��Ŀ����
6��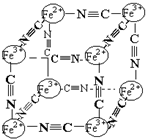 ������ѧ�г���X�����о�����ṹ����һ����ɫ����ɱ�ʾΪ��MxFey��CN��z���о��������Ľṹ������Fe2+��Fe3+�ֱ�ռ��������Ķ��㣬�����������ڣ���CNһλ������������ϣ��侧���е������ӽṹ��ͼʾ������˵����ȷ���ǣ�������
������ѧ�г���X�����о�����ṹ����һ����ɫ����ɱ�ʾΪ��MxFey��CN��z���о��������Ľṹ������Fe2+��Fe3+�ֱ�ռ��������Ķ��㣬�����������ڣ���CNһλ������������ϣ��侧���е������ӽṹ��ͼʾ������˵����ȷ���ǣ�������| A�� | �þ�����ԭ�Ӿ��� | |
| B�� | M������λ����������������ģ���+2�� | |
| C�� | M������λ����������������ģ���+1�ۣ���M+��ȱ�ʣ�������û��M+��ռ�����ĵİٷֱȣ�Ϊ50% | |
| D�� | ����Ļ�ѧʽ�ɱ�ʾΪMFe2��CN��3����MΪ+1�� |
���� A���þ����к���Fe2+��Fe3+��CN-���������ӣ��ݴ��жϾ������ͣ�
B�����ݾ����ṹ�����þ�̯��ȷ�������и����Ӹ������ٸ��ݻ��ϼ۴�����Ϊ����жϣ�
C�����ݾ����ṹ�����þ�̯��ȷ�������и����Ӹ����ȿ��жϣ�
D�����ݾ����ṹ�����þ�̯��ȷ�������и����Ӹ������ٸ��ݻ��ϼ۴�����Ϊ����жϣ�
��� A���þ����к���Fe2+��Fe3+��CN-���������ӣ����Ըþ���Ϊ���Ӿ��壬��A����
B�����ݾ����ṹ��֪�������к���Fe2+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��Fe3+�ĸ���Ϊ4��$\frac{1}{8}$=$\frac{1}{2}$��CN-�ĸ���Ϊ12��$\frac{1}{4}$=3������Fe2+��Fe3+��CN-�ĸ�����Ϊ1��1��6�����ݻ��ϼ۴�����Ϊ���֪��M�Ļ��ϼ�Ϊ+1�ۣ���B����
C�����ݾ����ṹ����B�ļ����֪��ÿ�������к���Fe2+0.5����Fe3+0.5����CN-3������B��֪M�Ļ��ϼ�Ϊ+1�ۣ����ݻ��ϼ۴���Ϊ�㣬��֪ÿ������ƽ������MҲ��0.5������M������λ������������������ϣ���������������һ����M+������һ������M+������M+��ȱ��Ϊ50%����C��ȷ��
D����B�ķ�����֪������Ļ�ѧʽ�ɱ�ʾΪMFe2��CN��6����MΪ+1�ۣ���D����
��ѡC��
���� ������Ҫ�����̯�����㻯ѧʽ������ṹ��ȷ����֪ʶ�㣬�е��Ѷȣ���һ�����ۺ��ԣ�

| A�� | v��NH3��=0.010mol/��L•s�� | B�� | v��O2��=0.10mo1/��L•s�� | ||
| C�� | v��NO��=0.10mo1/��L•s�� | D�� | v��H2O��=0.30mol/��L•s�� |
| A�� | 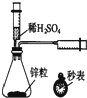 �ⶨһ��ʱ��������H2�ķ�Ӧ���� | B�� |  ��ȡ�����еĵ� | ||
| C�� |  ������ȼ�ղ�����SO2 | D�� | 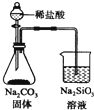 ֤���ǽ����ԣ�Cl��C��Si |
| A�� | ��ͨ��ʳ���к�3%��5%������������������ | |
| B�� | ������һ�����ᣬ�����Ա�̼����� | |
| C�� | �����ô׳�ȥ�տ�ˮ�ĺ��ڱڵ�ˮ�� | |
| D�� | ��һ�������£����������Ҵ�����������Ӧ |
| A�� | ʯӢ�ɵ��磬�����������ά | |
| B�� | �����ĵ��ʶ�������������� | |
| C�� | ���ξ����ܽ⡢���ˡ������ᾧ�����ɵõ��������Ȼ��� | |
| D�� | ������ʳ�õ�ʳ�ס�ֲ���͡����ﵰ�����ʶ��ǻ���� |
| �� ���� | I A | ��A | ��A | ��A | ��A | ��A | ��A | O |
| 1 | A | |||||||
| 2 | D | E | G | I | ||||
| 3 | B | C | F | H |
��2��A�ֱ���D��E��F��G��H�γɵĻ������У����ȶ���������HF
��3����B��C��E��F��G��H�У�ԭ�Ӱ뾶������Mg
��4��A��E��ɵ��������Ľṹʽ

��5���õ���ʽ��ʾB��H��ɻ�������γɹ���

��6��H������������ˮ�����C��������������Ӧ�����ӷ���ʽ6H++Al2O3=2Al3++3H2O��
| A�� | C6H5OH | B�� | H2O | C�� | HNO3 | D�� | C2H5OH |
 A��B��C��D���ɶ�����Ԫ���γɵĵ��ʣ�B��E��Ϊ��ɿ����ijɷ֣�������F����ɫ��Ӧ�ʻ�ɫ����G�У��ǽ���Ԫ�������Ԫ�ص�ԭ�Ӹ�����Ϊ1��2����һ�������£�������֮����ת����ϵ��ͼ��ͼ�в��ֲ���δ�г�����
A��B��C��D���ɶ�����Ԫ���γɵĵ��ʣ�B��E��Ϊ��ɿ����ijɷ֣�������F����ɫ��Ӧ�ʻ�ɫ����G�У��ǽ���Ԫ�������Ԫ�ص�ԭ�Ӹ�����Ϊ1��2����һ�������£�������֮����ת����ϵ��ͼ��ͼ�в��ֲ���δ�г����� ��
��