��Ŀ����
 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺��1����֪��N2��g��+O2��g���T2NO��g����H=+180.5kJ/mol
N2��g��+3H2��g���T2NH3��g����H=-92.4kJ/mol
2H2��g��+O2��g���T2H2O��g����H=-483.6kJ/mol
����17g ��������������ȫ����һ�����������ˮ�������ų�������Ϊ
226.3kJ
226.3kJ
��2��ij����С���о����������������������£��ı���ʼ�����������ʵ�����N2��g��+3H2��g���T2NH3��g����Ӧ��Ӱ�죮
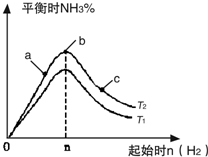
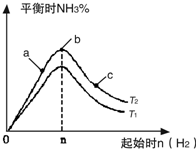
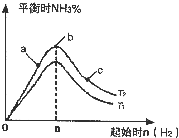
ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����
��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2
����
����
T1������ڡ��������ڡ��������ڡ�����ȷ���������ڱȽ���a��b��c����������ƽ��״̬�У���Ӧ��N2��ת������ߵ���
c
c
������ĸ����������ʼ��ϵ�м���N2�����ʵ���Ϊ
| n |
| 3 |
| n |
| 3 |
2.08
2.08
�������÷�Ӧ��298K��398Kʱ��ƽ�ⳣ���ֱ�ΪK1��K2����K1
��
��
K2���������=�����������ݶ��ڸ÷�Ӧ�йر�����ȷ����
bd
bd
��a�������������䣬��С�������ʱ����Ӧ���������淴Ӧ���ʼ�С����ƽ�⽫�����ƶ�
b�����������£�����ø���ϵ�¶Ȳ��ٸı䣬��Ӧ����ƽ��״̬
c�����������£���������������ܶȱ��ֲ��䣬��Ӧ����ƽ��״̬
d�������������䣬�������������Ϊԭ����2����������ƽ��ʱ��NH3��ƽ��Ũ�Ƚ���ԭƽ��Ũ�ȵ�һ�뻹ҪС��
��������1�����ݸ�˹��������Ӧ���ʱ䣬Ȼ����ݷ�Ӧ�ų������������ʵ��������ȣ�
��2���ٸ����¶�����ѧƽ���������ȷ�����У�
�ڸ����������������ʵ�����ѧƽ����������Ӧ�����ƶ���
�۸���a��b��c��������ʾ���������ش�
�۸��ݷ�Ӧ������ʵ���֮��Ϊ���ڻ�ѧ������֮��ʱ����ƽ��״̬ʱ������İٷֺ�������ݻ�ѧƽ��״̬�Ƿ�Ӧ���ת�������������IJ���������ش���������Ե�ƽ��Ũ�ȣ�Ȼ�����ƽ�ⳣ���ĸ������ش�
�ܸ��ݸ÷�ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�������ȵķ����ƶ���ƽ�������ƶ���ƽ�ⳣ�����٣�
��a������ѹǿ����Ӧ��������ƽ��������������ٵķ����ƶ���
b�����������£���ϵ�¶Ȳ��ٸı�����ƽ��״̬��
c�����������£�������ܶȱ���ʼ�ղ��䣬����˵����Ӧ�ﵽƽ�⣻
d������ѹǿ���٣�ƽ�����������ķ����ƶ���
��2���ٸ����¶�����ѧƽ���������ȷ�����У�
�ڸ����������������ʵ�����ѧƽ����������Ӧ�����ƶ���
�۸���a��b��c��������ʾ���������ش�
�۸��ݷ�Ӧ������ʵ���֮��Ϊ���ڻ�ѧ������֮��ʱ����ƽ��״̬ʱ������İٷֺ�������ݻ�ѧƽ��״̬�Ƿ�Ӧ���ת�������������IJ���������ش���������Ե�ƽ��Ũ�ȣ�Ȼ�����ƽ�ⳣ���ĸ������ش�
�ܸ��ݸ÷�ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�������ȵķ����ƶ���ƽ�������ƶ���ƽ�ⳣ�����٣�
��a������ѹǿ����Ӧ��������ƽ��������������ٵķ����ƶ���
b�����������£���ϵ�¶Ȳ��ٸı�����ƽ��״̬��
c�����������£�������ܶȱ���ʼ�ղ��䣬����˵����Ӧ�ﵽƽ�⣻
d������ѹǿ���٣�ƽ�����������ķ����ƶ���
����⣺��1��N2��g��+O2��g���T2NO��g����H=+180.5kJ/mol ��
N2��g��+3H2��g���T2NH3��g����H=-92.4kJ/mol ��
2H2��g��+O2��g���T2H2O��g����H=-483.6kJ/mol ��
�ɸ�˹���ɢ١�2-�ڡ�2+�ۡ�3�ã�4NH3��g��+5O2��g���T4NO��g��+6H2O��g����H=905.2kJ/mol��
��17g ��1mol��������������ȫ����һ�����������ˮ�������ų�������Ϊ
��905.2kJ=226.3 kJ��
�ʴ�Ϊ��226.3 kJ��
��2���ٷ�ӦΪ���ȷ�Ӧ���¶�����ѧƽ���������ȷ�����У���T1��T2��Ӧ�ﵪ���������ӣ���T1��T2���ʴ�Ϊ�����ڣ�
��b�����ƽ��״̬��c���ּ�������������ƽ�������ƶ���������ת�������ʴ�Ϊ��c��
�۵����������������ʵ���֮��Ϊ1��3ʱ��ƽ��״̬ʱ���İٷֺ������ƽ���ʱ����IJ������ͼʾ����ƽ��ʱ���������ʵ���Ϊn���ʼ���N2�����ʵ���Ϊ
����n=3mol��Ӧ�ﵽƽ��ʱH2��ת����Ϊ60%������ʼ����Ũ��Ϊ1��mol/l���仯������Ũ��Ϊ1.8mol/l���仯�ĵ���Ũ��Ϊ0.6mol/l��ƽ��ʱ������������������Ũ�ȷֱ���0.4mol/l��1.2mol/l��1.2mol/l����K=
=
=2.08���ʴ�Ϊ��
��2.08��
�ܸ÷�ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�������ȵķ����ƶ���ƽ�������ƶ���ƽ�ⳣ�����٣��ʴ�Ϊ������
��a�������������䣬��С�����������Ӧ����������Ũ����������Ӧ���������淴Ӧ��������ƽ�⽫����Ӧ�����ƶ�����a����
b�����������£���ϵ�¶Ȳ��ٸı�����ƽ��״̬����b��ȷ��
c�����������£�������ܶȱ���ʼ�ղ��䣬����˵����Ӧ�ﵽƽ�⣬��c����
d�������������䣬�������������Ϊԭ����2����NH3��Ũ�Ƚ�ΪԭŨ�ȵ�һ�룬�������ѹǿ���٣�ƽ�����������ķ����ƶ������淴Ӧ�����ƶ���������ƽ��ʱ��NH3��ƽ��Ũ�Ƚ���ԭƽ��Ũ�ȵ�һ�뻹ҪС����d��ȷ��
��ѡ��bd��
N2��g��+3H2��g���T2NH3��g����H=-92.4kJ/mol ��
2H2��g��+O2��g���T2H2O��g����H=-483.6kJ/mol ��
�ɸ�˹���ɢ١�2-�ڡ�2+�ۡ�3�ã�4NH3��g��+5O2��g���T4NO��g��+6H2O��g����H=905.2kJ/mol��
��17g ��1mol��������������ȫ����һ�����������ˮ�������ų�������Ϊ
| 1 |
| 4 |
�ʴ�Ϊ��226.3 kJ��
��2���ٷ�ӦΪ���ȷ�Ӧ���¶�����ѧƽ���������ȷ�����У���T1��T2��Ӧ�ﵪ���������ӣ���T1��T2���ʴ�Ϊ�����ڣ�
��b�����ƽ��״̬��c���ּ�������������ƽ�������ƶ���������ת�������ʴ�Ϊ��c��
�۵����������������ʵ���֮��Ϊ1��3ʱ��ƽ��״̬ʱ���İٷֺ������ƽ���ʱ����IJ������ͼʾ����ƽ��ʱ���������ʵ���Ϊn���ʼ���N2�����ʵ���Ϊ
| n |
| 3 |
| C2(NH3) |
| C3(H2)C(N2) |
| 1��22 |
| 1��23?0.4 |
| n |
| 3 |
�ܸ÷�ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�������ȵķ����ƶ���ƽ�������ƶ���ƽ�ⳣ�����٣��ʴ�Ϊ������
��a�������������䣬��С�����������Ӧ����������Ũ����������Ӧ���������淴Ӧ��������ƽ�⽫����Ӧ�����ƶ�����a����
b�����������£���ϵ�¶Ȳ��ٸı�����ƽ��״̬����b��ȷ��
c�����������£�������ܶȱ���ʼ�ղ��䣬����˵����Ӧ�ﵽƽ�⣬��c����
d�������������䣬�������������Ϊԭ����2����NH3��Ũ�Ƚ�ΪԭŨ�ȵ�һ�룬�������ѹǿ���٣�ƽ�����������ķ����ƶ������淴Ӧ�����ƶ���������ƽ��ʱ��NH3��ƽ��Ũ�Ƚ���ԭƽ��Ũ�ȵ�һ�뻹ҪС����d��ȷ��
��ѡ��bd��
������������Ҫ�����˸�˹���ɡ���ѧƽ�ⳣ������������Է�Ӧ���ʺ�ƽ���Ӱ�죬�Ѷ��еȣ����ݿα�֪ʶ������ɣ�

��ϰ��ϵ�д�
�����Ŀ
 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺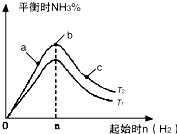 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺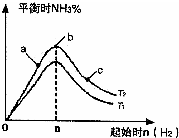 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺