��Ŀ����
4����֪��һ���������з�ӦA��4HCl��g��+O2��g���T2Cl2��g��+2H2O��g����ӦA�У�4mol HCI���������ų�115.6kJ������
��ӦB��

ͨ����������ijЩ�����л���ʱ�����������HC1��
���÷�ӦA����ʵ���ȵ�ѭ�����ã�
�ٷ�ӦA���漰�Ļ�ѧ�����У����зǼ��Լ����У�O2��Cl2��
��������������H2O��
�ڷ�ӦA���Ȼ�ѧ����ʽ��4HC��g��l+O2��g��$\frac{\underline{\;CuO/CuCl_{2}\;}}{400��}$ 2Cl2��g��+2H2O ��g����H=-115.6kJ•mol-1��
�۶Ͽ�1mol H-O ����Ͽ� 1mol H-Cl �������������ԼΪ31.9KJ����H2O ��H-O ����HCl��H-Cl�����ǿ����������ǿ��
���� ��ͬ�ַǽ���Ԫ��֮���γɷǼ��Թ��ۼ�����ͬ�ǽ���Ԫ��֮���γɼ��Թ��ۼ������ݵ�����������ԭ�Ӱ뾶С���縺��ֵ�������γɵĹ��ۼ�����ǿ�������γ����������
�ڷ�ӦA�У�4mol HCl���������ų�115.6kJ�������������Ȼ�ѧ����ʽ��д��������ע���ʾۼ�״̬�Ͷ�Ӧ�ʱ�д����
�۷�Ӧ�ȡ�H=��Ӧ���ܼ���-��������ܼ��ܣ��ݴ˼���H-O����H-Cl���ļ��ܲ��������Ͽ�1mol H-O����Ͽ�1mol H-Cl�����������
��� �⣺��O2����ԭ�Ӻ���ԭ��֮���ԷǼ��Թ��ۼ���ϣ�HCl����ԭ�Ӻ���ԭ��֮���Լ��Թ��ۼ���ϣ�Cl2����ԭ�Ӻ���ԭ��֮���ԷǼ��Թ��ۼ���ϣ�H2O����ԭ�Ӻ���ԭ��֮���Լ��Թ��ۼ���ϣ����������������⻯�����γ����������HCl��O2��Cl2��H2O���������γ��������H2O��
�ʴ�Ϊ��O2��Cl2��H2O��
�����ݷ�ӦA�У�4mol HCl���������ų�115.6kJ����������Ӧ���Ȼ�ѧ����ʽΪ��4HC��g��l+O2��g��$\frac{\underline{\;CuO/CuCl_{2}\;}}{400��}$ 2Cl2��g��+2H2O ��g����H=-115.6KJ/mol��
�ʴ�Ϊ��4HC��g��l+O2��g��$\frac{\underline{\;CuO/CuCl_{2}\;}}{400��}$ 2Cl2��g��+2H2O ��g����H=-115.6KJ/mol��
��E��H-O����E��HCl���ֱ��ʾH-O���ܡ�H-Cl���ܣ�
��ӦA�У�4mol HCl���������ų�115.6kJ����������Ӧ�ȡ�H=��Ӧ���ܼ���-��������ܼ��ܣ��ʣ�
4��E��H-Cl��+498kJ/mol-[2��243kJ/mol+4��E��H-O��]=-115.6kJ/mol��
�����ã�4E��H-Cl��-4E��H-O��=-127.6kJ/mol����E��H-O��-E��HCl��=31.9kJ/mol��
�ʶϿ�1mol H-O����Ͽ�1mol H-Cl�������������ԼΪ31.9kJ/mol��1mol=31.9kJ������H2O��H-O ����HCl��H-Cl��ǿ��
�ʴ�Ϊ��31.9��ǿ��
���� ���⿼���˻�ѧ�����ԡ����ܺ��ʱ��ϵ���㣬ͼ������жϡ��Ȼ�ѧ����ʽ��д��֪ʶ��ע���ʱ���ڷ�Ӧ��ļ��ܺͼ�ȥ������ļ��ܺͣ���Ŀ�Ѷ��еȣ�

 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�| A�� | 25% | B�� | 50% | C�� | 75% | D�� | ������ |
| A�� | ��ͭ˿����Ũ�����в����ȣ���Ӧ���ټ���ˮ���۲�����ͭ��Һ����ɫ | |
| B�� | ��ʢ��20g���ǵ��ձ��м��뼸��ˮ��������ȣ��ټ�������Ũ���ᣬѸ�ٽ��裬̽��Ũ�������ˮ�� | |
| C�� | �Ʊ�����������ʱ��������������Һ�еμ�����������Һ���ӱ߽��裬�����Ƶð�ɫ������������ | |
| D�� | ��������ͭ��[Cu2��OH��2C03]��ͭ�����������н��ݣ���ȥͭ�� |
| A�� | NO2������� | B�� | ʣ������֮һ���һ���������� | ||
| C�� | ��Һ��������ƿ | D�� | ����������һ������ |
| A�� | Zn��s��+CuSO4��aq���TZnSO4��aq��+Cu��s����H=-216 kJ•mol-1����Ӧ������������������������ | |
| B�� | ��һ�������£�A�TB��H��0��˵��A���ʱ�B�����ȶ� | |
| C�� | 101 kPaʱ��2H2��g��+O2��g���T2H2O��l����H=-571.6 kJ•mol-1����H2��ȼ����Ϊ571.6 kJ•mol-1 | |
| D�� | H+ ��aq��+OH- ��aq���TH2O��l����H=-57.3 kJ•mol-1����1 mol NaOH����Һ�뺬0.5 mol H2SO4��Ũ�����Ϻ�ų�57.3 kJ������ |
| A�� | ̼���� | B�� | ������ | C�� | ʯ��ˮ | D�� | �Ȼ�ͭ |
| A�� | ��״���£�22.4 L N2�� H2 ������к� NA��ԭ�� | |
| B�� | 3 mol ���� Fe ��ȫת��Ϊ Fe3O4��ʧȥ 8NA������ | |
| C�� | 2 L 0.5 mol•L-1�������Һ�У����� NA�������� | |
| D�� | ��״���£�2128 mL ��ȩ���� 2.85NA������ |

 ��E�ĵ���ʽ
��E�ĵ���ʽ ��y�ĽṹʽH-Cl��
��y�ĽṹʽH-Cl�� ����Ӧ�ڵĻ�ѧ����ʽΪ3Fe+4H2O��g��$\frac{\underline{\;����\;}}{\;}$Fe3O4+4H2��Ӧ�ݵ����ӷ���ʽΪ2Fe3++Fe=3Fe2+
����Ӧ�ڵĻ�ѧ����ʽΪ3Fe+4H2O��g��$\frac{\underline{\;����\;}}{\;}$Fe3O4+4H2��Ӧ�ݵ����ӷ���ʽΪ2Fe3++Fe=3Fe2+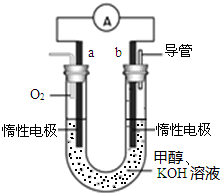 ��1��P2O5�Ƿ������Ը�������������岻����Ũ����������P2O5�������b
��1��P2O5�Ƿ������Ը�������������岻����Ũ����������P2O5�������b
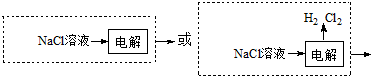 ��
��