��Ŀ����
11��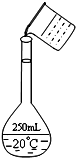 ʵ�����ù���NaOH����0.5mol/L��NaOH��Һ500mL����������������Ʒ�У����ձ� ��100mL��Ͳ ������ƿ ��ҩ�� �ݲ����� ��������ƽ�������룩 �ߵζ���
ʵ�����ù���NaOH����0.5mol/L��NaOH��Һ500mL����������������Ʒ�У����ձ� ��100mL��Ͳ ������ƿ ��ҩ�� �ݲ����� ��������ƽ�������룩 �ߵζ�����1������ʱ������ʹ�õ���������Ʒ�Т٢ۢܢݢޣ�����ţ�����ȱ�ٵ�һ�������ǽ�ͷ�ιܣ�
��2�������Ƶ�ת�ƹ�����ijѧ����������ͼ��������ָ�����еĴ���δ�ò�����������Ӧʹ��500mL������ƿ
��3������ʱ��һ��ɷ�Ϊ���¼������裺�ٳ��� �ڼ��� ���ܽ� ��ҡ�� ��ת �Ƣ�ϴ�Ӣ߶��ݢ���ȴ������ȷ�IJ���˳��Ϊ�ڢ٢ۢ�ݢޢߢܣ�
��4�����в�����ʹ���Ƶ���ҺŨ��ƫ�͵���A��B������ţ�
A��û�н�ϴ��Һת�Ƶ�����ƿ B��ת�ƹ�������������Һ����
C������ƿϴ����δ���� D������ʱ���ӿ̶���
��5��������ƿʹ�÷����У����в�������ȷ���ǣ�����ţ�B��C
A��ʹ������ƿǰ������Ƿ�©ˮ B������ƿ������ˮϴ�������ü�Һ��ϴ
C�����������ƹ��������ƽ���̵���ֽ�ϣ�ȷ�����������ձ����ܽ������ע������ƿ��
D�����ݺ�����ƿ������ʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��תҡ��
��6��ʵ���л���Ҫ2mol/L��NaOH��Һ950mL������ʱӦѡ�õ�����ƿ�Ĺ��ͳ�ȡNaOH�������ֱ���A������ţ�
A��1000mL��80g B��950mL��76g C��������72g D��500mL��42g��
���� ��1����������һ�����ʵ���Ũ����Һ��һ�㲽��ѡ����Ҫ��������
��2��������Һʱ��Ӧ����������Һ���ѡ������ƿ����ҺʱӦ�ò�����������
��3����������һ�����ʵ���Ũ����Һ��һ�㲽������
��4���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��5����������ƿ���켰ʹ�÷������
��6��ʵ��������Ҫ����2mol/L��NaOH��Һ950mL������û��950mL������ƿ��ʵ������ʱ��Ҫѡ��1000mL����ƿ�����Ƶ���ҺΪ1000mL 2mol/L��NaOH��Һ������ʵ�����Ƶ�����������Һ�������Ũ�ȼ�����������Ƶ�������
��� �⣺��1������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ���������������ƽ��ҩ�ס��ձ�����������500mL����ƿ����ͷ�ιܣ����Ա�����������٢ۢܢݢޣ�
��ȱ�ٵ���������ͷ�ιܣ�
�ʴ�Ϊ���٢ۢܢݢޣ� ��ͷ�ιܣ�
��2������0.5mol/L��NaOH��Һ500mLӦѡ��500mL����ƿ����ҺʱӦ�ò�����������
�ʴ�Ϊ��δ�ò�����������Ӧʹ��500mL������ƿ��
��3������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������ȷ��˳��Ϊ���ڢ٢ۢ�ݢޢߢܣ�
�ʴ�Ϊ����ݢޢߣ�
��4��A��û�н�ϴ��Һת�Ƶ�����ƿ���������ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ���Aѡ��
B��ת�ƹ�������������Һ�������������ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ���Bѡ��
C������ƿϴ����δ��������ʵ����ʵ�������Һ�����������Ӱ�죬��ҺŨ�Ȳ��䣬��C��ѡ��
D������ʱ���ӿ̶��ߣ�������Һ���ƫС����ҺŨ��ƫ�ߣ���D��ѡ��
��ѡ��AB��
��5��A��ʹ������ƿǰӦ�ü����Ƿ�©ˮ����A��ȷ��
B������ƿ��ˮϴ�������ô�����Һϴ�ӣ������Ӱ��������Һ��Ũ�ȣ���B����
C��������Һʱ����������Һ�壬Ӧ�����ձ����ܽ⣬��ҩƷ��ȫ�ܽ�ָ������£��ٰ���ҺС�ĵ�������ƿ�У���C����
D���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת��Σ�ҡ�ȣ���D��ȷ��
��ѡ��BC��
��6��ʵ��������Ҫ����2mol/L��NaOH��Һ950mL������û��950mL������ƿ��ʵ������ʱ��Ҫѡ��1000mL����ƿ�����Ƶ���ҺΪ1000mL 2mol/L��NaOH��Һ����Ҫ�������Ƶ�����m=cVM=1L��2mol/L��40g/mol=80.0g��
��ѡ��A��
���� ���⿼��������һ�����ʵ���Ũ�ȵ���Һ�ķ�������ȷ����ƿ�����Ĺ��ѡ���ǽ���ؼ���ע�����ʼ���Ӧ����������Һ������㣬��Ŀ�ѶȲ���

 ����ͬ�����Ծ�ϵ�д�
����ͬ�����Ծ�ϵ�д�| ʱ��t/h | 0 | 1 | 2 | 4 | 8 | 16 | 20 | 25 | 30 |
| ��ѹǿp/100kPa | 4.91 | 5.58 | 6.32 | 7.31 | 8.54 | 9.50 | 9.52 | 9.53 | 9.53 |
��1�������A��ƽ��ת���ʣ�Ӧ��ȡ�Ĵ�ʩΪ�����¶ȡ�����ѹǿ��
��2������ѹǿp����ʼѹǿp0���㷴Ӧ��A��ת���ʦ���A���ı���ʽΪ$\frac{P-P{\;}_{0}}{P{\;}_{0}}$��100%��ƽ��ʱA��ת����Ϊ94.1%����ʽ�����㷴Ӧ��ƽ�ⳣ��K1.5mol/L��
��3��������ѹǿp����ʼѹǿp0��ʾ��Ӧ��ϵ�������ʵ���n�ܺͷ�Ӧ��A�����ʵ���n��A����n��=$\frac{0.10P}{P{\;}_{0}}$mol��n��A��=0.10����2-$\frac{p}{p{\;}_{0}}$��mol��
���±�Ϊ��Ӧ��AŨ���뷴Ӧʱ������ݣ�����a=0.051����
| ��Ӧʱ��t/h | 0 | 4 | 8 | 16 |
| c��A��/��mol•L-1�� | 0.10 | a | 0.026 | 0.0065 |
| A�� | NH4+��NO3-��CO32-��Na+ | B�� | Na+��Ba2+��Mg2+��HCO3- | ||
| C�� | NO3-��Ca2+��K+��Cl- | D�� | NO3-��K+��ClO-��Ca2+ |
| A�� | ��22.4LHCl��������ˮ���1L��Һ�����ʵ����ʵ���Ũ��Ϊ1 mol•L-1 | |
| B�� | ����0.5L 0.1 mol•L-1��NaOH��Һ����Ҫ�����������ƹ���2.0g | |
| C�� | 0.5 L 2 mol•L-1���Ȼ�����Һ�У������Ӻ�����������Ϊ1.806��1024 | |
| D�� | �ܶ�Ϊ0.91 g•cm-3�İ�ˮ����������Ϊ25%���ӵ������ˮϡ�ͺ�������Һ����������С��12.5% |
| A�� | ����ϡ������Һ��Ӧ 2Fe+6H+�T2Fe2++6H2�� | |
| B�� | �����С�մ���Һ��Ӧ CH3COOH+HCO3-�TCH3COO-+H2O+CO2�� | |
| C�� | ������̼�������ռ���Һ��Ӧ CO2+2OH-�TCO32-+H2O | |
| D�� | �Ȼ�������������Ӧ�����Ȼ��� Fe2++Cl2�TFe3++2Cl- |
| A�� | ${\;}_{92}^{235}$U ԭ�Ӻ��к���92������ | |
| B�� | ${\;}_{92}^{235}$U ԭ�Ӻ�����143������ | |
| C�� | ${\;}_{92}^{235}$U ��������Ϊ92 | |
| D�� | ${\;}_{92}^{235}$U ��${\;}_{92}^{238}$U ��Ϊͬλ�� |
| A�� | ǰ�ߴ� | B�� | ���ߴ� | C�� | ��� | D�� | ����ȷ�� |