��Ŀ����
6����1����ͼ1��ʾ��һ��ԭ��أ��ش��������⣺��Zn�缫�Ǹ��������������������缫��ӦʽΪZn-2e-=Zn2+��
��Cu�缫�ĵ缫��Ӧʽ��Cu2++2e-=Cu��
��2����ͼ2��ʾ��һ�����أ�A��BΪ���Ե缫���������ҺΪNaCl��Һ���ش��������⣺
��д���õ����ܷ�Ӧ�Ļ�ѧ����ʽ��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+Cl2��+H2��
�ڵ��������������������������������Һ����ֺ�ɫ��
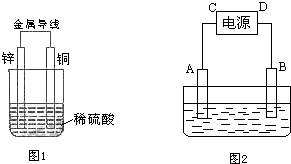
���� ��1��п��ͭ���ã�пΪ����������������Ӧ���缫����ʽΪZn-2e-=Zn2+��ͭΪ������������ԭ��Ӧ���缫����ʽΪCu2++2e-=Cu��
��2����ⱥ��ʳ��ˮʱ����������������ʧ���ӣ��������������ӵõ��ӣ���Һ�Լ��ԣ��Դ˽����⣮
��� �⣺��1����п��ͭ���ã�пΪ��������������������Ӧ���缫����ʽΪZn-2e-=Zn2+���ʴ�Ϊ������Zn-2e-=Zn2+��
��CuΪ����������������ԭ��Ӧ���缫����ʽΪCu2++2e-=Cu���ʴ�Ϊ��Cu2++2e-=Cu��
��2���ٵ缫�������ӷŵ磬ͬʱ�õ缫�����������������ӣ�Y�缫�������ӷŵ��������������Ե�ط�ӦʽΪ2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+Cl2��+H2����
�ʴ�Ϊ��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+Cl2��+H2����
�ں͵�Դ�ĸ��������ĵ缫���������õ缫�������ӷ����õ��ӵĻ�ԭ��Ӧ����2H++2e-=H2������Һ�Լ��ԣ�����̪��죬�ʴ�Ϊ������
���� ���⿼��ԭ��غ͵��صĹ���ԭ��֪ʶ��Ϊ��Ƶ���㣬������ѧ���ķ��������Ŀ��飬ע�����ԭ��غ͵��صĹ���ԭ���͵缫����ʽ����д���ѶȲ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
16��A��Al2O3��Fe3O4��Al��Cu�е�ij���ַ�ĩ��϶��ɣ��û����ɷֵķ���������ͼ��ʾ�����з���������ǣ�������
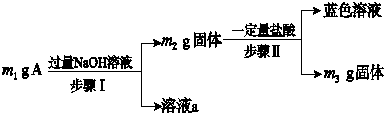

| A�� | ��m1��m2ʱ����Һa�е�������ֻ��1�� | |
| B�� | ������ɫ��Һ�����ӷ���ʽΪCu+2Fe3+�TCu2++2Fe2+ | |
| C�� | Ҫȷ����������Ƿ���Al����ȡA��������ϡ���� | |
| D�� | ��m2-m3=2.96 gʱ��Fe3O4����������Ϊ2.32 g |
17������������ȷ���ǣ�������
| A�� | 1molH2O������Ϊ18g/mol | |
| B�� | O2 ��Ħ������Ϊ32g | |
| C�� | 2mol NaOH ����1 Lˮ�������Һ��Ũ��Ϊ2mol/L | |
| D�� | 6.02��1023��CO���ӵ�����ԼΪ28g |
1���������ʷ����仯ʱ�����˷������Ӽ��������ͬ�����͵��ǣ�������
| A�� | ����ɱ��ֱ����ȱ�Ϊ���� | B�� | �����������ֱ����ȱ�Ϊ���� | ||
| C�� | �Ȼ������Ȼ���ֱ��ܽ���ˮ�� | D�� | ����������ֱ������ۻ� |
11���������ʲ�����������ˮ�������ǣ�������
| A�� | SO2 | B�� | Cl2 | C�� | ClO2 | D�� | O3 |
15�����й��ڳ����л����˵��������ǣ�������
| A�� | C5H12����������������ͬ���칹�� | |
| B�� | ����Ũ���ᡢŨ�����Ϲ�����ȡ�������Ƿ���ȡ����Ӧ | |
| C�� | ���ۺ���ά�ؾ����ã�C6H10O5��n��ʾ��������ǻ�Ϊͬ���칹�� | |
| D�� | ���ᡢ��������һ�������¾�����Cu��OH��2��Ӧ |
16��������Դ�Ĵ���ʹ�ûᵼ��ȫ���һ����ů����������������ǣ�������
| A�� | ��ʯ��Դ | B�� | ̫���� | C�� | ���� | D�� | ������ |
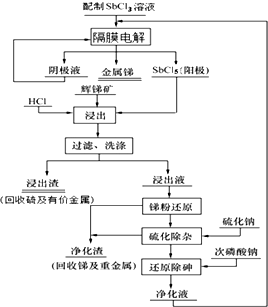 �ҹ�����̲���ռ�����һλ��ij�����������Ȼ���ˮ��Һ�������������ɵ����Ȼ�����Ϊ���������Ի����Sb2S3���������Խ������Ӷ�ʵ�ֽ���-���ı�·ѭ��������˴�ͳ��������С���ҵ���ϡ��ŷ���������⣮������ͼ��
�ҹ�����̲���ռ�����һλ��ij�����������Ȼ���ˮ��Һ�������������ɵ����Ȼ�����Ϊ���������Ի����Sb2S3���������Խ������Ӷ�ʵ�ֽ���-���ı�·ѭ��������˴�ͳ��������С���ҵ���ϡ��ŷ���������⣮������ͼ�� A��B��C�ǵ��ʣ�����A�dz����Ļ��ý�����B��C������ɫ���壬�����Ⱥ�����ɼף��������ʼ��ת����ϵ��ͼ��
A��B��C�ǵ��ʣ�����A�dz����Ļ��ý�����B��C������ɫ���壬�����Ⱥ�����ɼף��������ʼ��ת����ϵ��ͼ��