ЬтФПФкШн
ЁОЬтФПЁПвЛЖЈЮТЖШЯТЃЌдквЛИіШнЛ§ЮЊ1LЕФУмБеШнЦїжаЃЌГфШы1molH2(g)КЭ1mol I2(g)ЃЌЗЂЩњЗДгІH2(g)ЃЋI2(g)![]() 2HI(g)ЃЌОГфЗжЗДгІДяЕНЦНКтКѓЃЌЩњГЩЕФHI(g)еМЦјЬхЬхЛ§ЕФ50%ЃЌИУЮТЖШЯТЃЌдкСэвЛИіШнЛ§ЮЊ2LЕФУмБеШнЦїжаГфШы1molHI(g)ЗЂЩњЗДгІHI(g)
2HI(g)ЃЌОГфЗжЗДгІДяЕНЦНКтКѓЃЌЩњГЩЕФHI(g)еМЦјЬхЬхЛ§ЕФ50%ЃЌИУЮТЖШЯТЃЌдкСэвЛИіШнЛ§ЮЊ2LЕФУмБеШнЦїжаГфШы1molHI(g)ЗЂЩњЗДгІHI(g)![]()
![]() H2(g)ЃЋ
H2(g)ЃЋ![]() I2(g)ЃЌдђЯТСаХаЖЯе§ШЗЕФЪЧ(ЁЁЁЁ)
I2(g)ЃЌдђЯТСаХаЖЯе§ШЗЕФЪЧ(ЁЁЁЁ)
A.КѓвЛЗДгІЕФЦНКтГЃЪ§ЮЊ1
B.КѓвЛЗДгІЕФЦНКтГЃЪ§ЮЊ0.5
C.КѓвЛЗДгІДяЕНЦНКтЪБЃЌH2ЕФЦНКтХЈЖШЮЊ0.25 molЁЄLЃ1
D.КѓвЛЗДгІДяЕНЦНКтЪБЃЌHI(g)ЕФЦНКтХЈЖШ0.5 molЁЄLЃ1
ЁОД№АИЁПB
ЁОНтЮіЁП
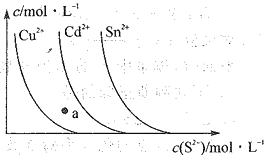
AКЭBЃЎвЛЖЈЮТЖШЯТЃЌНЋH2КЭI2Иї1molЕФЦјЬЌЛьКЯЮяГфШы1LЕФУмБеШнЦїжаЃЌЗЂЩњЗДгІH2(g)ЃЋI2(g)![]() 2HI(g)ЃЌДяЕНЦНКтКѓЩњГЩЕФHI(g)ЕФЬхЛ§ЗжЪ§ЮЊ50%ЃЌгЩгкИУЗДгІЧАКѓЦјЬхЗжзгЪ§ВЛБфЃЌЫљвдЦНКтЪБЦјЬхЕФзмЮяжЪЕФСПЮЊ2molЃЌдђHIЮЊ2molЁС50%=1molЃЌH2КЭI2ЕФЮяжЪЕФСПЯрЕШЃЌИїЮЊ0.5molЁЃЬхЛ§ЮЊ1LЃЌЙЪHIЁЂH2ЁЂI2ЕФХЈЖШКЭЮяжЪЕФСПЯрЕШЃЌЗжБ№ЮЊ1mol/LЁЂ0.5mol/LЁЂ0.5mol/LЃЌЫљвдH2(g)ЃЋI2(g)
2HI(g)ЃЌДяЕНЦНКтКѓЩњГЩЕФHI(g)ЕФЬхЛ§ЗжЪ§ЮЊ50%ЃЌгЩгкИУЗДгІЧАКѓЦјЬхЗжзгЪ§ВЛБфЃЌЫљвдЦНКтЪБЦјЬхЕФзмЮяжЪЕФСПЮЊ2molЃЌдђHIЮЊ2molЁС50%=1molЃЌH2КЭI2ЕФЮяжЪЕФСПЯрЕШЃЌИїЮЊ0.5molЁЃЬхЛ§ЮЊ1LЃЌЙЪHIЁЂH2ЁЂI2ЕФХЈЖШКЭЮяжЪЕФСПЯрЕШЃЌЗжБ№ЮЊ1mol/LЁЂ0.5mol/LЁЂ0.5mol/LЃЌЫљвдH2(g)ЃЋI2(g)![]() 2HI(g)ЕФЦНКтГЃЪ§ЮЊ
2HI(g)ЕФЦНКтГЃЪ§ЮЊ![]() =
=![]() =4ЁЃЮТЖШВЛБфЃЌЦНКтГЃЪ§ВЛБфЃЌЫљвд2HI(g)
=4ЁЃЮТЖШВЛБфЃЌЦНКтГЃЪ§ВЛБфЃЌЫљвд2HI(g) ![]() H2(g)ЃЋI2(g)ЕФЦНКтГЃЪ§ЮЊH2(g)ЃЋI2(g)
H2(g)ЃЋI2(g)ЕФЦНКтГЃЪ§ЮЊH2(g)ЃЋI2(g)![]() 2HI(g)ЕФЕЙЪ§ЃЌМД
2HI(g)ЕФЕЙЪ§ЃЌМД![]() ЃЌHI(g)
ЃЌHI(g)![]()
![]() H2(g)ЃЋ
H2(g)ЃЋ![]() I2(g)ЕФЦНКтГЃЪ§ЮЊ2HI(g)
I2(g)ЕФЦНКтГЃЪ§ЮЊ2HI(g) ![]() H2(g)ЃЋI2(g)ЕФ
H2(g)ЃЋI2(g)ЕФ![]() ДЮЗНБЖЃЌМД
ДЮЗНБЖЃЌМД![]() =0.5ЃЌЙЪAДэЮѓЃЌBе§ШЗЃЛ
=0.5ЃЌЙЪAДэЮѓЃЌBе§ШЗЃЛ
CЃЎ2LЕФУмБеШнЦїжаГфШы1molHI(g)ЗЂЩњЗДгІHI(g)![]()
![]() H2(g)ЃЋ
H2(g)ЃЋ![]() I2(g)ЃЌКЭЯђШнЛ§ЮЊ1LЕФУмБеШнЦїжаЃЌГфШы1molH2(g)КЭ1mol I2(g)ДяЕНЕШаЇЦНКтЃЌЦНКтЪБИїзщЗжЕФЬхЛ§ЗжЪ§ЯрЕШЃЌЫљвдКѓвЛЗДгІЦНКтКѓHIЕФЬхЛ§ЗжЪ§ЮЊ50%ЃЌМДЮяжЪЕФСПЮЊ1molЁС50%=0.5molЃЌH2КЭI2ЕФЮяжЪЕФСПЯрЕШЃЌИїЮЊ0.25molЁЃЬхЛ§ЮЊ2LЃЌЫљвдH2ЕФЦНКтХЈЖШЮЊ
I2(g)ЃЌКЭЯђШнЛ§ЮЊ1LЕФУмБеШнЦїжаЃЌГфШы1molH2(g)КЭ1mol I2(g)ДяЕНЕШаЇЦНКтЃЌЦНКтЪБИїзщЗжЕФЬхЛ§ЗжЪ§ЯрЕШЃЌЫљвдКѓвЛЗДгІЦНКтКѓHIЕФЬхЛ§ЗжЪ§ЮЊ50%ЃЌМДЮяжЪЕФСПЮЊ1molЁС50%=0.5molЃЌH2КЭI2ЕФЮяжЪЕФСПЯрЕШЃЌИїЮЊ0.25molЁЃЬхЛ§ЮЊ2LЃЌЫљвдH2ЕФЦНКтХЈЖШЮЊ![]() =0.125mol/LЃЌЙЪCДэЮѓЃЛ
=0.125mol/LЃЌЙЪCДэЮѓЃЛ
DЃЎИљОнвдЩЯМЦЫуКѓвЛЗДгІДяЕНЦНКтЪБЃЌHI(g)ЕФЦНКтХЈЖШЮЊ![]() =0.25mol/LЃЌЙЪDДэЮѓЃЛЙЪбЁBЁЃ
=0.25mol/LЃЌЙЪDДэЮѓЃЛЙЪбЁBЁЃ

 ЬьЬьЯђЩЯвЛБОКУОэЯЕСаД№АИ
ЬьЬьЯђЩЯвЛБОКУОэЯЕСаД№АИ аЁбЇЩњ10ЗжжггІгУЬтЯЕСаД№АИ
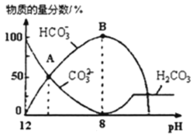
аЁбЇЩњ10ЗжжггІгУЬтЯЕСаД№АИЁОЬтФПЁПдквЛУмБеШнЦїжаЭЈШыAЁЂBСНжжЦјЬхЃЌдквЛЖЈЬѕМўЯТЗЂЩњЗДгІ2A(g)ЃЋB(g)![]() 2C(g) ІЄH<0ЃЌЕБЗДгІДяЕНЦНКтКѓЃЌИФБфвЛИіЬѕМў(x)ЃЌЯТБэжаСП(y)вЛЖЈЗћКЯЯТЭМжаЧњЯпЕФЪЧЃЈ ЃЉ
2C(g) ІЄH<0ЃЌЕБЗДгІДяЕНЦНКтКѓЃЌИФБфвЛИіЬѕМў(x)ЃЌЯТБэжаСП(y)вЛЖЈЗћКЯЯТЭМжаЧњЯпЕФЪЧЃЈ ЃЉ
x | y |
| |
A | ЮТЖШ | ЛьКЯЦјЬхЕФЦНОљЯрЖдЗжзгжЪСП | |
B | бЙЧП | AЕФАйЗжКЌСП | |
C | дйЭЈШыA | BЕФзЊЛЏТЪ | |
D | МгШыДпЛЏМС | AЕФжЪСПЗжЪ§ |
A.AB.BC.CD.D
ЁОЬтФПЁПбаОПН№ЪєгыЯѕЫсЕФЗДгІЃЌЪЕбщШчЯТЁЃ
ЪЕбщ (20C) | ЯжЯѓ |
Ђё | ЮоЩЋЦјЬх(гіПеЦјБфКьзиЩЋ)ЃЌШмвКБфЮЊРЖЩЋ |
Ђђ | 6mLЮоЩЋЦјЬх(ОМьВтЮЊH2)ЃЌШмвКМИКѕЮоЩЋ |
ЂХЂёжаВњЩњЕФЮоЩЋЦјЬхЪЧ______ЁЃ
ЂЦбаОПЂђжаЕФбѕЛЏМС
ЂйМзЭЌбЇШЯЮЊИУХЈЖШЕФЯѕЫсжаH+ЕФбѕЛЏадДѓгкNO3ЃЃЌЫљвдNO3ЃУЛгаЗЂЩњЗДгІЁЃввЭЌбЇвРОнЂёКЭЂђжЄУїСЫМзЕФЫЕЗЈВЛе§ШЗЃЌЦфЪЕбщжЄОнЪЧ______ЁЃ
![]() ввЭЌбЇЭЈЙ§ЗжЮіЃЌЭЦВтГіNO3ЃвВФмБЛЛЙдЃЌвРОнЪЧ______ЃЌНјЖјЫћЭЈЙ§ЪЕбщжЄЪЕИУШмвКжаКЌгаNH4+ЃЌЦфЪЕбщВйзїЪЧ______ЁЃ
ввЭЌбЇЭЈЙ§ЗжЮіЃЌЭЦВтГіNO3ЃвВФмБЛЛЙдЃЌвРОнЪЧ______ЃЌНјЖјЫћЭЈЙ§ЪЕбщжЄЪЕИУШмвКжаКЌгаNH4+ЃЌЦфЪЕбщВйзїЪЧ______ЁЃ
![]() ВЙШЋЂђжаNO3ЃБЛЛЙдЮЊ
ВЙШЋЂђжаNO3ЃБЛЛЙдЮЊ![]() ЕФЙ§ГЬЃКNO3Ѓ + ______eЃ + ______= NH4+ + ______H2O
ЕФЙ§ГЬЃКNO3Ѓ + ______eЃ + ______= NH4+ + ______H2O
ЂЧбаОПгАЯьВњЩњH2ЕФвђЫи
ЪЕбщ | ЯжЯѓ |
Ђѓ | 3.4m LЦјЬх(ОМьВтЮЊH2ЃЉЃЌШмвКТдДјЛЦЩЋ |
Ђє | 2.6m LЦјЬх(ОМьВтЮЊH2ЃЉЃЌЛЦЩЋШмвК |
Ђѕ | ЮоЩЋЦјЬх(гіПеЦјБфКьзиЩЋ)ЃЌЩюзиЩЋШмвК |
зЪСЯЃКFe[(NO)]2+дкШмвКжаГЪзиЩЋЁЃ ЂмЖдБШЂђЁЂЂѓЁЂЂєЃЌЮТЖШВЛЭЌЪБЪеМЏЕНЧтЦјЕФЬхЛ§ВЛЭЌЃЌдвђЪЧ______ЁЃ ЂнЂѕжаЮоЩЋЦјЬхЪЧЛьКЯЦјЬхЃЌдђвЛЖЈКЌгагіПеЦјБфКьзиЩЋЕФЦјЬхКЭ______ЁЃ ЂШИљОнЪЕбщЃЌН№ЪєгыЯѕЫсЗДгІЪБЃЌгАЯьЯѕЫсЕФЛЙдВњЮяВЛЭЌЕФвђЫига______ЁЃ