��Ŀ����
20���塢����Ҫ�����ں�ˮ�У��С�����Ԫ�ء������ƣ���ˮ�еĵ⸻���ں����У��ҹ����������������һ������ʳ���⣬���������Ƶ⣬��ȡ���;��������ʾ���ɺ���$\stackrel{����}{��}$������$\stackrel{ˮ}{��}$ˮ��Һ$\stackrel{����}{��}$$\stackrel{A}{��}$$\stackrel{B}{��}$���ⵥ��
�����й���������ȷ���ǣ�������
| A�� | ��Ԫ���ں�ˮ��û������̬���� | |
| B�� | ����ȡ�Ĺ������õ�������������H2O2 | |
| C�� | ����A����ȡ | |
| D�� | B���ᴿ�������������� |
���� �ɺ������յõ�����������ˮ���˵õ��������ӵ�ˮ��Һ��������������������Ϊ�ⵥ�ʣ�������ȡ����ȡ��Һ������õ��ⵥ�ʣ�
A����ˮ�еĵ�Ԫ���ǻ���̬��
B�����������������������������Ϊ�ⵥ�ʣ�������Ⱦ��
C������������֪������AΪ��ȡ������ȡ��Һ��õ����ⵥ�ʵ��л�����
D��ʵ�ֵⵥ�ʺ���ȡ���ķ�����Ը��ݵ������������������У�
��� �⣺�ɺ������յõ�����������ˮ���˵õ��������ӵ�ˮ��Һ��������������������Ϊ�ⵥ�ʣ�������ȡ����ȡ��Һ������õ��ⵥ�ʣ�
A����ˮ�еĵ�Ԫ���ǻ���̬������������̬����A��ȷ��
B����ȡ�Ĺ������õ�������������H2O2�����������������������������Ϊ�ⵥ�ʣ�������Ⱦ����B��ȷ��
C������������֪������AΪ��ȡ������ȡ��Һ��õ����ⵥ�ʵ��л������������Ԫ�أ���C��ȷ��
D������������B����ʱ����ʹ������������D����
��ѡD��
���� ���⿼��ѧ����Ԫ�صĴ����Լ�����ᴿ֪ʶ�����Ը�����ѧ֪ʶ���лش��ѶȲ���

��ϰ��ϵ�д�
 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д� ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�
�����Ŀ
6����FeCl3��CuCl2�����Һ�м���һ���������ۺ�ͭ�۳�ַ�Ӧ������Ӧ�����������ʣ�࣬������˵���в���ȷ���ǣ�������
| A�� | �����п϶���ͭ | B�� | �����п������� | ||
| C�� | ��Һ�п϶�������Fe3+ | D�� | ��Һ�п϶�������Cu2+ |
15���±�ΪԪ�����ڱ���һ���֣���ش��й����⣺
��1���ٺ͢ݵ�Ԫ�ط�����N��Si��
��2����������õĽ�����K���ǽ�������ǿ��Ԫ����F������дԪ�ط��ţ�
��3���������γ��������������Ԫ����Al����дԪ�ط��ţ����ֱ�д����Ԫ�ص�����������ޢ�����������ˮ���ﷴӦ�Ļ�ѧ����ʽ��2Al��OH��3+3H2SO4=Al2��SO4��3+6H2O��Al��OH��3+KOH=KAlO2+2H2O��
��4���ߺ͢�Ԫ���γɵ��ʵ������Ե�ǿ��ΪCl2��Br2�����û�ѧ���ű�ʾ��֤���ý��۵�һ��ʵ����ʵ�������ӷ���ʽ��ʾ��Cl2+2Br-=2Cl-+Br2��
| ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� | ||
| 4 | �� | �� |
��2����������õĽ�����K���ǽ�������ǿ��Ԫ����F������дԪ�ط��ţ�
��3���������γ��������������Ԫ����Al����дԪ�ط��ţ����ֱ�д����Ԫ�ص�����������ޢ�����������ˮ���ﷴӦ�Ļ�ѧ����ʽ��2Al��OH��3+3H2SO4=Al2��SO4��3+6H2O��Al��OH��3+KOH=KAlO2+2H2O��
��4���ߺ͢�Ԫ���γɵ��ʵ������Ե�ǿ��ΪCl2��Br2�����û�ѧ���ű�ʾ��֤���ý��۵�һ��ʵ����ʵ�������ӷ���ʽ��ʾ��Cl2+2Br-=2Cl-+Br2��
9��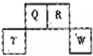 ������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ������T��������������������������ȣ�����˵����ȷ���ǣ�������
������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ������T��������������������������ȣ�����˵����ȷ���ǣ�������
 ������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ������T��������������������������ȣ�����˵����ȷ���ǣ�������
������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ������T��������������������������ȣ�����˵����ȷ���ǣ�������| A�� | W�����ڱ��е�λ���ǵ������ڣ���A�� | |
| B�� | ԭ�Ӱ뾶Q��R��T | |
| C�� | Wԭ�ӵõ�������ǿ��Qԭ�ӣ��ҿ��γɹ��ۻ�����QW2 | |
| D�� | ԭ��������R��1��Ԫ��ֻ��һ���⻯�� |
 ��
��
 ��E���Խ�����Ȫʵ�����Ҫԭ���ǣ���ΪNH3��������ˮ��
��E���Խ�����Ȫʵ�����Ҫԭ���ǣ���ΪNH3��������ˮ�� NH3��H2O+H+
NH3��H2O+H+ ��Bԭ��L��ĵ���������K���3����0.1mol C�����ܴ������û���2.24L��������״������ͬʱ���ĵ��Ӳ�ṹ�������ԭ�ӵĵ��Ӳ�ṹ��ͬ��D���ӵİ뾶��C���ӵ�С��D������B���ӵĵ��Ӳ�ṹ��ͬ��
��Bԭ��L��ĵ���������K���3����0.1mol C�����ܴ������û���2.24L��������״������ͬʱ���ĵ��Ӳ�ṹ�������ԭ�ӵĵ��Ӳ�ṹ��ͬ��D���ӵİ뾶��C���ӵ�С��D������B���ӵĵ��Ӳ�ṹ��ͬ��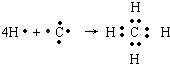 ��
��