��Ŀ����
11����ϩ�����л������е���Ҫ���ʣ������д��ڵ�˫��������ֵĽṹ��Ϊ����ṹ��������ṹ�Ķ�ϩ����Ϊ����ϩ�������������գ���1�����������к��й���ṹ����bd�������ţ�

��2�������ϩ���ļӳ�λ���ж��ֿ��ܣ���Ȼ�ĵ������巴Ӧ�IJ��������4�֣�
��3�������ϩ��������������뺬̼̼˫���������Ļ�������мӳɣ����磺

��Ҫ�Ʊ�
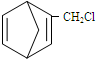 ��������ṹ��ԭ�Ͽ�����
��������ṹ��ԭ�Ͽ�����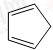 ��
�� ����д�ṹ��ʽ��
����д�ṹ��ʽ����4�����һ����1��3-����ϩ�ϳ���ȼ��
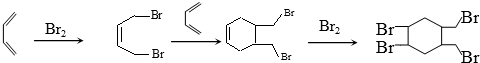
 �ĺϳ�·�ߣ��������Լ���ѡ����
�ĺϳ�·�ߣ��������Լ���ѡ�������ϳ�·�߳��õı�ʾ��ʽΪ��A$��_{��Ӧ����}^{��Ӧ�Լ�}$B��$��_{��Ӧ����}^{��Ӧ�Լ�}$Ŀ����� ��
 ��
��
���� ��1���ݹ���ṹ�ĺ��������
��2����Ȼ���������ϩ���ṹ��ʽΪ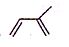 ����ѧ������2-��-1��3-����ϩ���ݴ˷�����
����ѧ������2-��-1��3-����ϩ���ݴ˷�����
��3��Ҫ�Ʊ�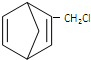 ����Ҫ���й���ṹ��5Ԫ����
����Ҫ���й���ṹ��5Ԫ����
��4����1��3-����ϩ�ϳ�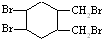 ����������1��3-����ϩ����1��1�ӳɣ�����1��4-����-2-��ϩ������1��3-����ϩ��Ӧ�õ�6Ԫ��������ӳ��Ƶã�
����������1��3-����ϩ����1��1�ӳɣ�����1��4-����-2-��ϩ������1��3-����ϩ��Ӧ�õ�6Ԫ��������ӳ��Ƶã�
��� �⣺��1�������д��ڵ�˫��������ֵĽṹ��Ϊ����ṹ�����Ϲ���ṹ��Ϊbd���ʴ�Ϊ��bd��
��2����Ȼ���������ϩ���ṹ��ʽΪ ��������ӳɲ������Ϊ��CH2BrCHBrC��CH3��=CH2��CH2=CHC��CH3��BrCH2Br��CH2BrCH=C��CH3��CH2Br��CH2BrCHBrC��CH3��BrCH2Br����4�֣��ʴ�Ϊ��4��
��������ӳɲ������Ϊ��CH2BrCHBrC��CH3��=CH2��CH2=CHC��CH3��BrCH2Br��CH2BrCH=C��CH3��CH2Br��CH2BrCHBrC��CH3��BrCH2Br����4�֣��ʴ�Ϊ��4��
��3����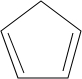 ��
��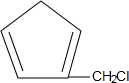 �����Ƶ�
�����Ƶ� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
�� ��
��
��4����1��3-����ϩ�ϳ� �ĺϳ�·��Ϊ
�ĺϳ�·��Ϊ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л�����ĺϳɣ�����ϩ�������ʣ���Ŀ�ѶȲ���ע������л�������ŵı仯�Լ���Ӧ������

��ϰ��ϵ�д�
�����Ŀ
2���������ӷ�Ӧ����ʽ����д��ȷ���ǣ�������
| A�� | ������ˮ�ķ�Ӧ��2Na+2H2O�T2NaOH+H2�� | |
| B�� | ̼������Һ��ϡ���ᷴӦ��CO2-3+2H+�TH2O+CO2�� | |
| C�� | ��Ba��OH��2��Һ����μ�������������Һ�����ԣ�2H++SO2-4+Ba2++2OH-�TBaSO4��+2H2O | |
| D�� | �������м���ϡ���FeS+2H+�TFe2++H2S�� |
6������˵�����ʾ��������ȷ���ǣ�������
| A�� | �����ʵ������������������ֱ���ȫȼ�գ����߷ų��������� | |
| B�� | �ɵ���Aת��Ϊ����B��H=+119kJ/mol����֪����B�ȵ���A�ȶ� | |
| C�� | ϡ��Һ�У�H+��aq��+OH-��aq��=H2O��l����H=-57.3kJ/mol | |
| D�� | ��25�桢101kPaʱ��2g H2��ȫȼ������Һ̬ˮ���ų�285.8kJ���������ʾH2ȼ���ȵĻ�ѧ����ʽΪ2H2��g��+O2��g��=2H2O��l����H=-571.6kJ/mol |
16����NAΪ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | �ܱ�������2molNO��1molO2��ַ�Ӧ������������ķ�����Ϊ2NA | |
| B�� | �ö��Ե缫���CuSO4��Һ���������0.1molCu��OH��2��ʹ��Һ��ԭ�����·��ת�Ƶ��ӵ���ĿΪ0.2NA | |
| C�� | 142 g Na2SO4��Na2HPO4���������У�������������Ϊ3NA | |
| D�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.4NA |
20��0.5molij�����������Ҵ�����������Ӧ����������������ԭ���������������28g����ԭ��������ǣ�������
| A�� | ���� | B�� | �Ҷ��� | C�� | ���� | D�� | ���� |
16����������VB2��-������ع���ʱ��ӦΪ��4VB2+11O2�T4B2O3+2V2O5���øõ��Ϊ��Դ��ѡ�ö��Ե缫�������ͭ��Һ��ʵ��װ����ͼ��ʾ�������·��ͨ��0.04mol����ʱ��Bװ���ڹ��ռ���0.448L���壨��״������������˵����ȷ���ǣ�������

| A�� | VB2�缫�����ĵ缫��ӦΪ��2VB2+11H2O-22e-�TV2O5+2B2O3+22H+ | |
| B�� | ���·�е�����c�缫����VB2�缫 | |
| C�� | �������У�c�缫�������к�ɫ�����������������ݲ��� | |
| D�� | ��Bװ���ڵ�Һ�����Ϊ100 mL����CuSO4��Һ�����ʵ���Ũ��Ϊ0.05 mol/L |
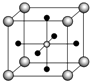 ������������Խ������Ϊ��Fe��Al��Ӧ�ù㷺�ġ�������������
������������Խ������Ϊ��Fe��Al��Ӧ�ù㷺�ġ�������������