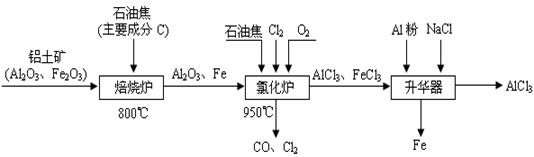
��Ŀ����
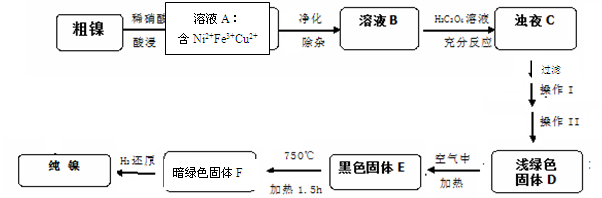
��14�֣�����һ��ʮ����Ҫ����ɫ����,��������һ�㺬��Fe��Cu�������ᡢ����Һ��Ӧ�IJ��������ʶ�Ӱ��ʹ�á��ֶԴ��������ᴿ������ķ�Ӧ�������£�
��֪��
��2Ni2O3(��ɫ) 4NiO(����ɫ) + O2��
4NiO(����ɫ) + O2��
��
������Ϣ�ش�
��1��28Ni�����ڱ��еĵ� ���ڣ�����ϡ���ᷴӦ�����ӷ���ʽΪ ��ʵ��������Ľ��������¶��йأ������¶��������Ľ������������¶ȸ���70�� ʱ�����Ľ������ֽ��ͣ���������Ni(OH)2����������ԭ��Ϊ ��
��2������ҺA�ľ��������У����Ƚ���ҺA��У�����PH=5.5���������5min,����һ��ʱ����˳�Fe(OH)3��Ϊ�˵õ���������ҺB����Ҫ������������ ���г��ӣ�����ȷ�𰸱�ţ���
A��NaOH B��Na2S C��H2S D��NH3�qH2O
��3����֪�����������ʵ��ܽ�ȹ�ϵ��NiC2O4>NiC2O4��H2O> NiC2O4��2H2O�������I��II������ �� �� D����E�Ļ�ѧ����ʽΪ ��
��4��1molE���������̵õ������������ϲμӷ�Ӧ������Ϊ mol��
��5���ⶨ��Ʒ�е��������Ļ���˼·�ǽ�Ni2+ת��Ϊ������ͨ������������������һ������Ni��������֪Ni2+����CO32-��C2O42-��S2-�������γɳ��������ⶨʱ��ѡ�ö���ͪ뿣�C4H8N2O2���������������ɷ���ʽΪC8H14N4O4Ni���ʺ�ɫ��������ʵ�����Ƕȷ�����Ҫԭ��Ϊ�� ��

��֪��
��2Ni2O3(��ɫ)
 4NiO(����ɫ) + O2��
4NiO(����ɫ) + O2����
| ���� | CuS | Cu(OH)2 | Ni(OH)2 | NiS |
| Ksp | 8.8��10��36 | 2.2��10��20 | 5.48��10-16 | 3.2��10-19 |
������Ϣ�ش�
��1��28Ni�����ڱ��еĵ� ���ڣ�����ϡ���ᷴӦ�����ӷ���ʽΪ ��ʵ��������Ľ��������¶��йأ������¶��������Ľ������������¶ȸ���70�� ʱ�����Ľ������ֽ��ͣ���������Ni(OH)2����������ԭ��Ϊ ��
��2������ҺA�ľ��������У����Ƚ���ҺA��У�����PH=5.5���������5min,����һ��ʱ����˳�Fe(OH)3��Ϊ�˵õ���������ҺB����Ҫ������������ ���г��ӣ�����ȷ�𰸱�ţ���
A��NaOH B��Na2S C��H2S D��NH3�qH2O
��3����֪�����������ʵ��ܽ�ȹ�ϵ��NiC2O4>NiC2O4��H2O> NiC2O4��2H2O�������I��II������ �� �� D����E�Ļ�ѧ����ʽΪ ��
��4��1molE���������̵õ������������ϲμӷ�Ӧ������Ϊ mol��
��5���ⶨ��Ʒ�е��������Ļ���˼·�ǽ�Ni2+ת��Ϊ������ͨ������������������һ������Ni��������֪Ni2+����CO32-��C2O42-��S2-�������γɳ��������ⶨʱ��ѡ�ö���ͪ뿣�C4H8N2O2���������������ɷ���ʽΪC8H14N4O4Ni���ʺ�ɫ��������ʵ�����Ƕȷ�����Ҫԭ��Ϊ�� ��
������ע�Ŀ��⣬ÿ��2�֣�
��1���ģ�1�֣���3Ni + 8H+ + 2NO3�� = 3Ni2+ + 2NO��+ 4H2O, �¶�����ʱ��Ni2+��ˮ��̶ȱ��
��2��C
��3��ϴ�ӡ������1�֣� 4NiC2O4��2H2O +3 O2= 2Ni2O3+ 8CO2�� +8H2O
��4�� 2
��5�����ɳ��������������С���������ȷ�ȸߡ�
��1���ģ�1�֣���3Ni + 8H+ + 2NO3�� = 3Ni2+ + 2NO��+ 4H2O, �¶�����ʱ��Ni2+��ˮ��̶ȱ��
��2��C
��3��ϴ�ӡ������1�֣� 4NiC2O4��2H2O +3 O2= 2Ni2O3+ 8CO2�� +8H2O
��4�� 2
��5�����ɳ��������������С���������ȷ�ȸߡ�
�����������1��28Ni������4����ӣ��ǵ�������Ԫ�أ���������ͼ��֪����ϡ���ᷴӦ������������NO��ˮ�����ӷ���ʽΪ3Ni + 8H+ + 2NO3�� = 3Ni2+ + 2NO��+ 4H2O��Ni(OH)2����Ni2+ˮ����õ��ģ������¶ȹ���ʱNi2+��ˮ��̶ȱ��������Ni(OH)2��������
��2��ABѡ���к���Na+������ҺB����Na+���ʣ���������������ѡ��D����Cu(OH)2 Ni(OH)2���ܶȻ�����������NH4+�����Ĵ�������������Һ������C��CuS��NiS���ܶȻ����ܴ�Cu2+������ȫ���Ҳ��������µ����ʣ���ȷ����ѡC��
��3������3�����ʵ��ܽ�ȵIJ��죬��ҺC����Ҫ��NiC2O4��2H2O�����Գ�������֮��Ӧϴ�ӡ��������������EΪ��ɫ������Ӧ��Ni2O3����D����E�Ļ�ѧ����ʽΪ4NiC2O4��2H2O +3 O2= 2Ni2O3+ 8CO2�� +8H2O��
��4��1mol Ni2O3����������ת��Ϊ2molNiO��2molNiO����������������ԭ��Ӧ�����ɵ���������Ҫ���������ʵ�����2mol��
��5����ΪNi2+�붡��ͪ뿣�C4H8N2O2�����ɵ�C8H14N4O4Ni�ʺ�ɫ�����ij��������������С���������ȷ�ȸߡ�

��ϰ��ϵ�д�
 �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�����Ŀ
 Fe3O4��4NaOH
Fe3O4��4NaOH