��Ŀ����
1�� ��ͼ��ʾ��U�ܵ���˱�ˮ�ͽ�����ռ���������������Ϊ1��4���Ļ�����壬�ٶ�������ˮ���ܽ�ȿ��Ժ��ԣ�������м���������Ļ�������װ�÷������й����ĵط����û�����建���ķ�Ӧһ��ʱ�䣮
��ͼ��ʾ��U�ܵ���˱�ˮ�ͽ�����ռ���������������Ϊ1��4���Ļ�����壬�ٶ�������ˮ���ܽ�ȿ��Ժ��ԣ�������м���������Ļ�������װ�÷������й����ĵط����û�����建���ķ�Ӧһ��ʱ�䣮��1������������ȷ�Ӧ��֣���ֻ����һ���л����д����ѧ����ʽ��CH4+4Cl2$\stackrel{����}{��}$CCl4+4HCl��
��2����������Сʱ�ķ�Ӧ��U���Ҷ˵IJ�������ˮ���仯��B��
A������ B������ C������ D����ȷ��
��3������ͨ��������ȼ�ϣ���ȼ�շ�Ӧ�Ļ�ѧ����ʽ��CH4+2O2$\stackrel{��ȼ}{��}$2H2O+CO2��
��4�����ü���-��������ȼ�ϵ�أ��������ҺΪKOH��Һ����д���õ�ص������ĵ缫��Ӧʽ2O2+4H2O+8e-�T8OH-�������ĵ缫��ӦʽCH4-8e-+10OH-�TCO32-+7H2O��
���� ��1��������������Ӧ��֣������е�H��ȫ��ȡ����ֻ�������Ȼ�̼��
��2�������������Ӧ��������ѹǿ��С���Ҷ˵IJ�������ˮ�����ͣ�
��3������ȼ�պ�����Ƕ�����̼��ˮ���ݴ���д����ʽ��
��4������ȼ�ϵ���У�������Ͷ��ȼ�ϼ��飬����ʧ���ӷ���������Ӧ��������Ͷ�������������õ��ӵĻ�ԭ��Ӧ���ܷ�Ӧ��ȼ�Ϻ�������Ӧ�Ļ�ѧ����ʽ����Ϊ������Ӧ������Ӧ�ĺͣ�ע�����ʻ�����������ʽ����д��
��� �⣺��1��������������Ӧ��֣�ֻ�������Ȼ�̼��Ϊȡ����Ӧ����Ӧ�Ļ�ѧ����ʽΪCH4+4Cl2$\stackrel{����}{��}$CCl4+4HCl���ʴ�Ϊ��CH4+4Cl2$\stackrel{����}{��}$CCl4+4HCl��
��2�������������Ӧ���ɵĶ��ȼ��顢���ȼ��顢���Ȼ�̼������״��Һ�壬ֻ��һ�ȼ�����Ȼ��������壬�Ȼ��⼫������ˮ��ʹ������ѹǿ��С���Ҷ˵IJ�������ˮ�����ͣ�
�ʴ�Ϊ��B��
��3������ȼ�պ�����Ƕ�����̼��ˮ����Ӧ�ķ���ʽΪCH4+2O2$\stackrel{��ȼ}{��}$2H2O+CO2��
�ʴ�Ϊ��CH4+2O2$\stackrel{��ȼ}{��}$2H2O+CO2��
��4���ڼ�����Һ�У�������Ͷ��ȼ�ϼ��飬����ʧ���ӷ���������Ӧ��CH4-8e-+10OH-�TCO32-+7H2O��������Ͷ�������������õ��ӵĻ�ԭ��Ӧ��2O2+4H2O+8e-�T8OH-��
�ʴ�Ϊ��2O2+4H2O+8e-�T8OH-��CH4-8e-+10OH-�TCO32-+7H2O��
���� ���⿼��ʵ��װ���ۺϣ�Ϊ��Ƶ���㣬���ռ����ȡ����Ӧ��ȡ�����������Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬��Ŀ�ѶȲ���ע����������¼���ʧ���Ӳ������������ӣ�Ҫ����̼������ӣ�Ϊ�״��㣮

| A�� | ��FeCl3��Һ�е��뼸��KSCN��Һ����Һ���Ѫ��ɫ | |
| B�� | ����ɫ�廯������ֽ���ɰ�ɫ | |
| C�� | ��Ѫ��ɫ��Fe��SCN��3��Һ�м�������KI���壬��Һ��ɫ��dz | |
| D�� | ��50mL��Ͳ��ȡ30mL����ɫ��NO2���岢��סע��ף���������ѹ������ѹ����Ͳ�е����壨�˹��̲������¶ȱ仯����������ɫ���� |
| A�� | �����������ᷴӦ���������Ƴɺ�ɫͿ�ϻ����� | |
| B�� | �̻���Ѥ���ͷ��Ǻ��ء��ơ��ơ�ͭ�Ƚ���Ԫ�صĻ�����Ļ�ѧ���ʵ�չ�� | |
| C�� | ҽ�þƾ����õ�����ֲ�ᆳ�ǻ��ٷ��;������Ƴɣ�Ũ��ͨ����75% | |
| D�� | ά����C��ˮ����ά���أ�����ǿ����ֿ������п��������� |
| A�� | 2H2��g��+O2��g��=2H2O��l������H=-484kJ/mol | B�� | H2O��g��=H2��g��+$\frac{1}{2}$O2��g������H=+242kJ/mol | ||
| C�� | 2H2��g��+O2��g��=2H2O��g������H=-484kJ/mol | D�� | H2��g��+$\frac{1}{2}$O2��g��=H2O��g������H=+242kJ/mol |
| A�� | ��������ѹǿ���ٱ仯 | |
| B�� | �����������������ٱ仯 | |
| C�� | ÿ����2mol SO3��ͬʱ������1mol SO2 | |
| D�� | ÿ����2mol SO2��ͬʱ������2mol SO3 |
| A�� | ���ͬλ��������ԭ�ӵķ��ţ�13H | |
| B�� | Mg2+�Ľṹʾ��ͼ�� | |
| C�� | �����ĵ���ʽ�� | |
| D�� | Ca��ClO��2�ĵ��뷽��ʽ��Ca��ClO��2=Ca2++2ClO- |
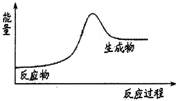
| A�� | H2��Cl2��ȼ�� | B�� | ʯ��ʯ�ֽ� | ||
| C�� | Na��H2O�ķ�Ӧ | D�� | Ba��OH��2•8H2O��NH4Cl����ķ�Ӧ |
| A�� | CH2=CH-CH3+Cl2$\stackrel{����}{��}$CH2=CHCH2Cl+HCl �ӳɷ�Ӧ | |
| B�� | 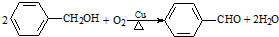 ������Ӧ ������Ӧ | |
| C�� |  �ӳɷ�Ӧ �ӳɷ�Ӧ | |
| D�� | 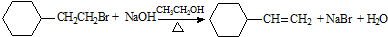 ȡ����Ӧ ȡ����Ӧ |
 ��
��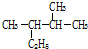 ��ϵͳ����������Ϊ2��3-�������飻
��ϵͳ����������Ϊ2��3-�������飻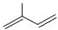 ��
��