��Ŀ����
17��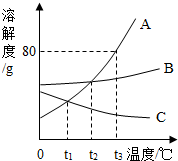 ��ͼ��ʾ��A��B��C���ֹ������ʵ��ܽ�����ߣ���ش��������⣺
��ͼ��ʾ��A��B��C���ֹ������ʵ��ܽ�����ߣ���ش��������⣺��1��S��ʾ���ʵ��ܽ�ȣ���t2��ʱSA=SB��SC
��2��t2��ʱ����80g A���ʷ���100g ˮ�У���ֽ���õ�A������Һ�DZ��ͣ���ͻ��ͣ���Һ�������¶ȵ����ߣ�A��B��C�������ʵı�����Һ�������ʴ���Һ����������C��
��3��t3��ʱ����80g A��B�������ʷֱ����100gˮ�У���ֽ��������t1�棬�õ�A������Һ�������������������=��������B���ʵ�������
���� ���ݹ�����ܽ�����߿��ԣ��ٲ��ij������ij�¶��µ��ܽ�ȣ��ڱȽϲ�ͬ��������ͬһ�¶��µ��ܽ�ȴ�С�����ж����ʵ��ܽ�����¶ȱ仯�ı仯����ȣ�
��� �⣺��1�����ܽ������ͼ���Կ���t2��ʱ��SA=SB��SC��
��2��t2��ʱA���ܽ��С��80g����100gˮ������ܽ� ��A������С��80g������80g A���ʷ���100g ˮ�У���ֽ���õ�A���ʵı�����Һ�������¶ȵ����ߣ�AB���ܽ������ʣ���������ܽ⣬C���ܽ�ȼ�С��������������
��3��t3��ʱ����80g A��B�������ʷֱ����100gˮ�У���ֽ��������t1�棬B���ܽ�ȴ���A���ܽ�ȣ��Ҿ�С��80g�������� ��100gˮ���ܽ��AС��B�����Եõ�A������Һ������С��B��Һ��������
�ʴ�Ϊ����1��t2��
��2�����ͣ�C��
��3������
���� �����ѶȲ��Ǻܴ���Ҫ�����˹������ʵ��ܽ����������ʾ�����弰�����ܽ�����߽����ص����⣬�Ӷ�����ѧ���������⡢��������������

��ϰ��ϵ�д�
 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�
�����Ŀ
7��þ�ڶ�����̼��ȼ�գ�������̼������þ���÷�Ӧ��Mg��CO2��C��MgO��������Ϊ��������
| A�� | 12��11��3��20 | B�� | 24��44��12��40 | C�� | 6��11��3��10 | D�� | 12��22��6��20 |
5����ǧ���������ɽ���һ���������У��۹������벻�£�Ҫ��������˼䣮��������ǫ�ġ�ʯ���������̺���һ���Ŀ�ѧ֪ʶ��ʫ���漰�й�ʯ��ʯ����ʯ�ҡ���ʯ��֮���ת����������ѧ��Ӧ�������⼸����Ӧ���漰�Ļ�����Ӧ�����ǣ�������
| A�� | ���Ϸ�Ӧ | B�� | �û���Ӧ | C�� | �ֽⷴӦ | D�� | ���ֽⷴӦ |
12��ijͬѧ����ѧ���ֻ�ѧ֪ʶ�������£������д����һ���ǣ�������
| A | B |
| ������������; O2��ȼ--�����и���� H2O2���ֽ�ų�O2--ʵ������O2 | �ճ������ �·���Բ�����--�����ͻ�ƾ���ϴ ��������ë֯��--ȡ��������ȼ����ζ |
| C | D |
| ��ȫ��ʶ ú��й©--��110�绰���� Ƥ��մ��Ũ����--��ˮ��ϴ����Ϳϡ���� | ��ѧ������ ά����C--ȱ��ʱ����Ѫ�� ���ࡢ������--���������ȡ��Ӫ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
2�����л�ѧ����ʽ��д��ȷ���ǣ�������
| A�� | Fe+2AgCl�T2Ag+FeCl2 | B�� | Al+H2SO4�TAlSO4+H2�� | ||
| C�� | C+2CuO$\frac{\underline{\;����\;}}{\;}$2Cu+CO2 | D�� | C+H2O$\frac{\underline{\;����\;}}{\;}$H2+CO |
7��ʵ������Ϊ�˼���2ƿʧȥ��ǩ��NaOH��Һ��Ca��OH��2��Һ����������ʵ�飺
��1����ȡ2mL��Һ���Թ��У��ֱ����Na2CO3��Һ��������Һ����ǣ���Ca��OH��2��Һ����Ӧ�Ļ�ѧ����ʽCa��OH��2+Na2CO3�TCaCO3��+2NaOH��
��2����ȡ2mL��Һ���Թ��У�ͨ��CO2Ҳ�ܼ���������������û�ѧҩƷ��ȡCO2�����ҵIJ������ò����ֱܷ�����Һ�д�����
��3��NaOH��Һ��ͨ��CO2�����Ա仯���ڷ�Ӧ�����Һ�м������������Բ���֤��NaOH��CO2��Ӧ����C��A��ϡ���� B��Ba��OH��2��Һ C����̪��Һ
��4��NaOH��Һ��ͨ��������CO2��������Һ�����ʣ�������Na2CO3��Ҳ�������Ȼ��ƣ�
��1����ȡ2mL��Һ���Թ��У��ֱ����Na2CO3��Һ��������Һ����ǣ���Ca��OH��2��Һ����Ӧ�Ļ�ѧ����ʽCa��OH��2+Na2CO3�TCaCO3��+2NaOH��
��2����ȡ2mL��Һ���Թ��У�ͨ��CO2Ҳ�ܼ���������������û�ѧҩƷ��ȡCO2�����ҵIJ������ò����ֱܷ�����Һ�д�����
��3��NaOH��Һ��ͨ��CO2�����Ա仯���ڷ�Ӧ�����Һ�м������������Բ���֤��NaOH��CO2��Ӧ����C��A��ϡ���� B��Ba��OH��2��Һ C����̪��Һ
��4��NaOH��Һ��ͨ��������CO2��������Һ�����ʣ�������Na2CO3��Ҳ�������Ȼ��ƣ�
| �� �� | �� �� | ���� |
| ����Ӧ�����Һ�м����������Ȼ�����Һ�� | ��Һ����� | ������ Na2CO3 |
| �ھ��ú��ò�����պȡ�ϲ���Һ������pH��ֽ�ϣ� | pH=7 |
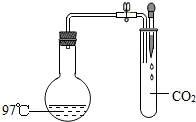 ��ͼ���ȹر�ֹˮ�У�����ͷ�ι��е�Һ�強ѹ���Թ��У�Ȼ���ֹˮ�У�������ƿ�ڵ�ˮ����������
��ͼ���ȹر�ֹˮ�У�����ͷ�ι��е�Һ�強ѹ���Թ��У�Ȼ���ֹˮ�У�������ƿ�ڵ�ˮ����������