��Ŀ����
8��һ�ֺ�����ﮡ��ܵ����͵��Ӳ��ϣ������в����ķ��������ɹۣ������е����Խ�����������ʽ���ڣ�����Co2O3•CoO����ʽ���ڣ������������ĵ����˫�棻﮻��������У��ӷ����л��������ܣ�CoO���Ĺ����������£�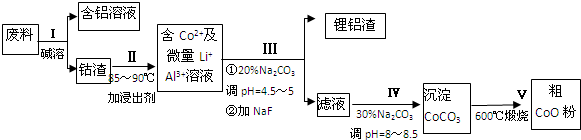
��1��Ϊ����߷����ܽ���Ч�ʣ����Բ�ȡ�Ĵ�ʩ�н��衢�ʵ�����¶ȡ����顢�ʵ������ҺŨ�ȵȣ�дһ�����ɣ�
��2�����̢��в���NaOH��Һ�ܳ������е�Al����Ӧ�����ӷ���ʽΪ2Al+2OH-+2H2O=2AlO2-+3H2����
��3�����̢��м���ϡH2SO4�ữ���ټ���Na2S2O3��Һ�����ܣ�������ܵĻ�ѧ��Ӧ����ʽΪ��������ֻ��һ�������4Co2O3•CoO+Na2S2O3+11H2SO4=12CoSO4+Na2SO4+11H2O����ʵ����ģ�ҵ����ʱ��Ҳ������������ܣ���ʵ�ʹ�ҵ�����в������ᣬ��ӷ�Ӧԭ������������������ܵ���Ҫԭ��Co2O3•CoO�������������Cl2����Ⱦ������
��4�����̢�õ����������Ҫ�ɷ���LiF��Al��OH��3����д������Al��OH��3��Ӧ�����ӷ���ʽ2Al3++3CO32-+3H2O=2Al��OH��3��+3CO2����
��5��̼������Һ�ڹ��̢��IV����������������ͬ����д���ڹ��̢�����������ǵ���pH���ṩCO32-��ʹCo2+����ΪCoCO3��
��6��ʵ����ģ����������V�У�����ʹ�õ����������ƾ��ơ����ż��⣬���������ǡ�������
���� �����������������ܽ�������������Һ�����ʣ������ӷ����з�����������˺�õ���Co2O3•CoO���������������ܽ���˺�õ���Co3+��Al3+��Li+����Һ��ͨ��������Һ��pH�õ�Al��OH��3��LiF�������ٹ��˵õ���Co3+����Һ���ٵμ�Na2CO3��Һ�õ�CoCO3�����������ˡ�ϴ�ӳ��������CoCO3���м��ȷֽ�ɵ�CoO��ĩ��
��1�������¶ȡ����衢��С����Ŀ����Ⱦ��ܼ��ٹ�����ܽ⣻
��2����������������Һ��Ӧ����ƫ�����ƺ�������
��3��Co3O4��Na2S2O3�����������·���������ԭ��Ӧ����CoSO4��Na2SO4��H2O��������л�ԭ�ԣ��ܱ�Co2O3•CoO���������ж�����������Ⱦ������
��4������������̼������ӷ���˫ˮ���������������Ͷ�����̼��
��5��̼������Һ�ڹ��̢�������������̼������ӷ���˫ˮ���������������Ͷ�����̼��̼������Һ�ڹ��̢��е���pH���ṩCO32-��ʹCo2+����ΪCoCO3��
��6������VΪ���ȹ��壬���������оƾ��ơ����żܡ������ǡ������ȣ�
��� �⣺�����������������ܽ�������������Һ�����ʣ������ӷ����з�����������˺�õ���Co2O3•CoO���������������ܽ���˺�õ���Co3+��Al3+��Li+����Һ��ͨ��������Һ��pH�õ�Al��OH��3��LiF�������ٹ��˵õ���Co3+����Һ���ٵμ�Na2CO3��Һ�õ�CoCO3�����������ˡ�ϴ�ӳ��������CoCO3���м��ȷֽ�ɵ�CoO��ĩ��
��1�������¶ȡ����衢��С����Ŀ����Ⱦ��ܼ��ٹ�����ܽ⣬����Ϊ������ܽ�������ԭ�ϵĽ���Ч�ʣ����Բ�ȡ�Ĵ�ʩ�У��ʵ������¶ȣ����裬��ĥ��ʯ���ʵ����ϡ�����Ũ�ȣ��ʴ�Ϊ�����衢�ʵ�����¶ȡ����顢�ʵ������ҺŨ�ȵȣ�
��2����������������Һ��Ӧ����ƫ�����ƺ���������Ӧ�����ӷ�Ӧ����ʽΪ��2Al+2OH-+2H2O=+2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O=2AlO2-+3H2����
��3��Co3O4��Na2S2O3�����������·���������ԭ��Ӧ����CoSO4��Na2SO4��H2O����Ӧ����ʽΪ��4Co3O4+Na2S2O3+11H2SO4=12CoSO4+Na2SO4+11H2O�����Լ���Na2S2O3�������ǻ�ԭCo3+��������л�ԭ�ԣ��ܱ�Co2O3•CoO���������ж�����������Ⱦ���������Բ������ᣬ
�ʴ�Ϊ��4Co2O3•CoO+Na2S2O3+11H2SO4=12CoSO4+Na2SO4+11H2O��Co2O3•CoO�������������Cl2����Ⱦ������
��4������������̼������ӷ���˫ˮ���������������Ͷ�����̼��ˮ������ӷ���ʽΪ��2Al3++3CO32-+3H2O=2Al��OH��3��+3CO2����
�ʴ�Ϊ��2Al3++3CO32-+3H2O=2Al��OH��3��+3CO2����
��5��̼������Һ�ڹ��̢�������������̼������ӷ���˫ˮ���������������Ͷ�����̼��̼������Һ�ڹ��̢��е���pH���ṩCO32-��ʹCo2+����ΪCoCO3��
�ʴ�Ϊ������pH���ṩCO32-��ʹCo2+����ΪCoCO3��
��6������VΪ���ȹ��壬���������оƾ��ơ����żܡ������ǡ������ȣ��ʴ�Ϊ�������ǡ�������
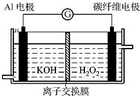
���� ������������������Ʊ�����ӵ�ص缫�������̣������˻��ϼ��жϡ����ӷ���ʽ��д���缫����ʽ��д����ѧ�����֪ʶ���漰�����ݽ϶࣬�ۺ��Խ�ǿ����ֿ�����ѧ�����ۺ������������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | 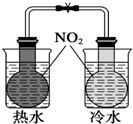 ֤���¶ȶ�ƽ���ƶ���Ӱ�� | B�� | 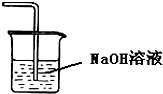 ����HClβ�� | ||
| C�� | 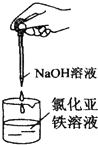 �Ʊ����������� | D�� | 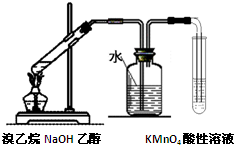 ��֤��������ȥ��Ӧ��������ϩ |
| A�� | NH4+��Na+��Br-��SO42- | |
| B�� | Al3+��Cl-��OH-��HCO3- | |
| C�� | pH=0����Һ�У�ClO-��SO42-��Fe2+��K+ | |
| D�� | ��ˮ�����c��OH-��=10-14mol•L-1����Һ�У�CH3COO-��SO42-��Na+��NH4+ |
| A�� | ���ȷ�Ӧ�������� | B�� | ���ȷ�Ӧ���ؼ�С | C�� | ���ȷ�Ӧ�������� | D�� | ���ȷ�Ӧ���ؼ�С |
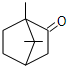

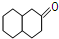


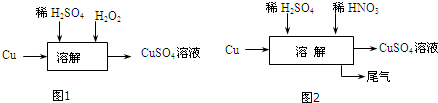
 ��������ͭ��������������Ӧ����㷺�Ľ������ϣ�
��������ͭ��������������Ӧ����㷺�Ľ������ϣ�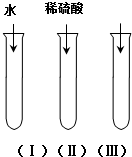 ����ͭ��CuO����һ�ֺ�ɫ���壬������ϡ�����У�ijͬѧ��֪����ϡ�����е�H2O��H+��SO42-�е�����������ʹ����ͭ�ܽ⣮�������һ��ͨ����ͼ��ʾ����ʵ����������̽�����
����ͭ��CuO����һ�ֺ�ɫ���壬������ϡ�����У�ijͬѧ��֪����ϡ�����е�H2O��H+��SO42-�е�����������ʹ����ͭ�ܽ⣮�������һ��ͨ����ͼ��ʾ����ʵ����������̽�����