��Ŀ����
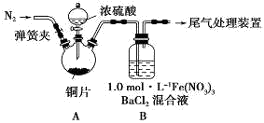
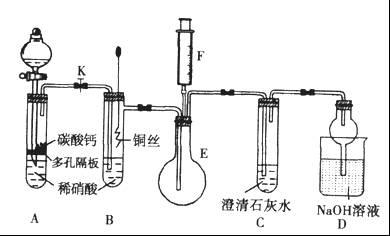
ij��ѧʵ��С���ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�á�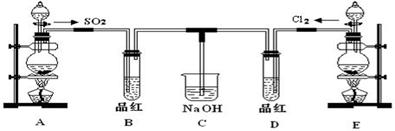
��1��ʵ������װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2��ʵ������װ��E�Ʊ�Cl2���䷴Ӧ�Ļ�ѧ��ѧ����ʽΪ��
MnO2+4HCl��Ũ�� MnCl2+Cl2��+2H2O������6 mol��HCl�μӷ�Ӧ����ת�Ƶĵ�������Ϊ ��
MnCl2+Cl2��+2H2O������6 mol��HCl�μӷ�Ӧ����ת�Ƶĵ�������Ϊ ��
��3���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�B�� ��D�� ��
��ֹͣͨ����,�ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�ΪB�� ��D�� ��
��4����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ��
��
��1����Һ©���IJ�����û��ȡ������2��1��806��1024����3NA����3����Ʒ����ɫ�� Ʒ����ɫ ����ɫ��Ʒ���ָֻ��ɺ�ɫ������������4��SO2+Cl2+2H2O=2HCl+H2SO4
���������������1����ʵ��ʱ��A�з�Һ©���Ļ�����©���е�Һ��û��˳�����£������ԭ���Ƿ�Һ©���Ͽڲ�����δ�������С�Ͳۿ�δ���루2��MnO2+4HCl��Ũ�� MnCl2+Cl2��+2H2O֪����6 mol��HCl�μӷ�Ӧ������3molCl����Ӧ����1��5molCl2��ת�Ƶ������ʵ���Ϊ3mol����ת�Ƶ�������3NA=1��806��1024��3���ٶ����������Ư�����ܺ���ɫ����������ɫ������������ˮ��Ӧ���ɴ��������Ư���ԣ��ʴ�Ϊ��Ʒ����ɫ��Ʒ����ɫ�ڴ������Ư���������ô������ǿ�����ԣ����������Ư���������ö����������ɫ����������ɫ���ʣ��������Ư�����������Եģ����������Ư��������ʱ�ģ�����ʱ��������Ư������Һ�ޱ仯�������������ɫ�������ɵ���ɫ���ʲ��ȶ�������ʱ�����±�ɺ�ɫ���ʴ�Ϊ����ɫ��Ʒ���лָ��ɺ�ɫ�������Ա仯��4��������ǿ�����ԣ����������л�ԭ�ԣ������ܰ�1��1����������ԭ��Ӧ��ʹ����ʧȥƯ���ԣ���Ӧ����ʽΪSO2+Cl2+2H2O�TH2SO4+2HCl����ǡ����ȫ��Ӧ��������Ư���ԡ�
MnCl2+Cl2��+2H2O֪����6 mol��HCl�μӷ�Ӧ������3molCl����Ӧ����1��5molCl2��ת�Ƶ������ʵ���Ϊ3mol����ת�Ƶ�������3NA=1��806��1024��3���ٶ����������Ư�����ܺ���ɫ����������ɫ������������ˮ��Ӧ���ɴ��������Ư���ԣ��ʴ�Ϊ��Ʒ����ɫ��Ʒ����ɫ�ڴ������Ư���������ô������ǿ�����ԣ����������Ư���������ö����������ɫ����������ɫ���ʣ��������Ư�����������Եģ����������Ư��������ʱ�ģ�����ʱ��������Ư������Һ�ޱ仯�������������ɫ�������ɵ���ɫ���ʲ��ȶ�������ʱ�����±�ɺ�ɫ���ʴ�Ϊ����ɫ��Ʒ���лָ��ɺ�ɫ�������Ա仯��4��������ǿ�����ԣ����������л�ԭ�ԣ������ܰ�1��1����������ԭ��Ӧ��ʹ����ʧȥƯ���ԣ���Ӧ����ʽΪSO2+Cl2+2H2O�TH2SO4+2HCl����ǡ����ȫ��Ӧ��������Ư���ԡ�
���㣺̽����ˮ��������Ư�����ã�̽������������Ʒ����Һ�ķ�Ӧ

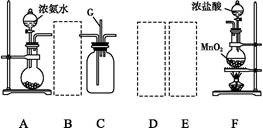
���ü�������������ȡ����Ӧ��ȡ����Ʒ����������ڹ�ҵ���ѳ�Ϊ��ʵ��ij��ѧ��ȤС����ʵ������ģ���������̣�����Ƶ�ģ��װ�����£�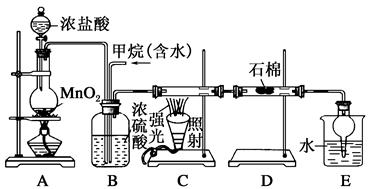
�������Ҫ��ش�
��1��Bװ�������ֹ��ܣ��ٿ��������ٶȣ��ھ��Ȼ�����壻��______________��
��2����V��Cl2��/V��CH4����x��������������������Ȼ��⣬��xֵӦ________��
��3��Dװ�õ�ʯ���о��Ȼ���KI��ĩ����������____________��
��4��Eװ�õ�������________������ţ���
| A���ռ����� | B���������� |
| C����ֹ���� | D�������Ȼ��� |
��6��Eװ�ó����������⣬�������л����E�з�����������ѷ���Ϊ________����װ�û���ȱ�ݣ�ԭ����û�н���β����������β����Ҫ�ɷ�Ϊ________������ţ���
a��CH4��b��CH3Cl��c��CH2Cl2��d��CHCl3��e��CCl4
��ҵβ���е�������ͨ�����ð������շ�����ԭ����NH3��NOx�ڴ��������·�Ӧ�����������ʡ�ijУ�С��ͬѧ��������װ�úͲ���ģ�ҵ�ϵ�������Ĵ������̡�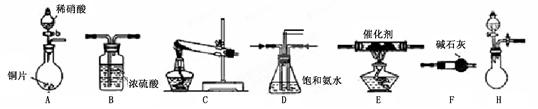
I��̽����ȡNH3�ķ���
��1��������װ���У�H�ܿ��١������ȡNH3��װ������Ҫ���ӵķ�Ӧ�Լ�Ϊ ��
��2��Ϊ̽�����õ�ʵ��Ч�����С��ͬѧ��������Cװ������ȡ�������ڿ���ʵ��������ͬ������£�����±���ʵ�����ݡ�
| �Լ������� | �����Լ� | NH3�����mL�� | |
| a | 6.0 g Ca(OH)2�������� | 5.4 g NH4Cl | 1344 |
| b | 5.4g (NH4)2SO4 | 1364 | |
| c | 6.0 g NaOH�������� | 5.4 g NH4Cl | 1568 |
| d | 5.4g (NH4)2SO4 | 1559 | |
| e | 6.0 g CaO�������� | 5.4 g NH4Cl | 1753 |
| f | 5.4 g (NH4)2SO4 | 1792 | |
�����������ݣ�����Ϊ���ַ�����ȡ������Ч����� ������ţ����Ӹ÷���ѡ���ԭ�Ϸ�������Ч���õĿ���ԭ���� ��
II��ģ��β������
�С��ͬѧѡ����������װ�ã�������˳�����ӳ�ģ��β������װ�ý���ʵ�顣
��1���������װ����ѡ������Ϊ�����Ľ��в��䣨��ѡװ�ò����ظ�����
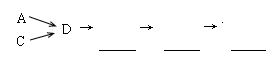
��2��A�з�Ӧ�����ӷ���ʽΪ ��
��3��Dװ�õ������У�ʹ�����Ͼ��ȡ����������ٶȡ� ��
��4��Dװ���е�Һ�廹�ɻ��� ������ţ���
a��H2O b��CCl4 c��ŨH2SO4 d��CuSO4��Һ
��5����С��ͬѧ����Ƶ�ģ��β������װ���л�����һ�����Ե�ȱ���� ��
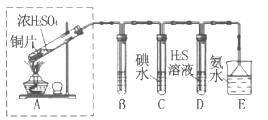
ij��ȤС���������ͼʵ��װ�ý���ʵ�顣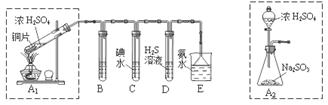
��̽��������Ⱦ��SO2������
��1��Ϊ��ʵ����ɫ������Ŀ�꣬�ܷ�����ͼA2����A1װ�� ����ܡ�����
��2��B��C��D�ֱ����ڼ���SO2��Ư���ԡ���ԭ�Ժ������ԣ���B����ʢ�Լ�Ϊ ��C�з�Ӧ�����ӷ���ʽΪ ��D�з�Ӧ�Ļ�ѧ����ʽΪ ��
��̽��ͭƬ��ŨH2SO4��Ӧ�IJ���
ʵ�������������ͭƬ���渽�ź�ɫ���塣�������ϵ�֪���˺�ɫ������ܺ���CuO��CuS��Cu2S��������CuS��Cu2S��������ϡ���ᣬ�ڿ��������ն�ת��ΪCu2O��SO2����С��ͬѧ�ռ�һ������ɫ���壬������ʵ�鷽��̽����ɷ֣�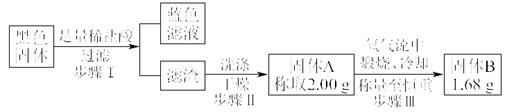
��3������� �м�������ϴ�Ӹɾ���ʵ�鷽����_____________________________��
��4����ɫ����ijɷ���________________��
�������
�ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH����SO32����SO42����HSO3���������ӡ�
��5����ˮ���չ���SO2�ķ�Ӧ�����ӷ���ʽΪ ��
��6����֪����������һ��������ˮ��SO2Ҳ������ˮ��
�����������Լ�Ϊ��С�ձ����Թܡ�����������ͷ�ιܡ�����װ�ú���ֽ��2 mol/L���ᡢ
2 mol/LHNO3��1 mol/LBaCl2��Һ��l mol/LBa(OH)2��Һ��Ʒ����Һ������ˮ�������ʵ��֤��������Һ���д���SO32����HSO3��������±���ʵ�������Ԥ������ͽ��ۣ�
| ʵ����� | Ԥ����������� |
| ����1��ȡ����������Һ����С�ձ��У��ý�ͷ�ι�ȡl mol/L BaCl2��Һ��С�ձ��μ�ֱ�������� | �����ְ�ɫ���ǣ�������Һ�� �д���SO32���� SO42���� |
| ����2����С�ձ��е���Һ���ˡ�ϴ�ӣ���������ˮ�Ѹ�����ֽ�ϵĹ��������һС�ձ��У�����µĹ��� �� | �� ������Һ���д��� SO32���� |
| ����3�� �� | �� ������Һ���д��� HSO3���� |
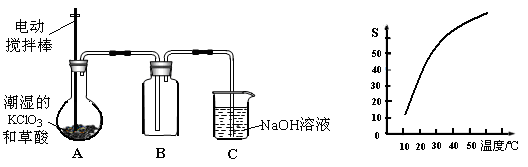
 8SO2��2Fe2O3�÷�Ӧ�б�������Ԫ����_______����Ԫ�ط��ţ������÷�Ӧת��2. 75mol����ʱ�����ɵĶ��������ڱ�״���µ����Ϊ_______L��
8SO2��2Fe2O3�÷�Ӧ�б�������Ԫ����_______����Ԫ�ط��ţ������÷�Ӧת��2. 75mol����ʱ�����ɵĶ��������ڱ�״���µ����Ϊ_______L��
 ��Һ��������________________��
��Һ��������________________��