��Ŀ����
��1��25��ʱ��ijNaCl��Һ��c(Cl�C)��1��10��4 mol��L�C1�������Һ��c(Na��)��c(OH��)��
��2��25��ʱ����0.1 mol��L�C1NaOH��Һ��0.06 mol��L�C1��H2SO4��Һ��������(���Ի�Ϻ�����ı仯)����������Һ��pH�� ��25��ʱ��pHֵΪ8��NaOH��Һ��pHֵΪ10��NaOH��Һ�������Ϻ���Һ��������Ũ����ӽ� ��
��3��25��ʱ������������Һ�У���pH=0������ ��0.1 mol��L�C1������ ��0.01 mol��L�C1��NaOH��Һ ��pH=11��NaOH��Һ����ˮ��������������Ũ��֮�Ȣ٩U�کU�۩U���ǣ� (����ĸ)
| A��1�U10�U100�U1000 | B��0�U1�U12�U11 |
| C��14�U13�U12�U11 | D��14�U13�U2�U3 |
������¶���ˮ��Kw�� ��
���ڸ��¶��²��ij��ҺpH��3�������Һ��c(H��)��c(OH��)��________��
�۸��¶��½�pH=2�������pH=11������������Һ�������ϣ�pH=______________
��5�� ��ˮ��c(H+)=5.0��10�C7 mol��L�C1�����ʱ��ˮ�е�c(OH�C) = �����¶Ȳ��䣬����ϡ����ʹc(H+)=5.0��10�C3 mol��L�C1����c(OH�C) = ���ڸ��¶�ʱ����ˮ�е���NaOH��Һ����Һ�е�c(OH�C)=5.0��10�C2 mol��L�C1������Һ��c(H+)= ��
��1��1000:1 ��2��2 2��10�C2 ��3�� A ��4��10�C13 107:1 6.5
��5��5.0��10�C7 mol��L�C1 5.0��10�C11 mol��L�C1 5.0��10�C12 mol��L�C1
���������������1��c(Na+)��c(Cl�C)��1��10��4 mol��L�C1��c(OH�C)��1��10��7mol��L�C1��c(Na��)��c(OH��)��1��10��4��1��10��7��1000:1����2����NaOH��Һ��H2SO4��Һ�������ΪV�������ڷ�Ӧ��H2SO4����������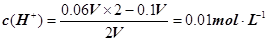 ����pH=�Clg0.01=2��pHֵΪ8��NaOH��Һ��c(OH��)=10��6 mol��L�C1��pHֵΪ10��NaOH��Һ��c(OH��)=10��4 mol��L�C1���������Ϻ�Ļ����Һ��
����pH=�Clg0.01=2��pHֵΪ8��NaOH��Һ��c(OH��)=10��6 mol��L�C1��pHֵΪ10��NaOH��Һ��c(OH��)=10��4 mol��L�C1���������Ϻ�Ļ����Һ��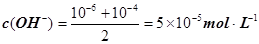 ��������Ũ��Ϊ
��������Ũ��Ϊ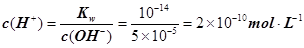 ����3�� ��10�C14 mol��L�C1����10�C13 mol��L�C1����10�C12 mol��L�C1����10�C11 mol��L�C1������ˮ��������������Ũ��֮�Ȣ٩U�کU�۩U��=1�U10�U100�U1000����ΪA����4�� ����c(OH�C)=10�C2 mol��L�C1��c(H+)=10�C11 mol��L�C1����Kw= c(H+)��c(OH�C)=10�C13������pH=3���c(H+)=10�C3 mol��L�C1����Kw= c(H+)��c(OH�C)=10�C13���c(OH�C)=10�C10 mol��L�C1�����Ը���Һ��c(H��)��c(OH��)��10�C3��10�C10��107��1���������е�c(H+)=10�C2 mol��L�C1������������Һ��c(OH�C)=10�C2 mol��L�C1������Һ��������ʱ�����ǡ����ȫ�кͣ���Һ�����ԣ�c(H+)=
����3�� ��10�C14 mol��L�C1����10�C13 mol��L�C1����10�C12 mol��L�C1����10�C11 mol��L�C1������ˮ��������������Ũ��֮�Ȣ٩U�کU�۩U��=1�U10�U100�U1000����ΪA����4�� ����c(OH�C)=10�C2 mol��L�C1��c(H+)=10�C11 mol��L�C1����Kw= c(H+)��c(OH�C)=10�C13������pH=3���c(H+)=10�C3 mol��L�C1����Kw= c(H+)��c(OH�C)=10�C13���c(OH�C)=10�C10 mol��L�C1�����Ը���Һ��c(H��)��c(OH��)��10�C3��10�C10��107��1���������е�c(H+)=10�C2 mol��L�C1������������Һ��c(OH�C)=10�C2 mol��L�C1������Һ��������ʱ�����ǡ����ȫ�кͣ���Һ�����ԣ�c(H+)= 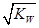 = 10�C6.5 mol��L�C1����pH=6.5����5��ˮ�����������Ũ�����ǵ�������������������Ũ�ȣ�����c(OH�C)=c(H+)=5.0��10�C7 mol��L�C1�����¶���Kw= c(H+)��c(OH�C)=5.0��10�C7��5.0��10�C7 =2.5��10�C13����c(H+)=5.0��10�C3 mol��L�C1����c(OH�C) =Kw/c(OH�C)=5.0��10�C11 mol��L�C1����c(OH�C)=5.0��10�C2 mol��L�C1������Һ��c(H+)=Kw/c(H+)=5.0��10�C12 mol��L�C1��
= 10�C6.5 mol��L�C1����pH=6.5����5��ˮ�����������Ũ�����ǵ�������������������Ũ�ȣ�����c(OH�C)=c(H+)=5.0��10�C7 mol��L�C1�����¶���Kw= c(H+)��c(OH�C)=5.0��10�C7��5.0��10�C7 =2.5��10�C13����c(H+)=5.0��10�C3 mol��L�C1����c(OH�C) =Kw/c(OH�C)=5.0��10�C11 mol��L�C1����c(OH�C)=5.0��10�C2 mol��L�C1������Һ��c(H+)=Kw/c(H+)=5.0��10�C12 mol��L�C1��
���㣺������Һ������Ũ�ȼ��㡢pH���㡢ˮ�����ӻ��ļ��㡣

(1)ˮ�ĵ���ƽ��������ͼ��ʾ����A���ʾ25��ʱˮ�ĵ����ƽ��ʱ������Ũ�ȣ�B���ʾ100��ʱˮ�ĵ����ƽ��ʱ������Ũ�ȡ���100��ʱ1 mol��L��1��NaOH��Һ�У���ˮ�������c(H��)��___________mol��L��1��25 ��ʱ����ˮ�ĵ���ƽ����ϵ�м�������NH4Cl���壬��ˮ�ĵ���ƽ���Ӱ����________(��ٽ����������ơ���Ӱ�족)��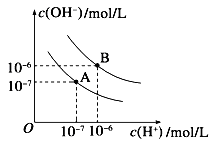
(2)����ƽ�ⳣ���Ǻ���������ʵ���̶�ǿ����������֪������ݡ�
| ��ѧʽ | ����ƽ�ⳣ��(25��) |
| HCN | K��4.9��10��10 |
| CH3COOH | K��1.8��10��5 |
| H2CO3 | K1��4.3��10��7��K2��5.6��10��11 |
c(CH3COO��)(�����������������)��
��25��ʱ���е�Ũ�ȵĢ�NaCN��Һ����Na2CO3��Һ����CH3COONa��Һ��������Һ��pH�ɴ�С��˳��Ϊ___________��(�����)
����NaCN��Һ��ͨ������CO2����������Ӧ�Ļ�ѧ����ʽΪ_________________________________��
����ȱ�ʾ����ʵ����ǿ��������ȵĶ��壺
�������ѵ���ĵ���ʷ���������Һ��ԭ�е���ʵ��ܷ���������100%��
��֪25��ʱ��������(��)�ĵ����(��ҺŨ�Ⱦ�Ϊ0.1 mol��L��1)���±���
| ��� | ����(��) | ����Ȧ� |
| A | ������Һ(��һ����ȫ����)�� �ڶ��� HSO4- H����SO42- H����SO42- | 10% |
| B | ����������Һ�� HSO4- H����SO42 H����SO42 | 29% |
| C | ��� CH3COOH CH3COO����H�� CH3COO����H�� | 1.33% |
| D | ��� HCl��H����Cl�� | 100% |
��1��25��ʱ��0.1 mol��L��1����������Һ�У�c(H��)�Ӵ�С��˳���� (�����)��
��2��25��ʱ��0.1 mol��L��1������Һ��HSO4-�ĵ����С����ͬ�¶���0.1 mol��L��1��������
��Һ��HSO4-�ĵ���ȣ���ԭ���� ��
��3������ĵ���ƽ�ⳣ��K�ı���ʽ�� ������ĵ���ƽ�ⳣ��
K�����Ȧ��Ĺ�ϵʽΪ��K= ���ú����Ĵ���ʽ��ʾ��
�Ӻ�ˮ����ȡþ������������þ����Ҫ��Դ����������ȡþ�Ĺ������漰�ļ������ʵ��ܶȻ�����������ѧ��֪ʶ�ش����м������⣺
| ���� | CaCO3 | MgCO3 | Ca(OH)2 | Mg(OH)2 |
| �ܶȻ� | 2.8��10�C9 | 6.8��10�C6 | 5.5��10�C6 | 1.8��10�C11 |
(1)�ڴӺ�ˮ����ȡþʱ�������õ�����(��Ҫ�ɷ���̼���)������Ϊ (��ܡ����ܡ�)��������ĥ�ɷ�ĩֱ��Ͷ�뺣ˮ�У������� ���������ֱ��Ͷ�룬Ӧ���������δ�������д����ѧ����ʽ ��
(2)ijͬѧ��ʵ����������ģ����������̣���ʵ������ʯ�ң����������ռ���棬����Ϊ (����ԡ������ԡ�)�õ�Mg(OH)2���ڼ��Լ�ʱ��������Һ���뺣ˮ�У���˼����һ�£����ڵõ��Ļ����ϵ�м���������ռ���Һ��������� (��ܡ����ܡ�)��Mg2+ת��ΪMg(OH)2������������ (�����ӷ���ʽ��ʾ)��
(3)��֪��ˮ��þ����Ũ��Ϊ1.8��10�C3mol��L�C1����Ҫʹþ���Ӳ�����������Һ��PH���ӦΪ ��
���仯�����й㷺��Ӧ�ã���SO2���ʵ��о��Ǹ��л�ѧ��ѧ��һ����Ҫ���ݡ�
I���Ա��о���һ����Ҫ���о�������������ĵ��ʼ����ֻ����ﰴ���±���ʾ�ֳ�3 �飬���2��������M�Ļ�ѧʽ�� ��
| ��1�� | ��2�� | ��3�� |
| S (����) | SO2��H2SO3��M��NaHSO3 | SO3��H2SO4��Na2SO4��NaHSO4 |
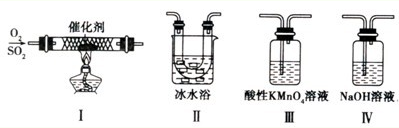
��1������ʵ�鷽������������ʵ������ȡ����SO2���� ��
A��Na2SO3��Һ��HNO3 B��Na2SO3����Ũ����
C���������ڴ�����ȼ�� D��ͭ���ȵ�Ũ����
��2��װ��C�������dz�ȥ�����SO2����ֹ��Ⱦ��������֪��������������Һ����SO2�� �����У������õ�Na2SO3��NaHSO3�Ļ����Һ�������£���ҺpH��n(SO32��)��n(HSO3��)�仯��ϵ���±�
| n(SO32��)��n(HSO3��) | 91��9 | 1��1 | 9��91 |
| pH | 8.2 | 7.2 | 6.2 |
������Һ��n(SO32��)��n(HSO3��) =10��1ʱ����Һ������Ũ�ȹ�ϵ��ȷ���� ��
A��c(Na+)+ c(H+)= 2c(SO32��)+ c(HSO3��)+ c(OH��)
B��c(Na+)��c(HSO3��)��c(SO32��)��c(OH��)��c(H+)
C��c(Na+)��c(SO32��)��c(HSO3��)��c(OH��)��c(H+)
��3��������װ����ͨ�������SO2��Ϊ����֤A��SO2��Fe3+������������ԭ��Ӧ������ȡA�з�Ӧ�����Һ�ֳ����ݣ������������̽��ʵ�飬�������۲��������ǵ�̽�����̣���ѡ�Լ���KMnO4��Һ��KSCN��Һ��BaCl2��Һ��ϡ���ᡢϡ���ᡢϡ�� �ᡢBa(NO3)2��Һ�����Ƶ���ˮ��
| ��� | ʵ�鷽�� | ʵ������ | ���� |
| ������ | ����һ����Һ�м���KMnO4��Һ��Һ | �Ϻ�ɫ��ȥ | SO2��Fe3+��Ӧ������Fe2+ |
| ������ | ���ڶ�����Һ�м��� | | SO2��Fe3+��Ӧ������Fe2+ |
| ������ | ���ڶ�����Һ�м��� | | SO2��Fe3+��Ӧ������SO42�� |
���������ٵó��Ľ����Ƿ���� ��ԭ�� ��
���������Ƶķ������뷽���۾��������ҵõ���Ӧ���ۣ����㽫�������������
��4��װ��B���ܱ���Br���Ļ�ԭ������SO2�������� ��
������ǿ�ᣬ��ѧ�ν�������ˮ��Һ�п�����ȫ���롣����ʵ�ǣ�������ˮ�еĵ�һ����������ȫ�ģ��ڶ������벢����ȫ����������Ϊ:H2SO4=H++HSO4-��HSO4- H+ + S042-��
H+ + S042-��
��ش������й�����:
��1��Na2SO4��Һ��_(������ԡ��������ԡ��������ԡ�)����������_
(�����ӷ���ʽ��ʾ)��
��2��H2SO4��Һ��BaC12��Һ��Ӧ�����ӷ���ʽΪ_ ��
��3����0��l0mol��L-1��Na2SO4��Һ�У���������Ũ�ȹ�ϵ��ȷ����_ (��д���)��
| A��c(Na+)=c(SO42-)+c��HSO4һ)+c(H2SO4) |
| B��c(OH-)="c(" HSO4-)+c(H+) |
| C��c( Na+)+c(H+)=c(OH-)+c(HSO4-)+2c(SO42-) |
| D��c( Na+)=2c(SO42-)+2c(HSO4-) |
��5����25��ʱ��0��10 mol��L-1H2SO4��Һ��pH=-lg0��11����0��10 mol��L-1��H2SO4��Һ��c(SO42-)= mol��L-1
 H����HA����HA��
H����HA����HA�� H++OH? ����ش�
H++OH? ����ش� Fe��2NaCl,�õ�طŵ�ʱ��������ӦʽΪ ________________ _____��
Fe��2NaCl,�õ�طŵ�ʱ��������ӦʽΪ ________________ _____��
