��Ŀ����
8��ʵ����Ҫ��98%���ܶ�Ϊ1.84g•cm-3������������3.68mol/L��������Һ500mL����1����ȷ��ȡ98%������100.0mL��
��2��Ҫ����3.68mol/L��������Һ500mL�������õ��������ǣ���д��ţ��٢ڢܢޢ࣮
��100mL��Ͳ ���ձ� ��1000mL����ƿ ��500mL����ƿ ����ƽ ��ͷ�ι� ����ƿ �ಣ����
��3��������3.68mol/L��������Һ��������������ȷ�����������д����������ʹ�����Ƶ�������ҺŨ��ƫ�͵���ABCD��
A����ϡ�͵�������Һת��������ƿ��δϴ���ձ��Ͳ�����
B�����ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ��
C���ý�ͷ�ι�������ƿ�м�ˮʱ��Һ����������̶��ߣ���ʱ�����ý�ͷ�ιܽ�ƿ��Һ��������ʹ��Һ��Һ����̶�������
D���ý�ͷ�ι�������ƿ�м�ˮʱ�����ӹ۲���Һ��Һ��������ƿ�̶�����
E��ϴ����ȡŨ�������Ͳ������ϴ��Һת�Ƶ�����ƿ��
��4��������ʱ�����ˮ��������������ƿ�̶��ߣ���ʱʵ��Ӧ��˼������У��������ƣ�
���� ��1������ϡ��ǰ�����ʵ������������������Һ�������
��2����������һ�����ʵ���Ũ����Һ�IJ��裺�������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װƿ����ǩ��ȷ����Ҫ��������
��3������c=$\frac{n}{V}$�������������ʵ����ʵ��������Һ�������Ӱ���жϣ�
��4����������ʱ��һ�����ֵĴ�������������������ƣ�
��� �⣺��1������Ҫ98%H2SO4�����ΪVmL��������Һϡ��ǰ����������������VmL��1.84g/cm3��98%=0.5L��3.68mol/L��98g/mol�����V=100ml��������Һ�����Ϊ100.0ml��
�ʴ�Ϊ��100.0��
��2������˳���ǣ��������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װƿ����ǩ��һ������Ͳ��ȡ���õ���ͷ�ιܣ����������ձ���ϡ�ͣ���ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ת����ϣ�����������ˮϴ���ձ���������2��3�β���ϴ��Һȫ��ת�Ƶ�����ƿ�У��ټ���������ˮ������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ������ƿ�����������µߵ�ҡ�ȣ������������ձ�����������100mL��Ͳ��500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ���٢ڢܢޢࣻ
��3��A����ϡ�͵�������Һת��������ƿ��δϴ���ձ��Ͳ���������������ʵ���ʧ����Ũ��ƫ�ͣ���A��ȷ��
B�����ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�⣬�ᵼ�����ʵ���ʧ����Ũ��ƫ�ͣ���B��ȷ��
C���ý�ͷ�ι�������ƿ�м�ˮʱ��Һ����������̶��ߣ���ʱ�����ý�ͷ�ιܽ�ƿ��Һ���������������IJ�ֹ��ˮ���������ʣ���Ũ��ƫ�ͣ���C��ȷ��
D���ý�ͷ�ι�������ƿ�м�ˮʱ�����ӹ۲���Һ��Һ��������ƿ�̶����У�����Һ���ƫ��Ũ��ƫС����D��ȷ��
E��ϴ����ȡŨ�������Ͳ������ϴ��Һת�Ƶ�����ƿ�У��ᵼ�����ʵ����ʵ���ƫ�࣬Ũ��ƫ�ߣ���E����
��ѡABCD��
��4��������ʱ��һ�����ֵĴ�������������������ƣ��ʴ�Ϊ���������ƣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

 ����ѧУ�ֲ����ܲ�ϵ�д�
����ѧУ�ֲ����ܲ�ϵ�д� �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д�| A�� | Cu��Cu2+ | B�� | CO32-��CO2 | C�� | NH3��NO | D�� | MnO4-��Mn2+ |
| A�� | ��������������Ӧ�����Ȼ��ƺ���ṹ���ȶ�����ǿ����ϵ���������� | |
| B�� | ����ȼ�տɿ��ɡ����桱�������ڲ�����������ѧ�ܣ�ת��Ϊ�����ͷų��� | |
| C�� | ��������ʹ�õ���Դ�����Ԫ����չ����������Դ���ϱ��������ã��������ܺ�̫���ܾ����������Դ | |
| D�� | ����Ȳ��ܷ����ķ�Ӧ��һ�������������ķ�Ӧ |
| A�� | ����ϡ���ᷴӦ��2Fe+6H+=2Fe3++3H2�� | |
| B�� | ̼��þ�����ᷴӦ��MgCO3+2H+=Mg2++CO2��+H2O | |
| C�� | ͭ����������Һ��Ӧ��Cu+Ag+=Cu2++Ag | |
| D�� | ����þ��Һ������������Һ��Ӧ��SO42-+Ba2+�TBaSO4�� |
| A�� | v��O2����=2v��SO3���� | |
| B�� | �����������ƽ��Ħ����������ʱ����仯 | |
| C�� | ��������ݻ��ǹ̶��ģ���ô���������ܶȲ���ʱ����仯 | |
| D�� | ��λʱ��������n mol SO3��ͬʱ����n mol O2 |
| A�� | ��AgNO3 ��NaBr ��AgBr | B�� | ��AgNO3 ��NaCl ��AgCl | ||
| C�� | ��AgCl ��AgNO3 ��NaCl | D�� | ��AgNO3 ��NaI ��AgI |
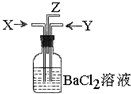 ̼��Ũ���Ṳ�Ȳ���������X��ͭ��Ũ���ᷴӦ����������Yͬʱͨ��ʢ������BaCl2��Һ��ϴ��ƿ�У���ͼװ�ã��������й�˵����ȷ���ǣ�������
̼��Ũ���Ṳ�Ȳ���������X��ͭ��Ũ���ᷴӦ����������Yͬʱͨ��ʢ������BaCl2��Һ��ϴ��ƿ�У���ͼװ�ã��������й�˵����ȷ���ǣ�������| A�� | ����ƿ�в����ij�����BaSO4 | B�� | Z���ܳ�������������CO2 | ||
| C�� | Z���ܿ�û�к���ɫ������� | D�� | ����ƿ�в����ij�����BaCO3 |