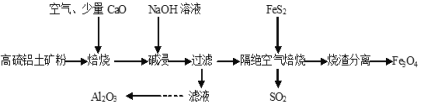
��Ŀ����
����Ŀ����������茶���[(NH4)2SO4��FeSO4��6H2O]�ֳ�Ħ���Σ��������Ҵ��������ڶ���������ijʵ��С�����ù�ҵ����м��ȡĦ���Σ����ⶨ�䴿�ȣ���������ͼ���¡�
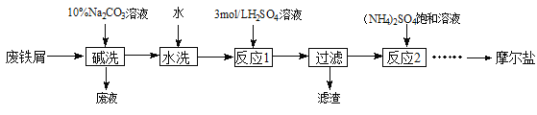
��֪������李�FeSO4��7H2O�������������ˮ�е��ܽ��
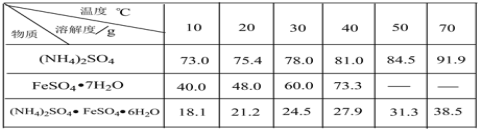
(1)��ϴ��Ŀ����_________��
(2)��Ӧ1�¶��������70~75�棬�ɲ�ȡ�Ĵ�ʩ��________��
(3)��Ӧ2�г���3 mol��L-1�����������Һ��pHΪ1��2����Ŀ����_______��
(4)�벹�������ɷ�Ӧ2������Һ�����������茶����ʵ�鷽����____�����ˡ����Ҵ�ϴ�ӡ����¸��
(5)Ϊ�ⶨ���������(NH4)2SO4��FeSO4��6H2O���崿�ȣ���������ʵ�飺
ȷ��ȡ0.4000g������������茶���������ƿ���ܽ⣬������ϡ�����ữ����0.0100 mol��L��1 ��KMnO4��Һ�ζ�����MnO![]() ǡ����ȫ����ԭΪMn2+ʱ������KMnO4��Һ�����Ϊ20.00 mL��
ǡ����ȫ����ԭΪMn2+ʱ������KMnO4��Һ�����Ϊ20.00 mL��
��ش��������⣺
�ٵζ��յ������Ϊ____________��
��������Ʒ��(NH4)2SO4��FeSO4��6H2O����(��Է�������Ϊ392)����������Ϊ______��
�������������Ļ����ϣ����д�ʩ�ܽ�һ����߲ⶨȷ�ȵ���_____������ĸ����
A���ʵ����ӳ�����Ʒ������ B�����������H2SO4��Һ
C���÷�̪��ָʾ�� D�����ʵ�飬ȡƽ��ֵ
���𰸡���ȥ���� ˮԡ���� ����ˮ�� ����Ũ������ȴ�ᾧ ��ɫ��Ϊdz��ɫ���Ұ�����ڲ��ٻָ�ԭɫ 98.0% AD
��������
����м�м���10%��̼������Һϴ��������֬��Ȼ����ˮϴ��մ����м�����̼���ƣ�����ϡ���ᣬˮԡ�����Ʊ�����������Һ��Fe+H2SO4=FeSO4+H2����Ϊ�˷�ֹ�������ӱ�������������м����������˳�����������м����Һ�м��뱥���������Һ��Ϊ����������������笠�����ˮ�⣬�����pH=1��2������Ũ������ȴ�ᾧ�����˼���Ħ���Ρ�
(1)��м�������һ���Ͳ�,̼������ҺΪ������Һ,��Ҫʹ����ϴ����м������ͣ�
�𰸣���ȥ����
(2)���µ���100�棬�ɲ�ȡ�Ĵ�ʩ��ˮԡ���ȣ���ʹ���Ⱦ��ȣ��¶��أ�
�𰸣�ˮԡ����
(3)��������狀�����������Ϊǿ�������Σ��������pHΪ1��2����ˮ�⣻
�𰸣�����ˮ��
(4)�����ܽ�ȱ���֪��������淋��ܽ�����¶����߶���������Ӧ�ò��ü���Ũ������ȴ�ᾧ�����ˡ����Ҵ�ϴ�ӡ����¸���ķ�ʽ��ã�
�𰸣�����Ũ������ȴ�ᾧ
(5)����KMnO4��Һ���еζ�������Ӧʱ���������Һ������������Ϊ�����ӣ���Ӧ�����ӷ���ʽΪ5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O���жϴﵽ�ζ��յ�������ǵ����һ�����Ը�����ر���Һ����ʱ����ƿ����Һ�ɻ�ɫ��Ϊdz��ɫ���Ұ�����ڲ��ٻָ�ԭɫ��˵���ﵽ�ζ��յ㣻���õ�ʧ��������Ƚ��м��㣻�ʵ���߳�����Ʒ�����������ݹ����ܽ�һ����߲ⶨȷ�ȣ�����ƽ������ȡƽ��ֵ�ܽ�һ����߲ⶨȷ�ȣ�
�ٻ�ɫ��Ϊdz��ɫ���Ұ�����ڲ��ٻָ�ԭɫ��
����0.4000g�����к�(NH4)2SO4��FeSO4��6H2O������Ϊxg��
���ݵ�ʧ��������ȿ�֪��
5��(NH4)2SO4��FeSO4��6H2O�ݡ�KMnO4
5��392g 1mol
xg 0.0100mol/L��20.00��10-3L
��x=0.392g
��������Ϊ![]() =98.0%��
=98.0%��
��A.�ʵ����ӳ�����Ʒ������,���ݹ����ܽ�һ����߲ⶨȷ��,��A��ȷ��
B.���������H2SO4��Һ��������ػ�����ᷴӦ����B����ȷ��
C.�÷�̪��ָʾ����������к͵ζ�ָʾ����������������ԭ��Ӧ������û��Ҫ��ָʾ������C ����ȷ;
D.���ʵ�飬ȡƽ��ֵ�ܽ�һ����߲ⶨȷ�ȣ���D��ȷ��
�𰸣���ɫ��Ϊdz��ɫ���Ұ�����ڲ��ٻָ�ԭɫ 98.0% AD

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ���±���Ԫ�����ڱ�һ���֣��г��˾���Ԫ�������ڱ��е�λ��:
�� ���� | IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
1 | �� | |||||||
2 | �� | �� | �� | �� | ||||
3 | �� | �� | �� | �� |
���û�ѧ����ش���������
(1)������Ԫ���У���������ǿ����____________��
(2)�١�����Ԫ������������Ӧ��ˮ������������ǿ����________�������ʻ�ѧʽ����д���͢������������Ӧ��ˮ���ﷴӦ�����ӷ���ʽ _______________________________��
(3)ֻ�ɢ��γɵ�18�������ĵ���ʽ _______________��
(4)д���ɢٺ͢��γɵ����ӻ�����Ļ�ѧʽ ____________________________��
(5)�õ���ʽ��ʾ�ߺ͢��γɵĻ�������γɹ���_________________________________��
(6)����Ԫ�آۺ͢ݿ��γ�һ����Է�������Ϊ66�Ĺ��ۻ�����Y��Y�����и�ԭ�Ӿ��ﵽ��8�����ȶ��ṹ����Y�Ľṹʽ _________________��
����Ŀ��ijǿ������ɫ��Һ�п��ܺ��±������е����������ӡ�
������ | Mg2+��NH4+��Ba2+��Al3+��Fe2+ |
������ | SiO32-��MnO4-��Cl-��NO3-��SO32- |
ʵ��I ��ȡ��������Һ��������ʵ�顣

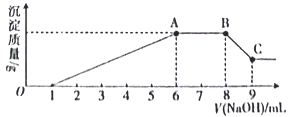
ʵ������Ϊ�˽�һ��ȷ������Һ����ɣ�ȡ100 mLԭ��Һ�������Һ�еμ�1 mol L-l�� NaOH��Һ����������������������������Һ����Ĺ�ϵ��ͼ��ʾ��
�ش��������⣺
��1��������ʵ��Ϳ����ƶϳ����ϱ��е�����һ�������ڵ���___________����
��2��ͨ��ʵ��I����ȷ������Һ��һ�����ڵ���������_____________����������X�ķ�����______________________������Z�Ļ�ѧʽΪ_________________��
��3��д��ʵ������ͼ����BC�ζ�Ӧ�����ӷ���ʽ��_____________________________________��
��4�� A���Ӧ�Ĺ�������Ϊ__________g��
��5��ͨ��������Ϣ���������Һ�������ӵ�Ũ��Ϊ______________mol L-l��