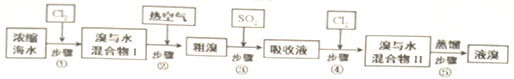
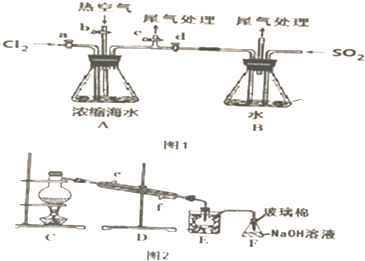
��Ŀ����
2����NA Ϊ�����ӵ�������ֵ������˵����ȷ���ǣ�������| A�� | ��״���£�1.12 L CCl4����C-Cl������ĿΪ0.2NA | |
| B�� | 1 L 1 mol/L AlCl3��Һ�к���NA��Al3+ | |
| C�� | ���³�ѹ�£�4.4 g CO2��N2O�Ļ�������к��е�ԭ������Ϊ0.3NA | |
| D�� | 80 mL 10 mol•L-1����������MnO2���ȷ�Ӧ��������Cl2������Ϊ0.2NA |
���� A����״���£����Ȼ�̼��״̬������̬��
B������������Һ�в���ˮ�⣬������������Ŀ���٣�
C��������̼������������Ħ����������44g/mol�����ߵķ����ж�����3��ԭ�ӣ�
D��Ũ������ϡ������ٷ�Ӧ����Ӧ���ɵ������������٣�
��� �⣺A����������Ȼ�̼������̬������ʹ�ñ���µ�����Ħ��������㣬��A����
B��1L 1mol/L AlCl3��Һ�к���1mol�Ȼ��������ڲ���������ˮ�⣬����Һ�к��е�������С��1mol�����е���������ĿС��NA����B����
C�����³�ѹ�£�4.4 g CO2��N2O�Ļ����������ʵ���Ϊ$\frac{4.4g}{44g/mol}$=0.1mol��0.1mol���ߵĻ�����к���0.3molԭ�ӣ����е�ԭ������Ϊ0.3NA����C��ȷ��
D��80mL 10mol•L-1�����к���0.8molHCl����������̵ķ�Ӧ������0.8molHCl������0.2mol����������Ũ������ϡ�����Ӧֹͣ����Ӧ���������������ʵ���С��0.2mol��������Cl2������С��0.2NA����D����
��ѡC��
���� ���⿼�鰢���ӵ��������йؼ�����жϣ���Ŀ�Ѷ��еȣ�ע�����պ������ʵ���Ϊ���ĵĸ���ѧ���밢���ӵ������Ĺ�ϵ����������������ѧ�������������������ѧ��������û���֪ʶ���ʵ�������������

 ����ѧ���ʱѧ����ϵ�д�
����ѧ���ʱѧ����ϵ�д� �ʰ�Ӣ��ͬ����ϰ��ϵ�д�
�ʰ�Ӣ��ͬ����ϰ��ϵ�д�| A�� | 28g | B�� | 56g | C�� | 14g | D�� | ������ |
| A�� | 18g D2O��18g H2O�к��е���������Ϊ10NA | |
| B�� | 2L 0.5 mol•L-1��������Һ�к��е�H+������Ϊ2NA | |
| C�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.2NA | |
| D�� | �ܱ�������2mol NO��1mol O2��ַ�Ӧ������ķ�����Ϊ2NA |
| A�� | ij���ȷ�Ӧ���Է����У���˸÷�Ӧ��������Ӧ | |
| B�� | ����Ũ�Ȼ�ʹ�ô��������������Ӱٷ���������ѧ��Ӧ���� | |
| C�� | ��ȼ����Ҫ�Ǽ�����ˮ�ڵ��¸�ѹ���γɵ�ˮ���ᄃ�壬��˿ɴ����ں��� | |
| D�� | ���ļ۵��ӵ����Ų�ʽΪ3d54s1������3d44s2����Ϊ�����ȶ� |
A CaO Na2O CuO CO2 B H2 N2 Cl2 Cu
C HCl HNO3 H2O H2SO4 D O2 NH3 NaCl KNO3
��1������������ʰ���1��Ҫ����࣬��д���еĿհף�
��1
| ��� | A | B | C | D |
| ����� | ���������� | ǿ�� | ||
| �����Ϸ���������� | Cu | O2 |
��3��ijͭ������ʴ�������γ�һ��ͭ�̣�����ͭ�������ͭ�̣�Ӧ��ѡ��A��
A��ϡHCl B��HNO3 C��ŨH2SO4
����ѡ������ʣ�д����������ͭ�̷�Ӧ�����ӷ���ʽ��Cu2��OH��2CO3+4H+=2Cu2++3H2O+CO2����
| ѡ�� | �������ʵ | ���� |
| A | ���ȵĴ�����Һϴȥ���� | Na2CO3��ֱ�Ӻ����۷�Ӧ |
| B | Ư���ڿ����о��ñ��� | Ư���е�CaCl2������е�CO2��Ӧ����CaCO3 |
| C | ʩ��ʱ����ľ�ң���Ч�ɷ�ΪK2CO3��������NH4Cl���ʹ�� | K2CO3��NH4Cl�������з�Ӧ���ɰ����ή�ͷ�Ч |
| D | �����ڳ�ʪ�Ŀ������������� | �����ڿ����з��������ⸯʴ |
| A�� | A | B�� | B | C�� | C | D�� | D |