��Ŀ����
6������˵�����ʾ��������ȷ���ǣ�������| A�� | �����ʵ������������������ֱ���ȫȼ�գ����߷ų��������� | |
| B�� | �ɵ���Aת��Ϊ����B��H=+119kJ/mol����֪����B�ȵ���A�ȶ� | |
| C�� | ϡ��Һ�У�H+��aq��+OH-��aq��=H2O��l����H=-57.3kJ/mol | |
| D�� | ��25�桢101kPaʱ��2g H2��ȫȼ������Һ̬ˮ���ų�285.8kJ���������ʾH2ȼ���ȵĻ�ѧ����ʽΪ2H2��g��+O2��g��=2H2O��l����H=-571.6kJ/mol |
���� A���������仯Ϊ�����Ϊ���ȹ��̣�
B���������������ߵIJ��ȶ��ش�
C����ϡ��Һ��ǿ����ǿ�Ӧ����1mol�ų�������Ϊ�к��ȣ�
D��ȼ��������һ�������£�1mol��ȼ����ȫȼ�������ȶ���������ʱ���ų���������
��� �⣺A���������仯Ϊ�����Ϊ���ȹ��̣����������������������������зֱ���ȫȼ�գ��ų������������࣬��ǰ�߷ų������࣬��A����
B���ɵ���Aת��Ϊ����B��H=+119kJ/mol����˵��A������������B��������������Խ��Խ�ȶ�����˵���A�ȵ���B�ȶ�����B����
C����ϡ��Һ��ǿ����ǿ�Ӧ����1mol�ų�������Ϊ�к��ȣ�ϡ��Һ�У�H+��aq��+OH-��aq��=H2O��l����H=-57.3kJ/mol��ʾ�к��ȣ���C��ȷ��
D��ȼ��������һ�������£�1mol��ȼ����ȫȼ�������ȶ���������ʱ���ų�����������25�桢101kPaʱ��2g H2��ȫȼ������Һ̬ˮ���ų�285.8kJ���������ʾH2ȼ���ȵĻ�ѧ����ʽΪH2��g��+$\frac{1}{2}$O2��g��=H2O��l����H=-285.8kJ/mol����D����
��ѡC��
���� ���⿼���˻�ѧ��Ӧ�е������仯�����ڻ���֪ʶ�Ŀ��飬��Ŀ�ѶȲ���ע������к��Ⱥ�ȼ���ȵĸ��

 ��У����ϵ�д�
��У����ϵ�д�| A�� | �����ụΪͬ���칹�� | B�� | ���ڸ߷��ӻ����� | ||
| C�� | ���ܷ���������Ӧ | D�� | ������ȼ�� |
| A�� | 11 | B�� | 7 | C�� | 5 | D�� | 3 |
 ������о���Ա������һ�ִ�����Ũ����ȩ��ˮ���·���--��Ĥ��ⷨ����ȩ�ֱ�����������������Ӧ��ת��Ϊ�Ҵ������ᣮʵ������һ��Ũ�ȵ���ȩ-Na2SO4��ҺΪ�������Һ��ģ����ȩ��ˮ�Ĵ������̣���װ����ͼ��ʾ������˵����ȷ���ǣ�������
������о���Ա������һ�ִ�����Ũ����ȩ��ˮ���·���--��Ĥ��ⷨ����ȩ�ֱ�����������������Ӧ��ת��Ϊ�Ҵ������ᣮʵ������һ��Ũ�ȵ���ȩ-Na2SO4��ҺΪ�������Һ��ģ����ȩ��ˮ�Ĵ������̣���װ����ͼ��ʾ������˵����ȷ���ǣ�������| A�� | �������У�����������Na+ | |
| B�� | ������ӦΪCH3CHO-2e-+2H+�TCH3COOH+H2O | |
| C�� | �����ܷ�Ӧ����ʽΪ2CH3CHO+H2O$\frac{\underline{\;���\;}}{\;}$CH3COOH+CH3CH2OH | |
| D�� | ����CH4-����ȼ�ϵ��Ϊֱ����Դ��ȼ�ϵ�ص�b��Ӧͨ����� |
| A�� | �٢ܶ�Ҫ�õ���ƿ | B�� | �ڢ۶�Ҫ�õ��¶ȼ� | ||
| C�� | �ۢܶ�Ҫ�õ��ƾ��� | D�� | �٢ڶ�Ҫ�õ�ָʾ�� |
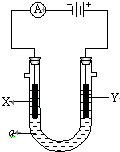 ���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ������ֱ����Դ��������ش��������⣺
���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ������ֱ����Դ��������ش��������⣺

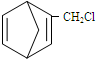 ��������ṹ��ԭ�Ͽ�����
��������ṹ��ԭ�Ͽ�����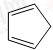 ��
�� ����д�ṹ��ʽ��
����д�ṹ��ʽ�� �ĺϳ�·�ߣ��������Լ���ѡ����
�ĺϳ�·�ߣ��������Լ���ѡ����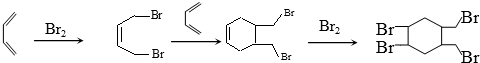 ��
��