��Ŀ����
11��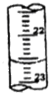 ijѧ����0.1000mol/L�������Һ�ⶨij�ռ���Ʒ�Ĵ��ȣ����ʲ������ᷴӦ����ʵ�鲽�����£�
ijѧ����0.1000mol/L�������Һ�ⶨij�ռ���Ʒ�Ĵ��ȣ����ʲ������ᷴӦ����ʵ�鲽�����£���1�����ƴ���Һ����2.50g�����������ʵĹ����ռ���Ʒ����500mL��Һ�����õIJ������������ձ�����ͷ�ιܡ��������⣬����Ҫ500mL����ƿ��
��2�����
��������ˮϴ����ʽ�ζ��ܣ�������ע���������Һ����0���̶������ϣ�
�̶��õζ��ܲ�ʹ�ζ��ܼ������Һ�壬����Һ������0���̶������£�����¼������
����ȡ20.00mL����Һע��ྻ����ƿ�У�������2�η�̪��Һ��
�����ñ�Һ�ζ����յ㣬��¼�ζ���Һ�������
�������ζ��������д�����ǣ����ţ��ô�������ᵼ�²ⶨ���ƫ���ƫ����ƫС������Ӱ�족����
����ȥ��ʽ�ζ��������ݵķ���Ӧ������ͼ�ı�������
�۲�����еζ�ʱ�۾�Ӧ�۲���ƿ����Һ��ɫ�ı仯���жϵζ��յ�������ǵ��μ����һ�α�Һʱ������Һ�ɺ�ɫ�պñ�Ϊ��ɫ���Ұ�����ڲ���ɫ��
��������������Ӷ�����������и��Ӷ��������ʹ�ⶨ���ƫС���ƫ����ƫС������Ӱ�족��
��3������ȷ��������й����ݼ�¼���£�����ȷ��������й����ݼ�¼�����
| �ζ����� | ����Һ��� | �����������mL�� | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 20.00 | 0.20 | 20.38 |
| �ڶ��� | 20.00 | 4.00 | 24.20 |
| ������ | 20.00 | 2.38 | a |
���� ��1����������һ�����ʵ���Ũ�ȵ���Һ�����Ʋ���ѡ��ʹ�õ�������
��2����ʢ��Һ����ʽ�ζ��ܱ�����ϴ��������ȡ����ҺŨ��ƫ�ͣ�
�ڸ��ݼ�ʽ�ζ����������ݵķ�����
�۵ζ�ʱ�۾�ע����ƿ����ɫ�仯�����ݴ���Һ�м����̪����ҺΪ��ɫ���кͷ�Ӧ��������Һ��ɫ��ʧ�����жϣ�
�ܵζ�ǰ���ӣ����¶���ƫ�ζ����ӻᵼ�¶���ƫС��
��3�����ݵζ��ܵĽṹ�;�ȷ�ȣ��ȷ������ݵ���Ч�ԣ����ݵζ��ܶ�������������Һ���������������������ƽ�������Ȼ����ݹ�ϵʽNaOH��HCl����������Ƶ����ʵ������ټ����ռ���Ʒ�Ĵ��ȣ�
��� �⣺��1����2.50g�����������ʵĹ����ռ���Ʒ����500mL��Һ�����ƹ���Ϊ�����㡢�������ܽ⡢��ȴ��ת�ơ�ϴ�ӡ�ҡ�ȵȣ���Ҫ��������������ƽ���ձ�����������500mL����ƿ����ͷ�ιܣ����õIJ������������ձ�����ͷ�ιܡ��������⣬����Ҫ500mL����ƿ��
�ʴ�Ϊ��500mL����ƿ��
��2����������ˮϴ����ʽ�ζ��ܣ�Ȼ��������ʽ�ζ����ñ�Һ������ϴ������ᵼ�±�ҺŨ�ȼ�С�����ĵı�Һ���ƫ�ⶨ���ƫ��
�ʴ�Ϊ����ƫ��
�ڼ�ʽ�ζ����������ݵķ������ѵζ��ܵĽ�ͷ�����������������������ݣ�
��ѡ������
�۵ζ�ʱ˫��Ӧע��۲���ƿ����Һ��ɫ�ı仯����ƿ�е����̪����ҺΪ��ɫ�����ŷ�Ӧ���У�����������������ǡ�÷�Ӧ����Һ����ɫ����ʧ�����Դﵽ�յ�����Ϊ�����μ����һ�α�Һʱ������Һ�ɺ�ɫ�պñ�Ϊ��ɫ���Ұ�����ڲ���ɫ��
�ʴ�Ϊ���۲���ƿ����Һ��ɫ�ı仯�����μ����һ�α�Һʱ������Һ�ɺ�ɫ�պñ�Ϊ��ɫ���Ұ�����ڲ���ɫ��
�ܲ���II�����Ӷ������ᵼ�µζ��ܵĶ���ƫ������и��Ӷ������ᵼ�µζ��ܶ���ƫС�����յ������ĵ��������ƫС���ⶨ���ƫ�ͣ�
�ʴ�Ϊ��ƫС��
��3���ٵζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL��
�ʴ�Ϊ��22.60��
������������������ֱ�Ϊ��20.38mL-0.20mL=20.18mL��24.20mL-4.00mL=20.20mL��a=22.6mL��22.6mL-2.38mL=20.22mL�����εζ����ݶ�����Ч�ģ��������������ƽ�����Ϊ��$\frac{20.18ml+20.20ml+20.22ml}{3}$=20.20mL��
���ݹ�ϵʽNaOH��HCl��֪��n��NaOH��=n��HCl��=0.1000mol•L-1��0.022mL=0.00202mol��
����20.00mL������Һ���У�m���ռ�Tn•M�T0.00202mol��40g/mol=0.808g��
����1000mL������Һ����m���ռ�T0.808g��$\frac{500ml}{20ml}$=2.02g��
�ռ�Ĵ��Ȧأ��ռ=$\frac{2.02g}{2.5g}$��100%=80.8%��
�ʴ�Ϊ��80.8��
���� ���⿼������к͵ζ�ʵ�飬��Ŀ�Ѷ��еȣ�ע�����ʵ���ԭ�������衢�����Լ�ע���������ʵ�����������

 ij�о�С���ñ�NaOH��Һ�ζ��״ף��ⶨʳ�ð״��д���ĺ���������˵����ȷ���ǣ�������
ij�о�С���ñ�NaOH��Һ�ζ��״ף��ⶨʳ�ð״��д���ĺ���������˵����ȷ���ǣ�������| A�� | ����ͼ����ȡһ������Ĵ���״�����ƿ�� | |
| B�� | �۲��ʽ�ζ��ܶ���ʱ�����ζ�ǰ���ӣ��ζ����ӣ������ᵼ��ʳ�ð״��д���ĺ���ƫС | |
| C�� | ��ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ�������ᵼ��ʳ�ð״��д���ĺ���ƫ�� | |
| D�� | �ζ�ʱ�۾�Ҫע���ŵζ�����NaOH��Һ��Һ��仯����ֹ�ζ����� |
| A�� | ������ˮ��ʹ����ʯ����ֽ�ȱ�죬����ɫ | |
| B�� | ������ˮ��ֻ����Cl2��H2O���� | |
| C�� | ��ˮ���������������ǿ | |
| D�� | ������ˮ�������ݳ�����������Cl2 |