��Ŀ����
A��B��C��D��E��FΪ��ѧ��ѧ�еij������ʣ�������A��1~2�ֶ�����Ԫ����ɣ���һ��������������ת����ϵ���������������
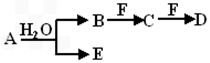
��1����������AΪ��ɫ����
����F��һ�ֽ������ʣ���д��B������F��Ӧ����C������E�����ӷ���ʽ��______________________________��
����CΪֱ���ͷ��ӣ�E����Ư���ԣ�����F��ɫ��Ӧ�ʻ�ɫ����д����ҵ���Ʊ�����F�Ļ�ѧ��Ӧ����ʽ��___________________��_________________��
��2����AΪ����ɫ���壬��A���ʵĵ���ʽΪ��_________________��
��������B��D��ʽ����ȣ�����C�Ĵ����ŷ�������������صĻ������⼴__________��
��������A��D��ʽ�����,�������ӷ���ʽ��ʾ����F��ˮ��Һ�����Ե�ԭ��__________��
��3��������A��һ��Ԫ��ԭ�ӵ�����������Ϊ�ڲ����������1/5����B��D�ֱ�����ˮ��������Һ��ǡ��������ϣ��ɵ�һ��������Ԫ�ص�����Һ����д��A�Ļ�ѧʽ________��
����F��һ�ֽ������ʣ���д��B������F��Ӧ����C������E�����ӷ���ʽ��______________________________��
����CΪֱ���ͷ��ӣ�E����Ư���ԣ�����F��ɫ��Ӧ�ʻ�ɫ����д����ҵ���Ʊ�����F�Ļ�ѧ��Ӧ����ʽ��___________________��_________________��
��2����AΪ����ɫ���壬��A���ʵĵ���ʽΪ��_________________��
��������B��D��ʽ����ȣ�����C�Ĵ����ŷ�������������صĻ������⼴__________��
��������A��D��ʽ�����,�������ӷ���ʽ��ʾ����F��ˮ��Һ�����Ե�ԭ��__________��
��3��������A��һ��Ԫ��ԭ�ӵ�����������Ϊ�ڲ����������1/5����B��D�ֱ�����ˮ��������Һ��ǡ��������ϣ��ɵ�һ��������Ԫ�ص�����Һ����д��A�Ļ�ѧʽ________��
��1����Fe+4H++NO3-=Fe3++NO��+2H2O����NH3+CO2+H2O+NaCl=NaHCO3��+NH4Cl��2NaHCO3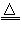 Na2CO3+H2O+CO2��
Na2CO3+H2O+CO2��
��2��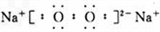 �����γ����ꣻ��Al3++3H2O
�����γ����ꣻ��Al3++3H2O Al(OH)3+3H+
Al(OH)3+3H+
��3��Mg3N2
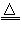 Na2CO3+H2O+CO2��
Na2CO3+H2O+CO2����2��
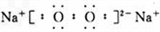 �����γ����ꣻ��Al3++3H2O
�����γ����ꣻ��Al3++3H2O Al(OH)3+3H+
Al(OH)3+3H+��3��Mg3N2

��ϰ��ϵ�д�
�����Ŀ
 [��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�
[��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�