��Ŀ����
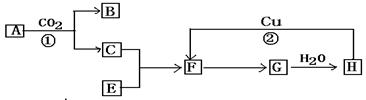
��X��Y��Z����Ԫ�أ���֪����X��Y��Z�ĵ����ڳ����¾�Ϊ���壻��X���ʿ���Z������ȼ�գ�������̬XZ����XZ��������ˮ�������X+��Z������ˮ��Һ��ʹ��ɫʯ����ֽ��죻��ÿ2��X2���ӿ���1��Y2���ӻ�������2��X2Y���ӣ�X2Y�ڳ�����Ϊ��ɫҺ�壻��Z��������X2Y�У�������Һ����Ư�����á�
��1���ƶ�X��Y����Ԫ�أ�X________��Y_________����Ԫ�����ƣ���
��2��д���ݹ����еĻ�ѧ��Ӧ�ķ���ʽ ��
��3��д����ҵ����ȡZ���ʵĻ�ѧ����ʽ ��
��4��дZ������ʯ���鷴Ӧ�Ļ�ѧ����ʽ ��
��1���� �� ��2��Cl+H2O HCl+HClO
HCl+HClO
��3��2NaCl��2H2O 2NaOH��H2����Cl2�� ��4��2Ca(OH)2 +2Cl2��Ca(ClO)2+CaCl2+2H2O
2NaOH��H2����Cl2�� ��4��2Ca(OH)2 +2Cl2��Ca(ClO)2+CaCl2+2H2O
�������������X�ĵ�����Z�ĵ�����ȼ�գ�����XZ������XZ��������ˮ����ˮ��Һ�е����X+��Z-��XZ��ˮ��Һ��ʹʯ����Һ��죬˵��XΪH2��ZΪCl2��XZΪHCl��������X�ĵ��ʿ���һ����Y�ĵ��ʻ�������������X2Y��X2Y������ΪҺ�壬˵��YΪO2��X2YΪH2O��Cl2����H2O�У���Ӧ����HCl��HClO��HClO����Ư�����á�
��1��ͨ�����Ϸ���֪��X��YԪ�����Ʒֱ����⡢�����ʴ�Ϊ���⣻����
��2��Cl2����H2O�У���Ӧ����HCl��HClO����Ӧ�Ļ�ѧ����ʽΪCl+H2O HCl+HClO��
HCl+HClO��
��3����ҵ���õ�ⱥ���Ȼ�����Һ�ķ�����ȡ��������Ӧ����ʽΪ2NaCl��2H2O 2NaOH��H2����Cl2����
2NaOH��H2����Cl2����
��4���������������Ʒ�Ӧ�����Ȼ��ơ�������ƺ�ˮ����Ӧ����ʽΪ2Ca(OH)2��2Cl2��Ca(ClO)2��CaCl2��2H2O
���㣺���������ƶϡ��Լ�������ҵ�Ʊ�������

 Ӧ������ҵ��ϵ�д�
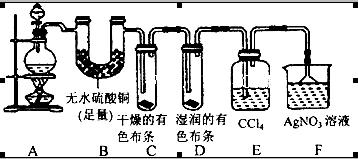
Ӧ������ҵ��ϵ�д�Ϊ�о�ͭ��Ũ����ķ�Ӧ��ij��ѧ��ȤС���������ʵ�顣
ʵ���Ӧ����Ķ���̽����
ʵ��װ����ͼ��ʾ�����̶�װ������ȥ��
��1��A�з�Ӧ�Ļ�ѧ����ʽΪ ��
��2��F�ձ��е���Һͨ���� ��
��3��ʵ������У���֤��Ũ��������Ԫ�ص�������ǿ����Ԫ�ص�������
��
��4��ʵ�������֤��Aװ���Թ��з�Ӧ���ò����Ƿ���ͭ���ӵIJ��������� ��
��5��Ϊ˵��Ũ�����е�ˮ�Ƿ�Ӱ��Bװ��������жϣ��������һ��ʵ�顣ʵ�鷽��Ϊ ��
ʵ���Ӧ����Ķ���̽��
��6����ͭ��Ũ���ᷴӦ�Ĺ����У������к�ɫ���ʳ��֣�������������������ϡ�
����1��
| ����/mol��L��1 | ��ɫ���ʳ��ֵ��¶�/�� | ��ɫ������ʧ���¶�/�� |
| 15 | Լ150 | Լ236 |
| 16 | Լ140 | Լ250 |
| 18 | Լ120 | ����ʧ |
a��ͭ��Ũ���ᷴӦʱ���漰�ķ�Ӧ���ܲ�ֹһ��
b������Ũ��ѡ���ʵ����ɱ����������г��ֺ�ɫ����
c���÷�Ӧ����������֮һ������Ũ�ȡ�15 mol/L
d������Ũ��Խ��ɫ����Խ����֡�Խ����ʧ
��7��Ϊ�������ͭ�IJ��ʣ����÷�Ӧ������Һ�кͺ����Ƴ�250.00 mL��Һ��ȡ����Һ25.00 mL��������KI��Һ���Ե�����ҺΪָʾ������b mol/L Na2S2O3��Һ�ζ����ɵ�I2��3��ʵ��ƽ�����ĸ�Na2S2O3��ҺV mL������Ӧ����ͭ������Ϊa g��������ͭ�IJ���Ϊ _������֪��2Cu2����4I��===2CuI��I2��2S2O
 ��I2===S4O
��I2===S4O ��2I����
��2I���� 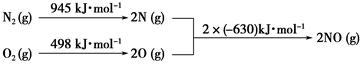

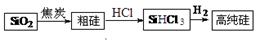
 Si(s) + 3HCl(g) ��H��0����һ�����ķ�Ӧ��ͨ��̶��ݻ����ܱ������н��з�Ӧ������˵����ȷ���� ������ĸ����
Si(s) + 3HCl(g) ��H��0����һ�����ķ�Ӧ��ͨ��̶��ݻ����ܱ������н��з�Ӧ������˵����ȷ���� ������ĸ����