��Ŀ����
4����пͭ�Ͻ�ķ���Ϊԭ���Ʊ�����ͭ���壨CuSO4•5H2O���IJ��ֹ���������ͼ��ʾ��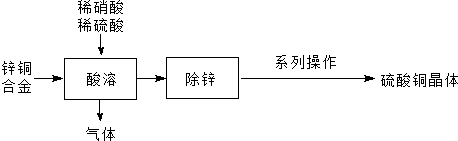
��1������ʱ��ͭת��ΪCu2+�����ӷ���ʽΪ3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��пͭ�Ͻ�Ҫ������Ƭ�ҷ�Ӧ����Ҫ���Ͻ��裬��Ŀ��������Ӵ��������ʹ��Ӧ��ֻ�ӿ췴Ӧ���ʣ���
��2��Ϊʹ���ܺ����Һ�в���NO3-�����ƻ���ϡ��Һʱ��Ӧ������������������ʵ���֮�Ȳ�С��3��2��
��3����ϵ�в�����Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����
��4���ⶨ��п�����Һ��Cu2+����Ũ�ȵķ���Ϊ��ȷ��ȡV1mL��п�����Һ����ƿ�У�����pH=3��4���������KI����ַ�Ӧ�μ�2�ε�����Һ���������μ�c mol/L Na2S2O3����Һ��ǡ����ȫ��Ӧ������Na2S2O3����ҺV2mL��
��֪��2Cu2++4I-�T2CuI��+I2 2S2O32-+I2�TS4O62-+2I-
�١�ǡ�÷�Ӧ��ʱ��Һ����ɫ�仯Ϊ��ɫ��Ϊ��ɫ��
��Cu2+���ӵ����ʵ���Ũ��Ϊ$\frac{c{V}_{2}}{{V}_{1}}$ mol/L����V1��V2��c��ʾ����
������п�����Һ���Ժ���NO3-���ӣ���ⶨ�����ƫ���ƫ����ƫС��������Ӱ�족����
���� �����������̿�֪��пͭ�Ͻ��������ᡢ�����л������������ԭΪ���������пͭ�����������ͭ���Ӻ�п���ӣ���ȥп���Ӻ������Һ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ɵ�����ͭ���壬
��1�����ݵ���غ��Ԫ���غ�д���ӷ���ʽ��һ���������ʣ�����ԽС�������Խ��ʹ��Ӧ����������Ҳ������Ӧ���ʣ�
��2��ʹ���ܺ����Һ�в���NO3-����ӦʹNO3-ȫ������������
��3�����ݴ���Һ�л��CuSO4•5H2O���壬����ͨ������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����������ͭ���壻
��4���١�ǡ�÷�Ӧ��ʱ����Һ�е�I2ǡ����Na2S2O3��ȫ��Ӧ��
�ڸ��ݷ�Ӧ�еĸ�����֮��Ĺ�ϵ�ɼ����Cu2+���ӵ����ʵ������ٸ���c=$\frac{n}{V}$����Ũ�ȣ�
��NO3-���Ӿ��������ԣ�����S2O32-��Ӧ��
��� �⣺�����������̿�֪��пͭ�Ͻ��������ᡢ�����л������������ԭΪ���������пͭ�����������ͭ���Ӻ�п���ӣ���ȥп���Ӻ������Һ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����ɵ�����ͭ���壬
��1������ʱ��ͭת��ΪCu2+�����ӷ���ʽΪ3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��һ���������ʣ�����ԽС�������Խ��ʹ��Ӧ����������Ҳ������Ӧ���ʣ�����пͭ�Ͻ�Ҫ������Ƭ�ҷ�Ӧ����Ҫ���Ͻ��裬��Ŀ��������Ӵ��������ʹ��Ӧ��ֻ�ӿ췴Ӧ���ʣ���
�ʴ�Ϊ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O������Ӵ��������ʹ��Ӧ��ֻ�ӿ췴Ӧ���ʣ���
��2��ʹ���ܺ����Һ�в���NO3-��ӦʹNO3-ȫ���������������ݷ�Ӧ3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��֪������������ʵ���Ϊ2molʱ����Ҫ����Ϊ3mol��������������������ʵ���֮�Ȳ�С��3��2��
�ʴ�Ϊ��3��2��
��3�����ݴ���Һ�л��CuSO4•5H2O���壬����ͨ������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����������ͭ���壬
�ʴ�Ϊ������Ũ������ȴ�ᾧ��
��4���١�ǡ�÷�Ӧ��ʱ����Һ�е�I2ǡ����Na2S2O3��ȫ��Ӧ����ʱ��Һ����ɫ�仯Ϊ��ɫ��Ϊ��ɫ��
�ʴ�Ϊ����ɫ��Ϊ��ɫ��
�ڸ��ݷ�Ӧ2Cu2++4I-�T2CuI��+I2��2S2O32-+I2�TS4O62-+2I-��֪��2Cu2+��I2��2S2O32-������Cu2+���ӵ����ʵ�����S2O32-�����ʵ�����ȣ�ΪcV2��10-3 mol������c=$\frac{n}{V}$��֪��Cu2+���ӵ����ʵ���Ũ��Ϊ$\frac{c{V}_{2}}{{V}_{1}}$mol/L��
�ʴ�Ϊ��$\frac{c{V}_{2}}{{V}_{1}}$��
��NO3-���Ӿ��������ԣ�����S2O32-��Ӧ��ʵ������ȥ��S2O32-�����ʵ�������V2ƫ����Cu2+���ӵ����ʵ���Ũ��Ҳƫ��
�ʴ�Ϊ��ƫ��
���� ���⿼�����ʵ��Ʊ��������IJⶨ��������ѧ��ʵ�������ͷ��������Ŀ��飬Ϊ�߿��������ͣ�ע�⣨4���⣬���ù�ϵʽ�������Ŀ�Ѷ��еȣ�

 �ݾ�ѵ������ϵ�д�
�ݾ�ѵ������ϵ�д� С����ȫ�ܼ��ϵ�д�
С����ȫ�ܼ��ϵ�д�| A�� | 80g CuO��Cu2S�Ļ���ﺬ��ͭԭ����һ��ΪNA | |
| B�� | ��⾫��ͭʱ������·��ת��NA�����ӣ������ܽ�32gͭ | |
| C�� | 1mol����1molCl2��ַ�Ӧ��ת�Ƶ�����Ϊ3NA | |
| D�� | ����FeI2����Һ��ͨ����������������1mol Fe2+������ʱ�÷�Ӧת�Ƶ��ӵ���Ŀ����ΪNA |
| A�� | ԭ������������Ϊ2��Ԫ��һ���������ڱ���A�� | |
| B�� | ����Ԫ��X��Y���γ�XY2�ͻ������X��Y��ԭ������֮�����Ϊ2��5 | |
| C�� | �Ȼ���ķе���廯��ķе�� | |
| D�� | Li��Na��K���ʵ��۵������� |
| A�� | Na+ NO3- SO42- | B�� | Ag+ NO3- Cl- | ||
| C�� | K+ H+ HCO3- | D�� | NH4+ Na+ OH- |
����˵����ȷ���ǣ�������
| A�� | �û������ͭ������Ϊ19.2 g | |
| B�� | ��û���ﷴӦ���ĵ�HNO3����Ϊ1.2 mol | |
| C�� | ����NaOH��Һ�������800 mL | |
| D�� | Cu2+������ȫ����Һ��c��NO3-��Ϊ0.7 mol/L |
| A�� | FeBr2��Һ��Cl2 | B�� | Ca��HCO3��2��Һ��KOH��Һ | ||
| C�� | NaHCO3��Һ��Al2��SO4��3��Һ | D�� | NaHSO4��Һ��Ba��OH��2��Һ |
| A�� | Na+��Ba2+��Cl-��SO42- | B�� | Na+��Ba2+��AlO2-��NO3- | ||
| C�� | NH4+��K+��Cl-��NO3- | D�� | Na+��K+��NO3-��SiO32- |
| A�� | H2SO4Ϊǿ�ᣬ�Ƴ�HClO4Ϊǿ�� | |
| B�� | Fe3Cl8���Ը�дΪFeCl2 •2FeCl3���Ƴ�Fe3I8���Ը�дΪFeI2 •2FeI3 | |
| C�� | NH3�ķе����PH3���Ƴ�CH4�е����SiH4 | |
| D�� | CO2 ͨ��Ba��NO3��2 ��Һ���������ɣ��Ƴ�SO2ͨ��Ba��NO3��2 ��Һ���������� |
| ʵ�鷽�� | ʵ������ |
| ����ɰֽ�����þ�����ˮ��Ӧ����ӦҺ�еμӷ�̪ | A������ˮ�棬�۳�С����ˮ���������ƶ���֮��ʧ����Һ��ɺ�ɫ |
| �������Ƶ�H2S������Һ�еμ����Ƶ���ˮ | B�������������壬���ڿ�����ȼ�գ���Һ���dz��ɫ |
| ��������з�̪��Һ����ˮ��Ӧ | C����Ӧ��ʮ�־��ң�������������ڿ�����ȼ�� |
| ��þ����2mol•L-1�����ᷴӦ | D�����ҷ�Ӧ����������������ڿ�����ȼ�� |
| ��������2mol•L-1�����ᷴӦ | E�����ɰ�ɫ��״�������̶�������ʧ |
| �����Ȼ�����Һ�еμ��������� | F�����ɵ���ɫ���� |
��1��ʵ��Ŀ�ģ���֤Na��Mg��Al��S��ClԪ�ش����ҽ��������μ������ǽ�����������ǿ�Ĺ��ɣ�
��2��ʵ����Ʒ���Թܡ��ƾ��ơ�ɰֽ�����ӡ�С������ͷ�ιܣ�
��3��ʵ�����ݣ�
| ʵ�鷽�� ������ţ� | ʵ������ ������ţ� | �йػ�ѧ����ʽ |
��5���������ʽṹ���ۼ�˵�������������۵�ԭ��ͬ����Ԫ�أ���ԭ����������ԭ�Ӻ˶Ժ�����ӵ�������������ǿ��ԭ��ʧȥ���ӵ������������õ�����������ǿ��