��Ŀ����
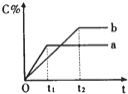
����Ŀ��700��ʱ,���ݻ�Ϊ2L���ܱ������г���һ������CO��H2O,������Ӧ��CO(g)+H2O(g)CO2(g)+H2(g)��Ӧ�����вⶨ�IJ������ݼ��±�(����t2
��Ӧʱ��/min | n(CO)/mol | n(H2O)/mol |
0 | 1.20 | 0.60 |
t1 | 0.80 | |
t2 | 0.20 |
����˵����ȷ����
A. ��Ӧ��t1 min�ڵ�ƽ������Ϊv(H2)=0.40/t1molL1min1
B. ����������������,��ʼʱ�������г���0.60 mol CO��0.10 mol H2O,�ﵽƽ��ʱn(CO2)=0.20 mol
C. ����������������,��ƽ����ϵ����ͨ��0.20 mol H2O,��ԭƽ�����,�ﵽ��ƽ��ʱCO��H2Oת��������
D. �¶�������800�棬�ﵽ��ƽ��ʱn(CO2)=0.34 mol��������ӦΪ���ȷ�Ӧ
���𰸡�D
��������A���ɱ������ݿ�֪��t1min�ڲμӷ�Ӧ��CO�����ʵ���Ϊ1.2mol-0.8mol=0.4mol��v��CO��=![]() =
=![]() mol/��Lmin��������֮�ȵ��ڻ�ѧ������֮�ȼ���v��H2��=v��CO��=
mol/��Lmin��������֮�ȵ��ڻ�ѧ������֮�ȼ���v��H2��=v��CO��=![]() mol/��Lmin������A����B��CO��H2O�����ʵ�����1��1��Ӧ������0.60 mol CO��0.1mol H2O���������ɵ�0.2mol n��CO2������B����C�����������������䣬��ƽ����ϵ����ͨ��0.20molH2O����ԭƽ����ȣ�ƽ�������ƶ����ﵽ��ƽ��ʱCOת��������H2Oת���ʼ�С����C����D��700��ʱ��t1minʱn��CO��=0.8mol��n��H2O��=0.6mol-0.4mol=0.2mol��t2minʱn��H2O��=0.2mol��˵��t2minʱ��Ӧ�Ѿ��ﵽƽ��״̬����ʱn��CO��=0.8mol��n��H2O��=0.2mol��n��CO2��=c��H2��=��n��CO��=1.2mol-0.8mol=0.4mol���¶�������800�棬�ﵽ��ƽ��ʱn(CO2)=0.34mol��˵�������¶ȣ�ƽ�����ƶ���������ӦΪ���ȷ�Ӧ����D��ȷ����ѡD��
mol/��Lmin������A����B��CO��H2O�����ʵ�����1��1��Ӧ������0.60 mol CO��0.1mol H2O���������ɵ�0.2mol n��CO2������B����C�����������������䣬��ƽ����ϵ����ͨ��0.20molH2O����ԭƽ����ȣ�ƽ�������ƶ����ﵽ��ƽ��ʱCOת��������H2Oת���ʼ�С����C����D��700��ʱ��t1minʱn��CO��=0.8mol��n��H2O��=0.6mol-0.4mol=0.2mol��t2minʱn��H2O��=0.2mol��˵��t2minʱ��Ӧ�Ѿ��ﵽƽ��״̬����ʱn��CO��=0.8mol��n��H2O��=0.2mol��n��CO2��=c��H2��=��n��CO��=1.2mol-0.8mol=0.4mol���¶�������800�棬�ﵽ��ƽ��ʱn(CO2)=0.34mol��˵�������¶ȣ�ƽ�����ƶ���������ӦΪ���ȷ�Ӧ����D��ȷ����ѡD��