��Ŀ����
ʵ�������Ȼ��ƹ�������1.00mol/L��NaCl��Һ0.5L���ش���������
��1�����ݼ�����������ƽ���ȡ�Ȼ��� g��
��2������ʱ�������õ��IJ����������ձ� ������ ��
��3��������Һ�����м���������a���ܽ⣬b��ҡ�ȣ�c��ϴ�ӣ�d����ȴ��e��������
f������Һ��������ƿ��g������ǩ��h�����ݣ�
����ȷ�IJ���˳���� ��
��4�����в������ʹ��Һ���ʵ���Ũ��ƫ�͵���
A��û�н�ϴ��Һת������ƿ�У�
B������ƿϴ�Ӻ�δ�����ﴦ����
C������ʱ����������ƿ�Ŀ̶��ߣ�
D����ˮ����ʱ��ˮ���������˿̶��ߣ�
��5��ȡ����NaCl��Һ10mL��ˮϡ�͵�100mL��ϡ�ͺ���Һ��NaCl�����ʵ���Ũ���� ��
��6��д������NaCl��Һ�к��������ӵ��йط�Ӧ����ʽ�� ��
��1�����ݼ�����������ƽ���ȡ�Ȼ���
��2������ʱ�������õ��IJ����������ձ� ������
��3��������Һ�����м���������a���ܽ⣬b��ҡ�ȣ�c��ϴ�ӣ�d����ȴ��e��������
f������Һ��������ƿ��g������ǩ��h�����ݣ�
����ȷ�IJ���˳����
��4�����в������ʹ��Һ���ʵ���Ũ��ƫ�͵���
A��û�н�ϴ��Һת������ƿ�У�
B������ƿϴ�Ӻ�δ�����ﴦ����
C������ʱ����������ƿ�Ŀ̶��ߣ�
D����ˮ����ʱ��ˮ���������˿̶��ߣ�
��5��ȡ����NaCl��Һ10mL��ˮϡ�͵�100mL��ϡ�ͺ���Һ��NaCl�����ʵ���Ũ����
��6��д������NaCl��Һ�к��������ӵ��йط�Ӧ����ʽ��
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺ʵ����
��������1������n=cV��m=nM�����㣻
��2���������Ʋ����Ǽ��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������Ҫ��������
��3�������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ���Բ���˳���������
��4������c=
��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��5��������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
��6������Cl-�ļ��鷽����������
��2���������Ʋ����Ǽ��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������Ҫ��������
��3�������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ���Բ���˳���������
��4������c=
| n |
| V |
��5��������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
��6������Cl-�ļ��鷽����������
���
�⣺��1������1.00mol/L��NaCl��Һ0.5L������Ȼ��Ƶ����ʵ���n=cV=1mol/L��0.5L=0.5mol/L������m=nM=0.5mol��58.5g/mol=29.3g���ʴ�Ϊ��29.3��
��2�����������м��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣮��ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ���������IJ����������ձ�����������500mL����ƿ����ͷ�ιܣ��ʴ�Ϊ��500mL����ƿ����ͷ�ιܣ�
��3�������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��֪��ȷ�IJ���˳���ǣ�e a d f c f h b g���ʴ�Ϊ��e a d f c f h b g��
��4��A��û�н�ϴ��Һת������ƿ�У��ᵼ�����ʵ���ʧ��ʹŨ��ƫ�ͣ���Aѡ��
B��������ƿδ���T����������Һ������ҺŨ����Ӱ�죬��ΪֻҪ����ʱ��ȷ������ˮ��ԭ�����еĻ��Ǻ�������ģ���Ũ����Ӱ�죬��B��ѡ��
C������ʱ����������ƿ�Ŀ̶��ߣ��ᵼ����Һ���ƫС����Ũ��ƫ�ߣ���C��ѡ��
D����ˮ����ʱ��ˮ���������˿̶��ߣ��ᵼ��Ũ��ƫ�ͣ���Dѡ��
��ѡAD��
��5����ϡ�ͺ���Һ��NaCl�����ʵ���Ũ����Xmol/L��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��
10mL��1mol/L=100mL��Xmol/L
���X=0.1mol/L���ʴ�Ϊ��0.1mol/L��
��6��ͨ���������������������ӵĴ��ڣ���ѧ����ʽ��NaCl+AgNO3�TAgCl��+NaNO3���ʴ�Ϊ��NaCl+AgNO3�TAgCl��+NaNO3��
��2�����������м��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣮��ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ���������IJ����������ձ�����������500mL����ƿ����ͷ�ιܣ��ʴ�Ϊ��500mL����ƿ����ͷ�ιܣ�
��3�������Ʋ����Ǽ��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��֪��ȷ�IJ���˳���ǣ�e a d f c f h b g���ʴ�Ϊ��e a d f c f h b g��
��4��A��û�н�ϴ��Һת������ƿ�У��ᵼ�����ʵ���ʧ��ʹŨ��ƫ�ͣ���Aѡ��
B��������ƿδ���T����������Һ������ҺŨ����Ӱ�죬��ΪֻҪ����ʱ��ȷ������ˮ��ԭ�����еĻ��Ǻ�������ģ���Ũ����Ӱ�죬��B��ѡ��
C������ʱ����������ƿ�Ŀ̶��ߣ��ᵼ����Һ���ƫС����Ũ��ƫ�ߣ���C��ѡ��
D����ˮ����ʱ��ˮ���������˿̶��ߣ��ᵼ��Ũ��ƫ�ͣ���Dѡ��
��ѡAD��
��5����ϡ�ͺ���Һ��NaCl�����ʵ���Ũ����Xmol/L��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��
10mL��1mol/L=100mL��Xmol/L
���X=0.1mol/L���ʴ�Ϊ��0.1mol/L��
��6��ͨ���������������������ӵĴ��ڣ���ѧ����ʽ��NaCl+AgNO3�TAgCl��+NaNO3���ʴ�Ϊ��NaCl+AgNO3�TAgCl��+NaNO3��
���������⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
���ж���ijЩ���ӵļ���˵������ȷ���ǣ�������
| A������ϡ���������ɫ���壬������ͨ�����ʯ��ˮ�У���Һ����ǣ�һ����CO32- |
| B��ij��Һ��NaOH��Һ���ȣ�����ʹʪ����ɫʯ����ֽ������壬˵��ԭ��Һ�д���NH4+ |
| C�������Ȼ�����Һ�а�ɫ�����������ټ����ᣬ��������ʧ��һ����SO42- |
| D��������ɫ��Ӧʵ�������Һ���Ƿ���K+ |
5.6g������һ��������ǻ�ϣ��ڿ����м��ȷ������ҷ�Ӧ����ȴ���������������ܽⷴӦ��IJ������壬������2��OL���壨��״������ͨ������С����Ӧ���ɵ����������������ԭ���ǣ�������
| A����������� |
| B��������FeS |
| C��������Fe2S3 |
| D�����Fe��������������� |
����NaOH��Na2CO3��Ba��OH��2������Һ�����ѡ�������Լ��еģ�������
| A��ʯ����Һ |
| B��ϡ���� |
| C��ϡ���� |
| D��CaCl2��Һ |
��ʢ����ˮ���Թ��У���������CCl4��������Ƭ�̺�����
| A��������Һ��Ⱥ�ɫ |
| B��������Һ��Ϊ�Ϻ�ɫ |
| C���ϲ�Ϊ��ɫ���²�Ϊ�Ⱥ�ɫ |
| D���²���ɫ���ϲ��Ϻ�ɫ |
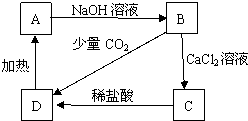 A��B��C��D��Ϊ����Ļ��������B��ˮ��Һ�׳�ˮ���������ת����ϵ��ͼ��ʾ��
A��B��C��D��Ϊ����Ļ��������B��ˮ��Һ�׳�ˮ���������ת����ϵ��ͼ��ʾ��