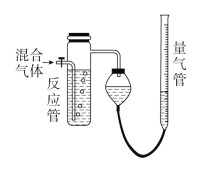
��Ŀ����
����Ŀ���ҹ�����Ϊ�˼���ȼú��ɵĴ�����Ⱦ��������ȼ��ú�����˶��ĸĽ���
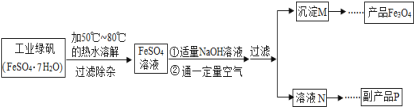
(1)Ϊ�˳�ȥú�еĺ�����ɲ���FeCl3������FeCl3��Һ��ϴú�ۣ��������·�Ӧ��FeS2��14FeCl3��8H2O=2FeSO4��13FeCl2��16HCl��Ϊ�˳������Fe2��������HCl��Ⱦ�������ù�ҵ����м���������Һ������������FeCl3�ķ�������д����һ�������йص����ӷ���ʽ��___________________________________��
(2)��һ�ַ����Dz��ù�������ȼú�м�����ʯ�ң�ʹ����ú��ȼ��ʱ���ɵ�SO2�����ݳ�������¯���У����û�ѧ����ʽ��ʾ��һ�����������̣�_______________________________��
(3)ij���в�����������������ú������ȼ�ϵ�����������������Ҫ�ɷ��DZ��飬д����ȼ�յĻ�ѧ����ʽ��________________________��
���𰸡�Fe��2H��=Fe2����H2��,2Fe2����Cl2=2Fe3����2Cl�� SO2��CaO![]() CaSO3��2CaSO3��O2
CaSO3��2CaSO3��O2![]() 2CaSO4 C3H8��5O2
2CaSO4 C3H8��5O2![]() 3CO2��4H2O
3CO2��4H2O
��������
(1)���빤ҵ����м������H��������HCl��Ⱦ����ͨ��Cl2������Fe2����
(2)������������ʯ�ҽ�SO2ת��ΪCaSO3��CaSO3�ڸ���ʱ������ΪCaSO4��
(3)C3H8ȼ�����ɶ�����̼��ˮ��
(1)���빤ҵ����м������H��������HCl��Ⱦ����ͨ��Cl2������Fe2����������Ӧ�����ӷ���ʽ�ֱ�ΪFe��2H��=Fe2����H2����2Fe2����Cl2=2Fe3����2Cl����
(2)������������ʯ�ҽ�SO2ת��ΪCaSO3��CaSO3�ڸ���ʱ������ΪCaSO4���û�ѧ����ʽ��ʾ��һ��������ΪSO2��CaO![]() CaSO3��2CaSO3��O2
CaSO3��2CaSO3��O2![]() 2CaSO4��
2CaSO4��
(3)C3H8ȼ�����ɶ�����̼��ˮ����Ӧ�Ļ�ѧ����ʽΪC3H8��5O2![]() 3CO2��4H2O��
3CO2��4H2O��

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�����Ŀ��ʵ������Ҫ����NaOH��Һ��������Һ��
������100mL 1.0mol��L-1 NaOH��Һ
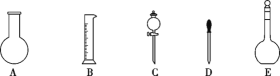
��1����ͼ��ʾ��������E������Ϊ___��������Һ�϶�����Ҫ��������___������ţ����������ӵIJ���������___�����������ƣ���
��2��������NaOH��Һʱ
�ٸ��ݼ�����������ƽ��ȡNaOH������Ϊ___g��
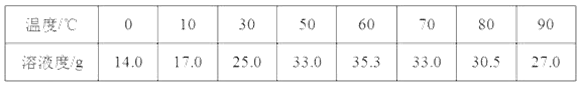
�����в�����������ҺŨ�ȵ�Ӱ����(����ƫ������ƫ��������Ӱ����)��
���� | Ũ��Ӱ�� |
����ƽ��ʹ�����룩����ʱ�����������������λ�÷ŵߵ��� | ___ |
û��ϴ���ձ��Ͳ����� | ___ |
����ʱ�����Ӷ��� | ___ |
����Eδ�����������ˮ | ___ |
������100mL 0.5mol��L-1 ������Һ
����������Ϊ98%���ܶ�Ϊ1.84g��cm-3��Ũ���������Ƹ���Һ����Ũ�������ʵ���Ũ��Ϊ___mol��L-1������Ũ��������Ϊ___mL (����������һλС��)��