��Ŀ����
20�������й�ʵ�������ơ������¼�����۽��Ͷ���ȷ���ǣ�������| ʵ�������� | �����¼ | ���۽��� | |
| A | ������Ũ������μ���Cu ��ϡ����Ļ������ | ��������ɫ���� | ���ᱻ��ԭΪNO2 |
| B | ������ǯ��ס�����ھƾ��� �ϼ��� | �����ۻ����������� | �۵㣺Al2 O3��Al |
| C | ��ij�Ȼ�������Һ�м��� Na2O2��ĩ | ���ֺ��ɫ���� | ����Na2O2��ĩǰ��ԭ�Ȼ� ������Һ�Ѿ����� |
| D | ����ɫʯ����Һ�г�����ʱ ��ͨ������ | ��Һ�ȱ�죬���� Ϊ��ɫ | ������Ư���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
���� A����������NO��NO��������Ӧ����NO2��
B�����ڼ�����������������Ӧ������������
C���������ƾ���ǿ�����ԣ��������������ӣ�
D��������ˮ��Ӧ���ɾ���Ư���ԵĴ����ᣬ����������Ư���ԣ�
��� �⣺A��Ũ�������ϡ������Һ�У�Ũ�Ƚ��ͣ���������NO��NO��������Ӧ����NO2����A����
B�����ڼ�����������������Ӧ�����������������ۻ����������䣬��˵���۵㣺Al2O3��Al����B��ȷ��
C���������ƾ���ǿ�����ԣ��������������ӣ�����֤���Ȼ�������Һ�Ѿ����ʣ���C����
D������ɫʯ����Һ�г�����ʱ��ͨ��������������ˮ��Ӧ��������ʹ����ᣬ��Һ�ȱ�죬����Ϊ��ɫ����������ֳ�Ư���ԣ���������������Ư���ԣ���D����
��ѡB��
���� ���⿼���Ϊ�ۺϣ��漰Ԫ�ػ���������ʵ��ۺ����������õĿ��飬Ϊ�߿��������ͣ�ע��������ʵ����ʵ���ͬ�Լ�ʵ��������ԡ������Ե����ۣ��ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
10������ͼʾʵ����ȷ���ǣ�������
| A�� | ����Cu��Ũ���ᷴӦ��������������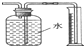 | |
| B�� | ��֤Na��ˮ��ӦΪ���ȷ�Ӧ��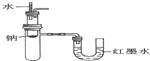 | |
| C�� | ��ȡ����İ����� | |
| D�� | �ռ�H2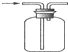 |
15�������±��и�����Ų����ɣ��йص�23��24��˵���в���ȷ���ǣ�������
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | �� | 23 | 24 |
| C2H6 | C2H6O | C2H4O2 | C3H8 | C3H8O | C3H6O2 | C4H10 | �� | M | N |
| A�� | MΪC9H20O | B�� | NΪC9H18O2 | ||
| C�� | M��N���ɷ����ӳɷ�Ӧ | D�� | M��N�����ܷ���ȡ����Ӧ |
5����ͼ��ʾ�������̶�������������������ƶ���M��N���������о�������Ӧ��3A��g��?xB��g��+xC��g����H=-192kJ•mol-1����M��N�ж�ͨ��3mol A �����壬��ʼM��N�ݻ���ͬ�������¶Ȳ��䣮����˵����ȷ���ǣ�������
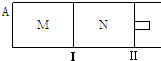

| A�� | ��x=2���ﵽƽ���B �����������ϵΪ���գ�M�����գ�N�� | |
| B�� | ��x��2���ﵽƽ���B��ת���ʹ�ϵΪ������M��������N�� | |
| C�� | ��x��2��C��ƽ�����ʵ���Ũ�ȹ�ϵΪ��c��M����c��N�� | |
| D�� | x����Ϊ��ֵ����ʼʱ��N�����г�������ֵ��A��ƽ���N������B��Ũ�Ⱦ���� |
12�������£����й��ڵ������Һ��˵����ȷ���ǣ�������
| A�� | ��pH=4��CH3COOH��Һ��ˮϡ��10������Һ�и�����Ũ�Ⱦ���С | |
| B�� | ��CH3COOH��Һ�ζ������ʵ���Ũ��NaOH��Һ��pH=7��V��CH3COOH����V��NaOH����Һ | |
| C�� | ��0.2mol/L��������Һ�м�������0.1mol•L-1��NH3•H2O��Һ��c��Cl-��+c��OH-��=c��H+��+c��NH3•H2O�� | |
| D�� | �ں�0.1mol NaHSO4��Һ�У�c��H+��=c��SO42-��+c��OH-�� |
11��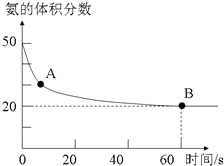 ������һ����Ҫ�Ļ���ԭ�ϣ��ڹ�ũҵ�ж��й㷺��Ӧ�ã�
������һ����Ҫ�Ļ���ԭ�ϣ��ڹ�ũҵ�ж��й㷺��Ӧ�ã�
��1��NH3��CO2��120��ʹ����������¿��Ժϳ����أ���Ӧ����ʽ���£�2NH3��g��+CO2��g��?CO��NH2��2��s��+H2O��g����ijʵ��С����һ���ݻ����������ܱ������г���CO2��NH3�ϳ����أ��ں㶨�¶��£����������NH3�ĺ�����ʱ��ı仯��ϵ��ͼ��ʾ��������������Ϊ���壩��A�������Ӧ����v����CO2������B����淴Ӧ����v����CO2��������ڡ�����С�ڡ����ڡ�����NH3��ƽ��ת����Ϊ75%��
��2����������泥�NH2COONH4���Ǻϳ����ع����е��м����ֽ������Ϊ2��1��NH3��CO2����������һ���ݻ����������ܱ������У��ں㶨�¶���ʹ�䷢����Ӧ���ﵽƽ�⣺2NH3��g��+CO2��g��?NH2COONH4��s����ʵ�����ڲ�ͬ�¶��µ�ƽ���������±���
��������Ӧ���ʱ䣺��H��0���ر��S��0�����������������=���������ݱ������ݣ��г�15.0��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ���ļ���ʽK=$\frac{1}{��1.6��1{0}^{-3}��^{2}����0.8��1{0}^{-3}��}$����Ҫ�����������
�������Ѵ�ƽ��״̬�����������з���������İ�������茶��壬��Ӧ���ת���ʽ����䣨���������С�����䡱����
��3����a mol NH4NO3����Һ�еμ�b L��ˮ����Һǡ�ó����ԣ���μӰ�ˮ�Ĺ�����ˮ�ĵ���ƽ�⽫
����������������������ƶ������μӰ�ˮ��Ũ��Ϊ$\frac{a}{200b}$mol•L-1��25��ʱ��NH3•H2O�ĵ���ƽ�ⳣ��Kb=2��10-5��
 ������һ����Ҫ�Ļ���ԭ�ϣ��ڹ�ũҵ�ж��й㷺��Ӧ�ã�
������һ����Ҫ�Ļ���ԭ�ϣ��ڹ�ũҵ�ж��й㷺��Ӧ�ã���1��NH3��CO2��120��ʹ����������¿��Ժϳ����أ���Ӧ����ʽ���£�2NH3��g��+CO2��g��?CO��NH2��2��s��+H2O��g����ijʵ��С����һ���ݻ����������ܱ������г���CO2��NH3�ϳ����أ��ں㶨�¶��£����������NH3�ĺ�����ʱ��ı仯��ϵ��ͼ��ʾ��������������Ϊ���壩��A�������Ӧ����v����CO2������B����淴Ӧ����v����CO2��������ڡ�����С�ڡ����ڡ�����NH3��ƽ��ת����Ϊ75%��
��2����������泥�NH2COONH4���Ǻϳ����ع����е��м����ֽ������Ϊ2��1��NH3��CO2����������һ���ݻ����������ܱ������У��ں㶨�¶���ʹ�䷢����Ӧ���ﵽƽ�⣺2NH3��g��+CO2��g��?NH2COONH4��s����ʵ�����ڲ�ͬ�¶��µ�ƽ���������±���
| �¶ȣ��棩 | 15.0 | 20.0 | 25.0 | 30.0 | 35.0 |
| ƽ��������Ũ�� ��10-3mol/L�� | 2.4 | 3.4 | 4.8 | 6.8 | 9.4 |
�������Ѵ�ƽ��״̬�����������з���������İ�������茶��壬��Ӧ���ת���ʽ����䣨���������С�����䡱����
��3����a mol NH4NO3����Һ�еμ�b L��ˮ����Һǡ�ó����ԣ���μӰ�ˮ�Ĺ�����ˮ�ĵ���ƽ�⽫
����������������������ƶ������μӰ�ˮ��Ũ��Ϊ$\frac{a}{200b}$mol•L-1��25��ʱ��NH3•H2O�ĵ���ƽ�ⳣ��Kb=2��10-5��
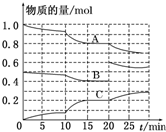 �ں㶨�¶�T�棬2L�����ܱ������з�����ӦaA��g��+bB��g��?cC��g����������A��B��C���ʵ����仯��ͼ��ʾ���ش��������⣺
�ں㶨�¶�T�棬2L�����ܱ������з�����ӦaA��g��+bB��g��?cC��g����������A��B��C���ʵ����仯��ͼ��ʾ���ش��������⣺