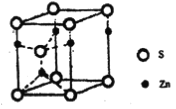
��Ŀ����
����Ŀ����ѧʵ���ұ������ϸ�Ĺ����ƶȺͿ�ѧ�Ĺ����������ش��������⣺
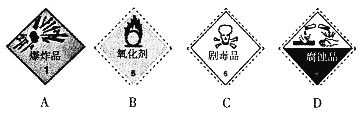
(1)Σ�ջ�ѧƷ��־���Ա�ʾ��ѧҩƷ��Σ�����������Ӧʹ�����б�־�е�_____(����)��
(2)�ء��ơ�þ�Ȼ��ý����Ż�ʱ��������Ʒ������������__________(����)��
A��ˮ B��ϸɳ C����ĭ����� D��������̼�����
����ѡ�����Ʒ�⣬����ʹ������������Ʒ����ء��ơ�þ�Ȼ��ý����Ż��ԭ����_________________��
(3)���ж�ҩƷ�ı��淽����ȷ����_________ (����)��
�ٽ�����������Ҵ������ͬһ������
�ڱ���Һ��ʱ�����м�������ˮ
���ò���ϸ��ƿʢװ�����
���ô���Ƥ���IJ����Լ�ƿʢװ����
������ɫ����ϸ��ƿʢװŨ����
(4)NaCN���ھ綾��ѧƷ��Ӧ�����ࡢ��������ʳ�û�ѧƷ�ֿ���š���������CN-�и�ԭ�Ӿ�����8�����ȶ��ṹ��NaCN�ĵ���ʽΪ______��NaCN��Һ�Լ��ԣ�ԭ����_____________(�����ӷ���ʽ��ʾ)��ǿ������NaC1O�ὫCN-����������N2��CO32-��C1-���������ʣ����ø÷�Ӧ���������ˮ(����)����Ӧ�����ӷ���ʽΪ_____________������һ����NaC1O����Ũ����ĺ����ˮ10L[c(CN-)=0.2mol/L]�������в�����״����21 L��������ù��̵������ʴﵽ_________����
���𰸡�BD B �ء��ơ�þ�Ȼ��ý�������ȼ�ղ������ˮ��������̼��Ӧ���ٽ����� �ڢ� ![]() CN-+H2O
CN-+H2O![]() HCN+OH- 2CN-+5ClO-+2OH-=2CO32-+N2��+5C1-+H2O 93.75
HCN+OH- 2CN-+5ClO-+2OH-=2CO32-+N2��+5C1-+H2O 93.75
��������
(1)����Ũ����Ļ�ѧ���ʺͳ��û�ѧΣ��Ʒ�ı�־˼����
(2)�Ӽء��ơ�þ��ȼ�ղ�������ʷ����жϣ�
(3)���ʱ���ʱ��Ҫ�Ӽ���ҩƷ����ʧ��ҩƷ���ʡ���ҩƷ���Լ�ƿ��ƿ����Ӧ�ȽǶȷ�����
(4)NaCN�����ӻ�����������ʺ��еĸ���Ԫ�ص�ԭ�ӽṹ����������CN-�и�ԭ�Ӿ�����8�����ȶ��ṹ�����������ε���ɼ�����Һ�ʼ��ԣ�����ε�ˮ�������д����ʽ�����ݵ����غ㡢����غ㼰ԭ���غ���д������ԭ��Ӧ�����ӷ���ʽ��
����ʵ�ʲ�N2���������۲�N2�����ıȾ��Dz��ʷ������㡣
(1)����������ɫ���ӷ����д̼�����ζ��Һ�壬���к�ǿ�������ԣ���ǿ�����������Ӧ��ʹ�� ��
�� ������ѡ����BD��
������ѡ����BD��
(2)A.�ء��ơ�þ������ˮ��Ӧ����Ӧ�����������ܹ�ȼ�գ�����ȼ�ղ���������ơ��������ؿ�����ˮ��Ӧ��������������ȼ���ã���˲�����ˮ���A����
B.ϸɳ�������������ȼ�ղ��ܷ�Ӧ��ϸɳ��״̬Ϊ���壬�ܶȴ������Ż����ϣ�����ʹ����������������ﵽ����Ŀ�ģ�B��ȷ��
C.��ĭ�����������������к���ˮ��������̼�������������ء��ơ�þ������ˮ��Ӧ��ȼ�ղ���������ơ��������ؿ����������̼��Ӧ��þ���ڶ�����̼��ȼ�գ����Բ�������ĭ��������C����
D.������̼�����������Ķ�����̼�ܹ���������ơ��������ط�Ӧ��������������ȼ���ã�þ���ڶ�����̼��ȼ�գ����Բ����ö�����̼�������D����
�ʺ���ѡ����B��
����ѡ�����Ʒ�⣬����ʹ������������Ʒ����ء��ơ�þ�Ȼ��ý����Ż��ԭ���Ǽء��ơ�þ�Ȼ��ý�������ȼ�ղ������ˮ��������̼��Ӧ���ٽ����ƼӾ磻
(3)�ٸ�����ؾ���ǿ�������ԣ��Ҵ����л�ԭ�ԣ�����������������ԭ��Ӧ�����±��ʣ��ʶ��߲��ܴ����ͬһ�����У��ٲ��������⣻
��Һ��е�ͣ����ӷ����ڱ���Һ��ʱ�����м�������ˮ���γ�ˮ�⣬�ɼ�����Ļӷ���ʧ���ڷ������⣻
��������ܹ��벣���ijɷ�SiO2������Ӧ,��ʴ���������Բ���ʹ�ò���ƿ��ţ��۲��������⣻
�������dz����л��ܼ��������ܽ���Ƥ�������Բ���ʹ��������Ҫʹ�ò��������ܲ��������⣻
��Ũ����������ֽ⣬Ϊ��ֹ��ֽ⣬������ɫ����ϸ��ƿʢװŨ���ᣬ��ֹ�䰵�����ݷ������⣻
�ʢڢݱ��淽���õ�������ѡ���Ǣڢݣ�
(4)NaCN�����ӻ�����������Na+��CN-������Cԭ���������4�����ӣ�Nԭ���������5�����ӣ���CN-�и�ԭ�Ӿ�����8�����ȶ��ṹ����CN-��C��Nԭ���γ�3�Թ��õ��Ӷԣ�Naԭ��ʧȥ�������ӱ�ΪNa+��Cԭ�ӻ��1�����ӣ��Ӷ�ʹCN-�е�Cԭ�Ӵﵽ8�����ȶ��ṹ����NaCN�ĵ���ʽΪ![]() ��NaCN��ǿ�������Σ�����Һ�У�CN-����ˮ�ⷴӦ������ˮ���������H+������ʹ��Һ��c(OH-)>c(H+)����Һ�Լ��ԣ����ӷ���ʽΪ��CN-+H2O
��NaCN��ǿ�������Σ�����Һ�У�CN-����ˮ�ⷴӦ������ˮ���������H+������ʹ��Һ��c(OH-)>c(H+)����Һ�Լ��ԣ����ӷ���ʽΪ��CN-+H2O![]() HCN+OH-��ǿ������NaC1O�ὫCN-����������N2��CO32-��NaC1O����ԭΪC1-���������ʣ�CN-ʧȥ5�����ӣ�ClO-���2�����ӣ����ݵ����غ��֪CN- ϵ����2��CO32-ϵ����2��N2��ϵ����1����ClO-��Cl-��ϵ������5���ٸ��ݵ���غ��֪OH-ϵ����2��������ԭ���غ㣬�ɵ�H2Oϵ����1���÷�Ӧ�����ӷ���ʽΪ2CN-+5ClO-+2OH-=2CO32-+N2��+5C1-+H2O��
HCN+OH-��ǿ������NaC1O�ὫCN-����������N2��CO32-��NaC1O����ԭΪC1-���������ʣ�CN-ʧȥ5�����ӣ�ClO-���2�����ӣ����ݵ����غ��֪CN- ϵ����2��CO32-ϵ����2��N2��ϵ����1����ClO-��Cl-��ϵ������5���ٸ��ݵ���غ��֪OH-ϵ����2��������ԭ���غ㣬�ɵ�H2Oϵ����1���÷�Ӧ�����ӷ���ʽΪ2CN-+5ClO-+2OH-=2CO32-+N2��+5C1-+H2O��
n(CN-)=0.2mol/L��10L=2mol������Nԭ���غ��֪�����Ϸ�Ӧ�õ����������ʵ���Ϊn(N2)=![]() n(CN-)=1mol�����ڱ�״���µ����V=22.4L��ʵ�ʵõ����������Ϊ21 L�����Ըù��̵������ʴﵽ
n(CN-)=1mol�����ڱ�״���µ����V=22.4L��ʵ�ʵõ����������Ϊ21 L�����Ըù��̵������ʴﵽ![]() =93.75%��
=93.75%��

 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д�����Ŀ������������ѧʵ���ҳ����Լ������������ȶ����������·�Ӧ��
��2AgNO3(s)= 2Ag(s)+ 2N02(g)+O2(g) ��H1
��2NO2(g)![]() N2O4(g) ��H2
N2O4(g) ��H2
(1)2AgNO3(s)= 2Ag(s)+N2O4(g)+O2(g) ��H=___________(�ú���H1����H2��ʽ�ӱ�ʾ)��
(2)ʵ����������������Һ�IJ��������ǣ���һ������������������Ũ�����У���ˮϡ����ָ������������ᡱ��������____________________________________________��
(3)һ���¶��£���5L�ĺ����ܱ�������Ͷ��34 g AgNO3(s)����ȫ�ֽ��û������������ʵ�����n)��ʱ��(t)�Ĺ�ϵ��ͼ��ʾ��
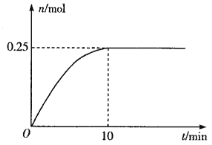
�����������˵����ϵ�ﵽƽ��״̬����___________(����ĸ)
a.Ag�۵��������ٸı� b.O2��Ũ�Ȳ��ٸı�
c.NO2������������ٸı� d.���������ܶȲ��ٸı�
�ڷ�Ӧ��ʼ��10min��N2O4��ƽ����Ӧ����Ϊ___________mol��L��1��min��1��
�����ﵽƽ��ʱ������������ѹǿp=3.0MPa���ڸ��¶���2NO2(g)![]() N2O4(g)��ƽ�ⳣ��Kp=___ (MPa)��1(�������2λС��)��[��ʾ����ƽ��ʱ����ַ�ѹ���Ũ�ȼ����ƽ�ⳣ����ѹǿƽ�ⳣ��(Kp)����ֵķ�ѹ(P1)=ƽ��ʱ��ѹ(P)������ֵ��������(
N2O4(g)��ƽ�ⳣ��Kp=___ (MPa)��1(�������2λС��)��[��ʾ����ƽ��ʱ����ַ�ѹ���Ũ�ȼ����ƽ�ⳣ����ѹǿƽ�ⳣ��(Kp)����ֵķ�ѹ(P1)=ƽ��ʱ��ѹ(P)������ֵ��������(![]() )]
)]
�ܴﵽƽ������������г���������NO2��ƽ��2NO2(g)![]() N2O4(g)___________(��������ҡ�����)�ƶ���NO2��ƽ��ת����___________���������С�����䡱����
N2O4(g)___________(��������ҡ�����)�ƶ���NO2��ƽ��ת����___________���������С�����䡱����
(4)��֪������������ܶȻ����±���ʾ��
������ | AgI | AgSCN | AgCl | Ag2CrO4 | Ag2S |
�ܶȻ� | 8.5��10��17 | 1.2x10��12 | 1.8��10��10 | 1.1��10��11 | 6.7��10��50 |
��ɫ | ��ɫ | ��ɫ | ��ɫ | ��ɫ | ��ɫ |
ʵ���ҳ���AgNO3��Һ�ζ�Cl������ѡ��___________��ָʾ��(����ĸ)
A.KI B. NH4SCN C. Na2CrO 4 D.K2S
(5)��ҵ�ϣ����õ�ⷨ���������������к���Cu��Au�����ʣ���AgNO3��Һ���������Һ�����Ե���ǿ��aA��ͨ��bmin���Ƶþ���������Ϊckg����þ���װ�õĵ���Ч��Ϊ___________(ֻҪ���г�����ʽ����)��[��֪��1mol���Ӵ�96500C(����)����������Ч�ʵ��������õ�������ͨ����������֮��]