+Ca2+�TCaCO3��+H2CO3 | |
| D�� | ��Ӧ�����Һ�д��ڣ�c��Na+��+2c��Ca2+��=c��HCO3-��+2c��CO32-��+c��Cl-��+c��H2CO3�� |
���� A��NaHCO3���ܵ�������ˮ�⣻
B������CaCl2����CO32-��
C��HCO3-��Ca2+����CaCO3�������ӣ������ӽ��HCO3-����H2CO3��
D�����ݵ���غ������
��� �⣺A��NaHCO3���ܵ�������ˮ�⣬ˮ���Լ��ԣ����������ԣ�NaHCO3��Һ�ʼ��Ե�ԭ����HCO3-��ˮ��̶ȴ��ڵ���̶ȣ���A��ȷ��
B����Һ�д���HCO3-?CO32-+H+������CaCl2����CO32-���ٽ�HCO3-�ĵ��룬��B��ȷ��
C��HCO3-��Ca2+����CaCO3�������ӣ������ӽ��HCO3-����H2CO3����Ӧ�����ӷ���ʽ��2HCO3-+Ca2+�TCaCO3��+H2CO3����C��ȷ��
D����Һ�д��ڵ���غ㣬c��Na+��+c��H+��+2c��Ca2+��=c��HCO3-��+2c��CO32-��+c��Cl-��+c��OH-������Һ��pH=7��c��H+��=c��OH-����c��Na+��+2c��Ca2+��=c��HCO3-��+2c��CO32-��+c��Cl-������D����
��ѡD��
���� ���⿼���˵������Һ�з�Ӧʵ�ʣ������ܽ�ƽ�������Ӧ�ã��������Һ������Ũ�ȴ�С������غ㡢�����غ��֪ʶ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
17���������ġ�ǧ�����������࣬������ɰʼ���𡱣�����ʫ�䷴ӳ�˶��Խ���̵���д������˵������ȷ���ǣ�������
| A�� | ��ǧ���������Ļ���ԭ���ǻ�ѧ�е���ȡ | |
| B�� | 24K�𣨴��𣩵ĸ������������ܶ���18K����Խ | |
| C�� | ��ͭ����ƽ𣬽�Ʋ������ˣ���ӿ�ͭ���ĸ�ʴ���� | |
| D�� | �����£����ױ����������������κ����ʷ�����ѧ��Ӧ |
18�����ʵ���У���Ӧ������ͽ��۶���ȷ�������߾��������ϵ���ǣ�������
| ѡ�� | ʵ�� | ���� | ���� |
| A | ������̼��Ʒ�ĩ���뵽����NH4Cl��Һ�� | �������壬��ĩ�ܽ� | NH4Clˮ��ʹ��Һ������ |
| B | ��BaSO4�����ĩ���뱥��Na2CO3��Һ�У����裬���ˣ�ϴ�ӣ��������м���ϡ���� | �������壬���������ܽ� | Ksp��BaCO3����Ksp��BaSO4�� |
| C | ����ɫ����ͭ��ĩ���и��¼��� | ��ɫ��ɺ�ɫ | CuO�ֽ�����ͭ���� |
| D | ��ij��Һ�μӹ�����ϡ���� | �д̼�����ζ�����������Һ�г��ֳ��� | ��Һ��һ������S2-��SO32- |
| A�� | A | B�� | B | C�� | C | D�� | D |
15���±��С��Լ����롰����������Ӧ��ϵ������ǣ�������
| ѡ�� | �Լ� | ���������� |
| A | AgNO3��Һ | �μӰ�ˮ�����������������μӰ�ˮ�������ܽ� |
| B | ���� | �μ�NaOH��Һ���������壻�����μӣ�������ɫ���� |
| C | CuSO4��Һ | �ӹ���NaOH��Һ��������ɫ�������ټ���ȩ�����ȣ�������ɫ���� |
| D | ��ˮ | �μ�NaOH��Һ����Һ��ɫ��ȥ���ټ�HCl����Һ��ɫ�ָ� |
| A�� | A | B�� | B | C�� | C | D�� | D |
12��ij����Һ����Ҫ�ɷ�ΪNaClO�������йظû����Һ��������ȷ���ǣ�������
| A�� | ����Һ�У�H+��NH4+��SO42-��Br-���Դ������� | |
| B�� | ����Һ�У�Ag+��K+��NO3-��CH3CHO���Դ������� | |
| C�� | �����Һ�м���Ũ���ᣬÿ����1molCl2��ת�Ƶ���ԼΪ6.02��1023�� | |
| D�� | �����Һ�е�������FeSO4��Һ����Ӧ�����ӷ���ʽΪ��2Fe2++ClO-+2H+�TCl-+2Fe3++H2O |
16���й���ͼ�ĸ����õ绯ѧװ�õ���������ȷ���ǣ�������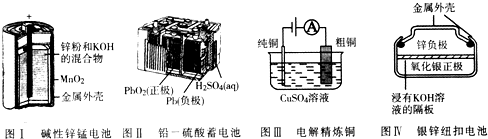

| A�� | ͼI��ʾ����У�п���缫��ӦΪZn-2e-�TZn2+ | |
| B�� | ͼ����ʾ��طŵ�����У�ÿת��1mo1���ӣ�����1mol PbSO4 | |
| C�� | ͼIII��ʾװ�ù��������У��������Һ��Cu2+Ũ��ʼ�ղ��� | |
| D�� | ͼ����ʾ����У�Ag2O�ڵ�ع��������б�����ΪAg |
14���������в������������ý�����ȷ���ǣ�������
| ��� | ���������� | ���� |
| A | ����ˮ�м��������������ú�ˮ��Ϊ��ɫ | ����Br2�����˼ӳɷ�Ӧ |
| B | ��ij��ɫ��Һ�еμ������ữ��BaCl2��Һ��������ɫ���� | ԭ��Һ��һ������SO42- |
| C | ��25mL��ˮ�ͷ�ˮ�зֱ����5��FeCl3������Һ��ǰ��Ϊ��ɫ������Ϊ���ɫ | �¶����ߣ�Fe3+��ˮ��̶����� |
| D | ��1mL KSCN��Һ��1mL ͬŨ��FeCl3��Һ��ֻ�ϣ��ټ�������KSCN��Һ����Һ��ɫ���� | ֤����Һ�д���ƽ�⣺Fe3++3SCN?Fe��SCN��3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
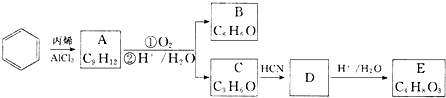
 ��A�ĺ��б�����ͬ���칹�壨��A�⣩��7�֣�
��A�ĺ��б�����ͬ���칹�壨��A�⣩��7�֣�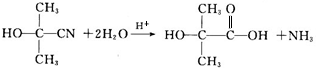 ��
��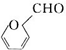 ����һ����Ҫ�Ļ���ԭ�ϣ���ͬ��ȩ�뱽�ӷ�Ӧ���ɷ�ȩ��֬����Ҳ���뱽�ӷ�Ӧ���ɿ�ȩ��֬��д���÷�Ӧ�Ļ�ѧ����ʽ��
����һ����Ҫ�Ļ���ԭ�ϣ���ͬ��ȩ�뱽�ӷ�Ӧ���ɷ�ȩ��֬����Ҳ���뱽�ӷ�Ӧ���ɿ�ȩ��֬��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
�� ��G
��G ��
��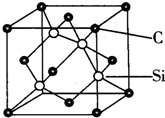 ͭ���ء��������Ԫ�صĻ�����������������̫���ܵ�ص���Ҫ���ϣ���ش�
ͭ���ء��������Ԫ�صĻ�����������������̫���ܵ�ص���Ҫ���ϣ���ش�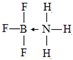 ��
��