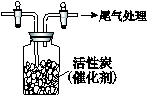��Ŀ����
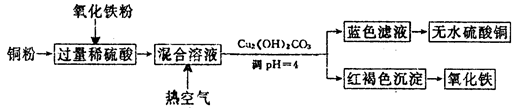
��14�֣��������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��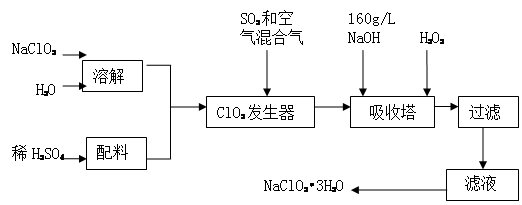
��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2?3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 gNaOH��������ˮ������Һ�����Ϊ1L��
��1�� 160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ������ ������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ�
b��ϡ��ClO2�Է�ֹ��ը��
c����NaClO3������ClO2
��3���������ڵķ�Ӧ�Ļ�ѧ����ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH�Ƿ�����ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2
�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6�� ����Һ�еõ�NaClO2?3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������ c������ d������ e����ȴ�ᾧ
Ҫ�õ�������NaClO2?3H2O���������еIJ��������������� ��������������ƣ�
��1�� 4mol/L��2�֣� ��2�� b��2�֣�
��3��2NaOH+2ClO2+H2O2 ��2 NaClO2+2H2O+O2��2�֣�����ֹH2O2�ֽ⣨2�֣�
��4�������ⶨ����������Һ��pH��2�֣�
��5�� a ��1�֣� ��6�� b��e��d��ȫ��2�֣��д����÷֣����ؽᾧ��1�֣�
���������������1��Ũ��g/L��ʾ1L��Һ���������������Ķ��٣�160g/LNaOH��Һ��ʾ1L����������Һ����160gNaOH������Һ���Ϊ1L����160gNaOH�����ʵ���Ϊ160g��40g/mo=4mol�����Ը���Һ�������Ƶ����ʵ���Ũ��c��NaOH��=4mol��1L==4mol?L?1��
��2������Ϣ�ڿ�֪����ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10%���°�ȫ���������й������������Ӧ��ϡ��ClO2�Է�ֹ��ը����ѡ��b��
��3������������Ϣ��֪��������������NaClO2������һ����ClO2��NaClO2�����ϼ۽��ͣ�����ԭ����H2O2�ض�����������������������Ӧ����ʽΪ2NaOH+2ClO2+H2O2=2 NaClO2+2H2O+O2��H2O2���ȶ����¶ȹ��ߣ�H2O2���ֽ⣬���������¶Ȳ��ܳ���20�棬��Ŀ���Ƿ�ֹH2O2�ֽ⡣
��4��NaOH��������Һ�ʼ��ԣ������ǵ�����Һ��ǿ�����ԣ�ѡ��ָʾ����pH��ֽ���������������ɫ���⣬����������Ҫ����pH�������ⶨ��ҺpH��
��5����ԭ��Ҫ���У���ԭ��̫ǿ���ὫClO2��ԭΪ���ͼ�̬���Ӱ��NaClO2������������������ᴿ�������Լ����ܸ��ź���������Na2O2����ˮ�൱��H2O2��Na2S��FeCl2��ԭ�Խ�ǿ����������NaClO2����Ƚ����ѣ���ѡ��a��
��6������Һ�еõ����ᾧˮ�ľ��壬ֻ�ܲ�ȡ������Ũ������ȴ�ᾧ������ͨ�����˵õ��־��壮���Բ���˳��Ϊbed���õ��Ĵ־��徭���ؽᾧ�ɵõ����ȸ��ߵľ��塣
���㣺���⿼�黯ѧ���̵ķ��������ʵ���Ũ�ȡ�����������

 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д���16�֣������������ҹ�����Դ������ռ�ϴ���أ������ŷų���SO2�����һϵ�л�������̬���⣬ֱ���ŷź�SO2���������γ����꣬Σ��������
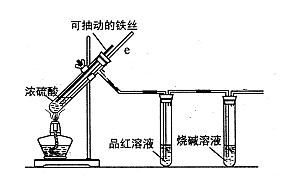
��1���û�ѧ����ʽ��ʾSO2�γ�����������ķ�Ӧ�� ��2��
��2����ҵ����Na2SO3��Һ���������е�SO2��������ͨ��1.0 mol��L-1��Na2SO3��Һ����ҺpH���ϼ�С������ҺpHԼΪ6ʱ������SO2�����������½���Ӧ�������ռ���
�� ��ʱ��Һ��c(SO32�C)��Ũ����0.2 mol��L-1������Һ��c(HSO3�C)��_______mol?L-1��
�� ��pHԼΪ6�����ռ���ͨ��������O2���ɽ����е�NaHSO3ת��Ϊ�������ʣ���Ӧ�Ļ�ѧ����ʽ�� ��2��
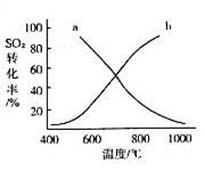
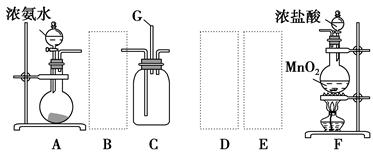
�� ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ��ģ��ʵ�����պ���������ʵ������ͼ��ʾ���� �����������SO2������Ч�ʡ�2��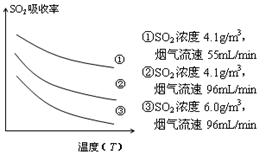
��3�������ֿ��ŵ�Na2SO3ҩƷ�Ѳ��ֱ������������û�ѧС��������֪Ũ�ȵ�����KMnO4��Һ��ȷ���京�������岽�����£�
����i����ȡ��Ʒ1.000 g��
����ii������Ʒ�ܽ����ȫת�Ƶ�250 mL����ƿ�У����ݣ����ҡ�ȡ�
����iii����ȡ25.00 mL��Ʒ��Һ��250 mL��ƿ�У���0.01000 mol��L��1 KMnO4����Һ�ζ����յ㡣
�����������������ظ�2�Ρ�
�� д������iii��������Ӧ�����ӷ���ʽ_________________________________��
�� ������0.01000 mol��L��1 KMnO4��Һʱ�����Ӷ��ݣ������ղ��ҩƷ��Na2SO3�ĺ���________(�ƫ����ƫС������Ӱ�족)��
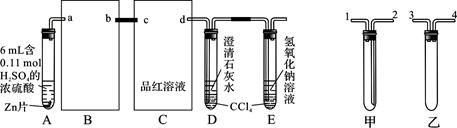
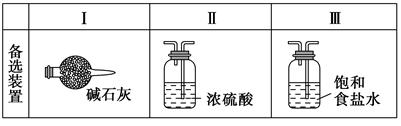
�� ijͬѧ����������������еζ�ʵ��(�гֲ�����ȥ)�������������� (����ĸ)��

A B C D E
�� �ζ�������±���ʾ��
| �ζ����� | ������Һ �����/mL | ����Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 2.20 | 20.20 |