��Ŀ����
9����ҵ�ϳ�����������ʢװ��Ũ���ᣮΪ�о����ʲ�������Ũ����ķ�Ӧ��ijѧϰС�����������̽�������1������ȥ�������������������̼�ظ֣�������Ũ�����У�10���Ӻ���������ͭ��Һ�У�Ƭ�̺�ȡ���۲죬�������������Ա仯����ԭ���dz�����Ũ����ʹ���ۻ���
��2������ȡ����6.0g����15.0mLŨ�����У����ȣ���ַ�Ӧ��õ���ҺX���ռ�������Y��
�ټ�ͬѧ��ΪX�г�Fe3+����ܺ���Fe2+����Ҫȷ�����е�Fe2+��Ӧѡ�ã�ѡ����ţ�d��
a��KSCN��Һ����ˮ b��NaOH��Һ c��Ũ��ˮ d������KMnO4��Һ
����ͬѧȡ336mL����״��������Yͨ��������ˮ�У�������Ӧ��SO2+Br2+2H2O�T2HBr+H2SO4��Ȼ���������BaCl2��Һ�����ʵ�������ø������2.33g���ɴ���֪����Y��SO2���������Ϊ66.7%��
��������ʵ����SO2��������Ľ������ͬѧ��Ϊ����Y�л����ܺ���H2��Q���壮Ϊ�����������̽��ʵ��װ�ã�ͼ�мг�����ʡ�ԣ���
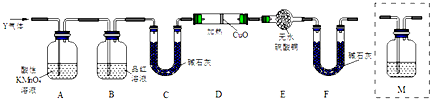
��3��װ��B���Լ��������Ǽ�����������Ƿ������
��4����Ϊ����Y�л�����Q��������C+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2��+2SO2��+2H2O���û�ѧ����ʽ��ʾ����
��5��Ϊȷ��Q�Ĵ��ڣ�����װ��������M��c��ѡ����ţ���
a��A֮ǰ b��A-B�� c��B-C�� d��C-D��
��6���������Y�к���H2��Ԥ��ʵ������Ӧ��D�к�ɫ��ĩ��죬E�а�ɫ��ĩ������
���� ��1������Ũ�����ǿ�����Խ��
��2���ٶ��������Ӿ���ǿ�Ļ�ԭ�ԣ��ܹ�ʹ���Եĸ��������ɫ������Fe2+��������+2������������ʹ�ữ�ĸ��������ɫ�����飻
�����ɵ�SO2���л�ԭ�ԣ�ͨ��������ˮ�У�����SO2+Br2+2H2O=2HBr+H2SO4��������Ԫ�ص��غ㣬��Ϲ�ϵʽ��SO2��BaSO4���SO2�����������
��3������������ʹƷ����Һ��ɫ���Ӷ�������������Ƿ������
��4�����������£�C��Ũ���ᷴӦ���ɶ�����̼��
��5����ȷ�������ڶ������������£�������ͨ�����ʯ��ˮ�����Ƿ��ж�����̼��
��6���������л�ԭ�ԣ��ܻ�ԭ�ԣ��ܻ�ԭ��ɫ������ͭ���ɺ�ɫ��ͭ���ʣ�ͬʱ����ˮ��ˮ��ʹ��ˮ����ͭ����ɫ��
��� �⣺��1��Ũ�������ǿ�������ԣ������������������ۻ����γ���������Ĥ��ֹ��Ӧ���У�
�ʴ�Ϊ��������Ũ����ʹ���ۻ���
��2����a����Һ�к������������ӣ�������������Ժ�ɫ���Զ��������Ӽ�����ɸ��ţ���a��ѡ��
b����Һ�к������������ӣ���NaOH��Ӧ���ɺ��ɫ�������������Ӻ�NaOH��Ӧ���ɰ�ɫ�����������ȶ�������ת��Ϊ����ɫ����ת��Ϊ���ɫ���������������Ƿ����������ӣ�����Ũ��ˮ���������ɫ��������b��ѡ��
c���������Ӻ�Ũ��ˮ��Ӧ���ɰ�ɫ�����������ȶ�������ת��Ϊ����ɫ����ת��Ϊ���ɫ�����������Ӻ�Ũ��ˮ��Ӧ���ɺ��ɫ���������������Ƿ����������ӣ�����Ũ��ˮ���������ɫ��������c��ѡ��
d�����������Ӿ���ǿ�Ļ�ԭ�ԣ��ܹ�ʹ���Եĸ��������ɫ�����������Ӳ����л�ԭ�ԣ�����ʹ���������ɫ�����Կ�������������������ӣ���dѡ��
��ѡ��d��
��SO2���л�ԭ�ԣ�ͨ��������ˮ�У�����SO2+Br2+2H2O=2HBr+H2SO4��
n��������壩=$\frac{0.336L}{22.4L/mol}$=0.015mol��
SO2 ��BaSO4
1mol 233g
n 2.33g
n=0.01mol��
�� SO2�����������$\frac{0.01}{0.015}$��100%=66.7%��
�ʴ�Ϊ��66.7%��
��3��A��ȥ����������������ʹƷ����Һ��ɫ������B���Լ���A���Ƿ���ȫ��ȥ��������
�ʴ�Ϊ������SO2�Ƿ������
��4�����������£�C��Ũ���ᷴӦ���ɶ�����̼������Q�Ƕ�����̼����Ӧ����ʽΪC+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$2SO2��+CO2��+2H2O��
�ʴ�Ϊ��C+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$2SO2��+CO2��+2H2O��
��5��ʵ�����ó���ʯ��ˮ���������̼������������Ҳ��ʹ����ʯ��ˮ����ǣ�Ҫ���������̼Ӧ���ų���������ĸ��ţ�������ȷ�������ڶ������������£�������ͨ�����ʯ��ˮ�����Ƿ��ж�����̼���ü���װ��Ӧ����B-C֮�䣬
��ѡc��
��6���������л�ԭ�ԣ��ܻ�ԭ�ԣ��ܻ�ԭ��ɫ������ͭ���ɺ�ɫ��ͭ���ʣ�ͬʱ����ˮ��ˮ��ʹ��ˮ����ͭ����ɫ�����Ǽ���ˮ��������Ӧ���������D������ͭ��죬E����ˮ����ͭ��������֤������������
�ʴ�Ϊ��D�к�ɫ��ĩ��죬E�а�ɫ��ĩ������
���� �����ۺϿ���Ԫ�ػ�����֪ʶ����������ʵ�鷽����ƣ���Ϥ������ʵ������ǽ���ؼ���ע����������ӡ����������ӵļ��鷽������Ŀ�Ѷ��еȣ�

| A�� | ��ȡ10.6gNaCO3������100mL����ƿ�У���ˮ�ܽ⣬���� | |
| B�� | ��ȡ10.6gNaCO3������100mL����ˮ�����裬�ܽ� | |
| C�� | ��Һʱδ�ò�����������ֱ�ӵ�������ƿ�� | |
| D�� | ���ݺ�����ת��ҡ�� |
| A�� | HR | B�� | H2R | C�� | RH4 | D�� | RH3 |
| ʵ����ʵ | ���� | |
| A | Cl2��ˮ��Һ���Ե��� | Cl2�ǵ���� |
| B | SO2ͨ�����ᱵ��Һ���ְ�ɫ���� | BaSO3������ǿ�� |
| C | NaHCO3��Һ��NaAlO2��Һ��ϲ�����ɫ���� | ���ԣ�HCO3-��Al��OH��3 |
| D | �����°�����ȼ��N2���ڷŵ�ʱ����O2��Ӧ | �ǽ����ԣ�P��N |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��ԭ�ӵ�ԭ�ӽṹʾ��ͼ�� | |
| B�� | C2H4�Ľṹ��ʽ��CH2CH2 | |
| C�� | ԭ�Ӻ�����10�����ӵ���ԭ�ӣ�${\;}_{8}^{18}$O | |
| D�� | CH4 ���ӵĽṹģ�ͣ� |
| A�� | CH3CH=C��CH3�� CH3 3-��-2-��ϩ | |
| B�� | CH3CH��CH3��CH��Cl��CH3 3-��-2-�ȶ��� | |
| C�� | CH3CH��OH��CH2CH3 2-�ǻ����� | |
| D�� | CH3CH��C2H5��CH2CH2CH3 2-�һ����� |
| A�� | 2g H2���е���ԭ����ĿΪNA | |
| B�� | ���³�ѹ�£�22.4 LO2���еķ�����ĿΪNA | |
| C�� | 1 molZnת��ΪZn2+ʧȥ�ĵ�����ĿΪNA | |
| D�� | 1 L 1mol•L-1KOH��Һ�к��еļ�������ĿΪNA |
| ѡ�� | ���� | ���� | �����Լ� | ��Ӧ��������ӷ��� |
| A | �� | ���� | �ռ���Һ | ��Һ |
| B | �� | �� | ϡ���� | ���� |
| C | ���� | �Ȼ��� | ����ʳ��ˮ | ϴ�� |
| D | ���� | ��ϩ | ���� | ���������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
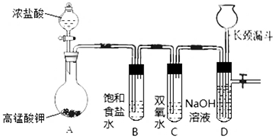 Cl2��H2O2�Ǹ��н����������������������������Cl2��������ǿ��H2O2���ܽ�H2O2������Ϊ����֤�ý��ۣ�ѧ���������ͼ��ʾ��ʵ��װ�ý���ʵ�飨�г�װ����ȥ������ش��������⣺
Cl2��H2O2�Ǹ��н����������������������������Cl2��������ǿ��H2O2���ܽ�H2O2������Ϊ����֤�ý��ۣ�ѧ���������ͼ��ʾ��ʵ��װ�ý���ʵ�飨�г�װ����ȥ������ش��������⣺ HCl+HClO��2HClO$\frac{\underline{\;����\;}}{\;}$2HCl+O2�������÷���ʽ�ش𣩣��������ɿ��Բ��öԱ�ʵ������
HCl+HClO��2HClO$\frac{\underline{\;����\;}}{\;}$2HCl+O2�������÷���ʽ�ش𣩣��������ɿ��Բ��öԱ�ʵ������