��Ŀ����
��11 �֣���ѧ��һֱ�����о����¡���ѹ�¡��˹��̵������·���������ʵ�鱨�����ڳ��¡���ѹ�����������£�N2�ڴ�������������Fe2O3��TiO2��������ˮ������Ӧ�����ɵ���Ҫ����ΪNH3����Ӧ�Ļ�ѧ����ʽ���£�N2(g)+ 3H2O(l)  2NH3(g)+ O2(g)���ش��������⣺
2NH3(g)+ O2(g)���ش��������⣺
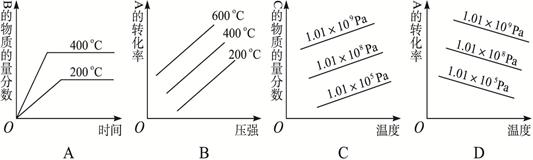
��1����һ���о�NH3���������¶ȵĹ�ϵ������ʵ�����ݼ��±������ա�N2ѹ��1.0��105 Pa����Ӧʱ��3 h������÷�Ӧ������ӦΪ ��Ӧ������ȡ����ȡ���
��2����Ŀǰ�㷺ʹ�õĹ�ҵ�ϳɰ�������ȣ��÷����й̵���Ӧ�������������������䷴Ӧ����������NH3�������Ľ��飺���� ��
��3���ϳɰ���ҵ��ԭ����N2�ɴӿ����з���õ���H2���ü����ڸ�������ˮ������Ӧ�Ƶá������ڸ�������ˮ������Ӧ��Ӧ����ʽΪ��CH4(g)��H2O(g)��CO(g)��3H2(g)���������ʵ�ȼ������
�����£�
H2(g) ����H =��285.8 kJ?mol��1��
CO(g) ����H =��283.0 kJ?mol��1��
CH4(g) ����H =��890.3 kJ?mol��1��
��֪1mol H2O(g)ת��Ϊ1mol H2O(l)ʱ�ų�44.0 kJ������д��CH4��H2O�ڸ����·�Ӧ���Ȼ�ѧ����ʽ__________________________________��
��4����������Ѱ���ʺϵĴ����͵缫���ϣ���N2��H2Ϊ�缫��Ӧ���HCl����NH4ClΪ�������Һ�Ƴ�����ȼ�ϵ�أ���д���õ缫��������Ӧʽ
��5�����ɵ�NH3���������̬���ʣ���(NH4)2SO4��NH4Cl����Щ������ �ԣ�ԭ���ǣ������ӷ���ʽ��ʾ��___________________________��ʹ��ʱ������________________���ʺ�ʩ��
 2NH3(g)+ O2(g)���ش��������⣺
2NH3(g)+ O2(g)���ش��������⣺��1����һ���о�NH3���������¶ȵĹ�ϵ������ʵ�����ݼ��±������ա�N2ѹ��1.0��105 Pa����Ӧʱ��3 h������÷�Ӧ������ӦΪ ��Ӧ������ȡ����ȡ���
| T/K | 303 | 313 | 323 |
| NH3������/��10-6 mol�� | 4.8 | 5.9 | 6.0 |
��3���ϳɰ���ҵ��ԭ����N2�ɴӿ����з���õ���H2���ü����ڸ�������ˮ������Ӧ�Ƶá������ڸ�������ˮ������Ӧ��Ӧ����ʽΪ��CH4(g)��H2O(g)��CO(g)��3H2(g)���������ʵ�ȼ������
�����£�
H2(g) ����H =��285.8 kJ?mol��1��
CO(g) ����H =��283.0 kJ?mol��1��
CH4(g) ����H =��890.3 kJ?mol��1��
��֪1mol H2O(g)ת��Ϊ1mol H2O(l)ʱ�ų�44.0 kJ������д��CH4��H2O�ڸ����·�Ӧ���Ȼ�ѧ����ʽ__________________________________��
��4����������Ѱ���ʺϵĴ����͵缫���ϣ���N2��H2Ϊ�缫��Ӧ���HCl����NH4ClΪ�������Һ�Ƴ�����ȼ�ϵ�أ���д���õ缫��������Ӧʽ
��5�����ɵ�NH3���������̬���ʣ���(NH4)2SO4��NH4Cl����Щ������ �ԣ�ԭ���ǣ������ӷ���ʽ��ʾ��___________________________��ʹ��ʱ������________________���ʺ�ʩ��
�� 11 �֣�
��1������
��2�������¶�. ����N2��Ũ��
��3��CH4(g)��H2O(g)��CO(g)��3H2(g) �� H����206.1 kJ?mol��1
��4�� N2+8H+ + 6e�� =" 2" NH4+
��5������ NH4++H2O NH3 H2O+H+ ����
NH3 H2O+H+ ����
��1������
��2�������¶�. ����N2��Ũ��
��3��CH4(g)��H2O(g)��CO(g)��3H2(g) �� H����206.1 kJ?mol��1
��4�� N2+8H+ + 6e�� =" 2" NH4+
��5������ NH4++H2O
 NH3 H2O+H+ ����
NH3 H2O+H+ ������

��ϰ��ϵ�д�
 ͬ��������ϰϵ�д�
ͬ��������ϰϵ�д�
�����Ŀ
 H2O(l) �Ħ�H =" 40.69" kJ��mol-1
H2O(l) �Ħ�H =" 40.69" kJ��mol-1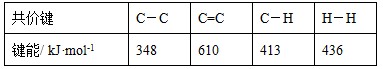
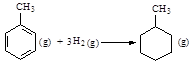 �Ħ�HΪ��384 kJ��mol-1
�Ħ�HΪ��384 kJ��mol-1 2OH?+H2��+Cl2��
2OH?+H2��+Cl2��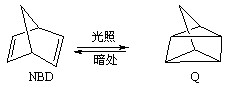 �����ø÷�Ӧ��������̫���ܣ���NBD�����ܱ�Q�����ܸ�
�����ø÷�Ӧ��������̫���ܣ���NBD�����ܱ�Q�����ܸ� 2NH3(g)+ 3/2O2(g)����H=+630kJ��mol-1
2NH3(g)+ 3/2O2(g)����H=+630kJ��mol-1 ��g��+
��g��+ 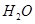 ��g��=
��g��=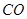 ��g��+
��g��+ ��g��
��g�� 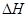 = +
= +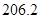
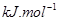
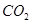 ��g��=
��g��=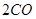 ��g��+
��g��+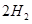 ��g��
��g��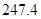
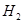 ��g�����Ȼ�ѧ����ʽΪ ��
��g�����Ȼ�ѧ����ʽΪ ��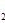 O���ȷֽ�Ҳ�ɵõ�H
O���ȷֽ�Ҳ�ɵõ�H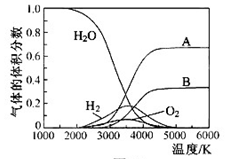
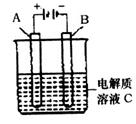
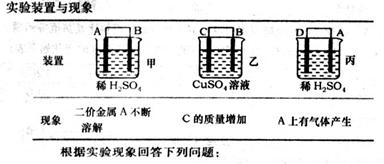
 CO2+NO ��H =" a" kJ/mol�ﵽƽ������¶ȣ�����������ɫ��dz�������ж���ȷ����
CO2+NO ��H =" a" kJ/mol�ﵽƽ������¶ȣ�����������ɫ��dz�������ж���ȷ����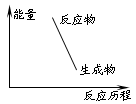
 2C(g)+Q (Q��0)������ͼ����ȷ���� ?
2C(g)+Q (Q��0)������ͼ����ȷ���� ?